The third week of March brings a broad set of updates across GI Oncology, spanning colorectal, pancreatic, gastroesophageal, and rare gastrointestinal cancers, highlighting how clinical strategies and research approaches continue to evolve.
This week’s posts include key discussions on the ESOPEC trial in esophageal cancer and the ORCHESTRA trial in metastatic colorectal cancer, alongside insights into targeted strategies in rare pancreatic cancer and innovative approaches such as federated learning in rare tumors.
Additional highlights feature the PERISCOPE II trial in gastric cancer, AI-driven treatment selection in metastatic PDAC, and early signals from a first-line immunotherapy-only trial in MSS colorectal cancer (BBOpCo). Advances in translational research and clinical care are further reflected through organoid-based models, the perioperative FLOT plus durvalumab strategy in gastroesophageal cancer, and biomarker-driven adjuvant aspirin strategies in PIK3CA-mutated colorectal cancer.
Together, these posts underscore the shift toward precision oncology, innovative trial design, and multidisciplinary care in GI malignancies.
Kristel Muijs – Radiation Oncologist, University Medical Center Groningen (UMCG)
“The phase III ESOPEC trial comparing perioperative FLOT chemotherapy with CROSS-based neoadjuvant chemoradiotherapy has sparked important discussion in the treatment of resectable esophageal adenocarcinoma.
In our Letter to the Editor, we highlight an important aspect that may influence the interpretation of the ESOPEC findings: the quality of radiotherapy and its potential impact on outcomes.
In the ESOPEC nCRT arm, the reported pathologic complete response rate was 10%, while distant metastases occurred in 50% of patients. These outcomes appear less favorable than those observed in contemporary real-world population datasets, where pCR rates around ~18% and lower distant recurrence rates have been reported.
One key mechanism is radiation-induced lymphopenia, which has emerged as an important determinant of treatment response and long-term outcomes across thoracic malignancies, including esophageal cancer.
In our letter, we raise the possibility that radiotherapy planning approaches used in ESOPEC may have resulted in higher cardiac radiation exposure, potentially contributing to greater immune suppression and less favorable outcomes in the nCRT arm.
For this reason, we believe caution is warranted before broadly abandoning neoadjuvant chemoradiotherapy, particularly for patients with limited nodal disease or those seeking organ preservation strategies.
Continued discussion is essential as we refine multimodal treatment strategies and optimize the role of high-quality radiotherapy in esophageal cancer care.”
Arndt Vogel – Medical Oncologist, Head of the Center for Personalized Medicine, MHH at Medical University of Hanover
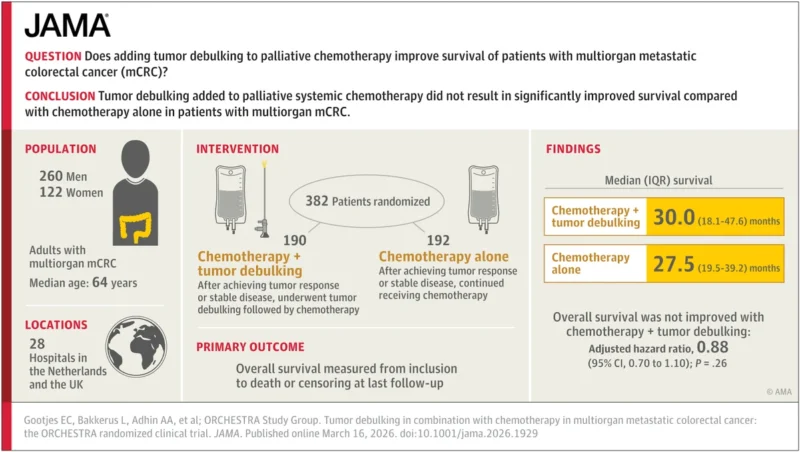
“Tumor Debulking in Combination With Chemotherapy in Multiorgan Metastatic Colorectal Cancer: The ORCHESTRA Randomized Clinical Trial
even with modern chemotherapies, tumor debulking does not add anything….”
Nelson Dusetti – Research Director, INSERM | Pancreatic Cancer & Translational Oncology | Co-founder of Predicting Med, developing transcriptomic tools for precision oncology
“We are pleased to share that our latest work is now available online in Cancer Treatment Reviews: ‘Targeted Therapeutic Strategies in Rare Subtypes of Pancreatic Cancer: Histology, Molecular Profiles, and Emerging Opportunities.’
In this article, we explore the biological and molecular heterogeneity of rare pancreatic cancer subtypes and discuss emerging targeted therapeutic approaches that may open new avenues for personalized treatment.
A special acknowledgment to Enza Scarlato and Davide Melisi for involving me in this work and for their friendly and fruitful collaboration. This publication reflects a strong collaborative effort between the Università degli Studi di Verona, the Institut Paoli-Calmettes and the CRCM – Centre de Recherche en Cancérologie de Marseille.”
Ane Appelt – Professor of Clinical Medical Physics, Rigshospitalet & Technical University of Denmark
“I’m SO pleased to share the latest work from CRUK RadNet Leeds, now published in Nature Communications.
One of the biggest challenges in modern oncology is that patient groupings are becoming smaller, not larger. As we increasingly sub-stratify disease, many patient groups effectively become ‘rare cancers’ – making it difficult to generate robust evidence using conventional approaches, such as cancer registries.
Anal cancer is a clear example. It is uncommon and geographically dispersed, with many centres only treating a few patients a year. Yet decisions still need to be made for individual patients, frequently on the basis of limited evidence.
We set out to address that problem using federated learning – enabling collaboration without sharing patient-level data.
Through the atomCAT2 consortium, we were able to:
- Link 16 international centres across nine countries
- Build the largest contemporary international anal cancer cohort to date (n = 1705)
- Develop and externally validate prognostic models across institutions
- Do all of this while keeping data securely within each centre
This approach allowed us to bypass many of the traditional barriers in multi-centre research – particularly around data governance, privacy, and data-sharing agreements – while still generating robust, generalisable insights.
Equally important to us was how this work was done. From the outset, this was designed as an open and reproducible science project:
- Pre-registration on OSF
- Prospective study protocol publication
- Pre-specified statistical analysis plan and sample size calculation
- Transparent reporting (TRIPOD)
- Open-source federated learning infrastructure and code
- Open access publication
We hope this demonstrates that large-scale, high-quality international research in rare cancers is possible – without compromising on scientific rigour or patient privacy.
(Oh, and we got a mention when House of Lords debated the Rare Cancers bill – so others seem to appreciate our efforts as well)
This has been a long and complex effort, and it simply would not have happened without the leadership and persistence of Stelios Theophanous. Also a huge thank you to the many collaborators and contributors across the atomCAT consortium, especially my co-leads Leonard Wee and Eirik Malinen.”
Giuseppe Santabarbara – Medical Oncologist, Azienda Ospedaliera di Rilievo Nazionale San Giuseppe Moscati
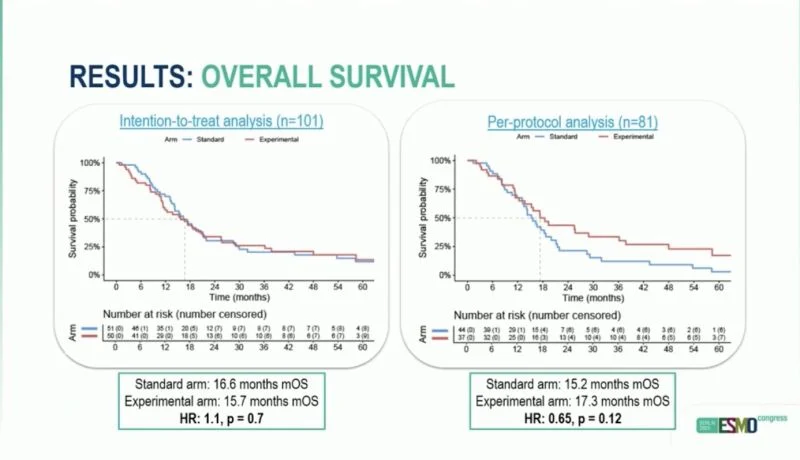
“The PERISCOPE II trial clarifies that for gastric cancer with limited peritoneal spread, the combination of gastrectomy and CRS/HIPEC does not extend survival and significantly increases the risk of severe toxicity and death. These results solidify systemic therapy alone as the standard of care for these patients.”
Federico Nichetti – MD, Medical Oncologist
“Our latest CINDERELLA study is out Leveraging on data from 41 cancer centers and 704 patients, we used Interpretable AI (IAI) to move beyond ‘one-size-fits-all’ second-line chemotherapy for metastatic PDAC.
Key Findings:
- FOLFIRINOX showed the longest survival for highly fit patients.
- Nal-IRI + 5FU/LV offers a distinct long-term benefit for specific subgroups.
- AI Optimization: Our Optimal Policy Tree (OPT) identifies the best regimen based on ECOG PS, primary tumor site, and CA19.9.
With the RASolute-302 trial potentially establishing a new second-line standard, our study provides a critical real-world benchmark.
This proof of concept demonstrates how IAI-based models can be applied to any setting to refine treatment selection and maximize patient benefit.”
Nicholas DeVito, MD – Assistant Professor of Medicine, Duke University, Division of Medical Oncology
“Our first-line immunotherapy-only trial in Stage IV MSS CRC (BBOpCo) is pushing boundaries and changing lives, but it is only the beginning. It is a foot in the door for a traditionally immunotherapy resistant disease, paving the way for novel biomarkers and combinations of tomorrow. More importantly, this achievement is proof that we can make daring but rational strides in cancer with forward thinking studies that speak directly to patients’ values and needs.”
Alexandre Bréant – GI Oncology Lead, Roche France (Squad Lead)
“A look back at the RocheOncoDays 2026 (2/3): an opening to the future of oncology
On March 6, I had the privilege of moderating the inaugural plenary session of the RocheOncoDays, dedicated to organoids. A theme that perfectly illustrates the ambition and culture of Roche in oncology: to develop cutting-edge technologies, which accelerate therapeutic research and the personalization of the care pathway, and to do so in close collaboration with the academic ecosystem.
By way of introduction, I would like to remind you of two structuring figures, which fully justify investing in the development of organoids.
In oncology,For each drug that eventually reaches patients, 20 to 30 molecules have been tested in humans, and ultimately failed
each year the industry (pharma & biotech) invests ~$80 billion in R&D
These figures should question us. At first glance, you might say to yourself, ‘Certainly, but it works, that’s the price to pay’. Perhaps, but is it sustainable?Is it sustainable, and ethical, for patients to expose them so much to molecules that happen to be ineffective or dangerous?
Is it sustainable for the industry to maintain this level of inefficiency?
Is it sustainable for the medical community and public payers, in a world where human and financial resources are increasingly constrained?
Probably not, and this therefore forces us to be more efficient in the development of therapeutic innovations.Through brilliant presentations, Lauriane Cabon of the Institute of Human Biology (research center dedicated to organoids, in which Roche has invested nearly CHF 1 billion) and Michel Ducreux of Gustave Roussy, as investigator of the organotreat trials, showed us that organoids are one way (among many others!) to help us make research more efficient and therapeutic strategies more personalized.”

Daniel Acosta Eyzaguirre – Medical Oncologist | Clinical Researcher, Drug Development | Digestive and Endocrine Tumors Unit (Gastric Cancer), Vall d’Hebron University Hospital | Vall d’Hebron Institute of Oncology (VHIO)
“A new Standard of Care for gastroesophageal cancer patients approved by EMA!
FLOT+Durvalumab in resectable gastroesophageal cancer.
This perioperative milestone represents a vital step forward in increasing cure rates for our patients.”
Caio Dabbous de Liz – Clinical Medical Oncologist
“I am pleased to share our recently published study,
‘Adjuvant Aspirin and Cyclooxygenase-2 Inhibitors in Resected, PIK3CA-Mutated Colorectal Cancer,’
a systematic review and meta-analysis of randomized controlled trials published in Critical Reviews in Oncology/Hematology.In this work, we evaluated randomized evidence on the role of NSAIDs in a molecularly selected subgroup of colorectal cancer patients harboring PIK3CA mutations.
Our findings demonstrate a consistent disease-free survival benefit with adjuvant aspirin or selective COX-2 inhibitors, supporting the biological rationale for biomarker-driven strategies in this setting.
We also provide a critical appraisal of current limitations, particularly regarding overall survival, and contextualize the results within ongoing randomized trials.
These findings contribute to the broader discussion on incorporating precision approaches into adjuvant colorectal cancer management.
Grateful to have led this work in collaboration with an excellent team.”
Find out 10 Must-Read Posts in GI Oncology from the second week of March on OncoDaily.





