On March 16, 2026, AstraZeneca announced that the European Commission had approved durvalumab (Imfinzi) plus perioperative FLOT chemotherapy for adult patients with resectable early-stage and locally advanced (Stages II, III, IVA) gastric and gastroesophageal junction (GEJ) cancers.
With this decision, durvalumab becomes the first and only perioperative immunotherapy regimen approved in this setting in the European Union.
Dave Fredrickson, Executive Vice President, Oncology Haematology Business Unit, AstraZeneca, added:
“This approval marks our third perioperative approval in Europe for an Imfinzi-based regimen, underscoring AstraZeneca’s commitment to transforming outcomes in early-stage disease, where cure is possible. For patients with early gastric and gastroesophageal cancers, this immunotherapy-based regimen delivers a durable survival benefit that increases over time.”

Durvalumab (Imfinzi): Mechanism and Background
Durvalumab (Imfinzi) is an immunotherapy developed by AstraZeneca, a global, science-led biopharmaceutical company based in Cambridge, UK.
Durvalumab is a human monoclonal antibody that binds to PD-L1 and blocks its interaction with PD-1 and CD80, helping counter tumour immune-evasion and releasing the inhibition of immune responses.
What Drove the Approval?
The approval by the European Commission followed a positive opinion from the Committee for Medicinal Products for Human Use (CHMP) and was based on results from the Phase III MATTERHORN trial, which evaluated perioperative durvalumab in combination with FLOT chemotherapy compared with FLOT alone in patients with resectable gastric and gastroesophageal junction cancers.
Imfinzi and FLOT chemotherapy is also approved in the US and other countries based on the MATTERHORN results.
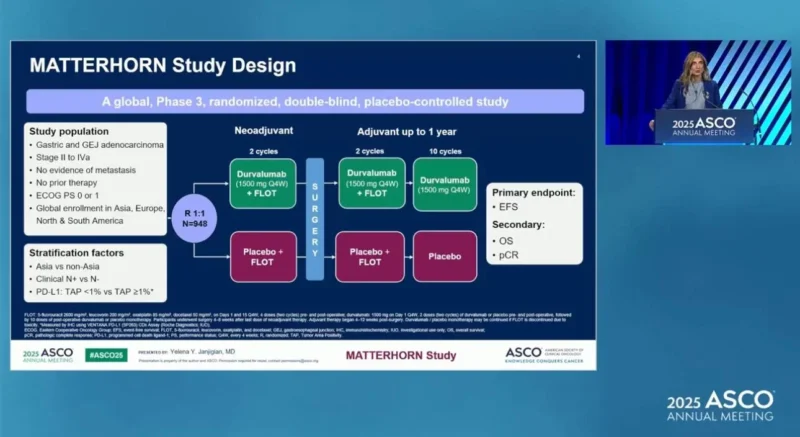
MATTERHORN Trial Design
MATTERHORN (NCT04592913) is a large, global, randomized, double-blind, placebo-controlled Phase III study designed to assess whether the addition of durvalumab to standard FLOT chemotherapy could improve outcomes in patients with previously untreated, resectable gastric or GEJ adenocarcinoma. The trial was led by Dr. Yelena Y. Janjigian (Memorial Sloan Kettering Cancer Center) in collaboration with an international network of investigators across Europe, Asia, and North America.
A total of 948 patients with Stage II–IVA disease were randomized 1:1. Patients received durvalumab plus FLOT chemotherapy for two cycles prior to surgery, followed by postoperative treatment on the same perioperative schedule, or placebo plus FLOT chemotherapy following the same schedule.
The treatment strategy incorporated immunotherapy across both neoadjuvant and adjuvant phases, reflecting a comprehensive perioperative approach.
The primary endpoint was event-free survival (EFS), with overall survival (OS) and pathologic complete response (pCR) among the key secondary endpoints.
Interim MATTERHORN Results from ASCO 2025
The planned interim analysis of MATTERHORN was presented during the ASCO 2025 Plenary Session and published in The New England Journal of Medicine, with a median follow-up of 31.5 months.
This interim analysis provided the first clear signal that adding durvalumab could meaningfully improve disease control in the perioperative setting. Patients treated with durvalumab plus FLOT experienced a significant reduction in the risk of recurrence, progression, or death, along with improvements in early survival outcomes.
Key findings included:
- EFS: 29% reduction in risk (HR 0.71; 95% CI 0.58–0.86; p<0.001)
- 2-year EFS: 67.4% vs 58.5%
- 2-year OS: 75.7% vs 70.4%
- pCR rate: 19.2% vs 7.2%
A time-dependent analysis of overall survival suggested that the benefit became more pronounced after the first year of treatment, supporting the hypothesis of a delayed but durable immunotherapy effect.
From a surgical and safety standpoint, the addition of durvalumab did not compromise treatment delivery. Rates of grade 3–4 adverse events were nearly identical between groups, and there were no meaningful differences in surgical delays or initiation of postoperative therapy.
Final MATTERHORN Results
The final analysis, presented by Professor Josep Tabernero at the ESMO Congress 2025 in Berlin, provided the definitive survival data that supported regulatory approvals.
These results confirmed that the early benefits observed in the interim analysis translated into a significant and durable overall survival advantage. The addition of durvalumab to perioperative FLOT reduced the risk of death by 22%, with consistent benefit observed across clinically relevant subgroups.
Key outcomes included:
- OS: HR 0.78 (95% CI 0.63–0.96)
- 3-year OS: 69% vs 62%
- Benefit observed independent of PD-L1 expression
In addition to survival, the final dataset highlighted deeper pathological responses. Patients receiving durvalumab were more likely to achieve nodal clearance (ypN0 58.2% vs 44.8%), supporting a deeper pathological response overall. Importantly, improvements in event-free survival were consistent across all levels of pathological response, suggesting that the benefit of immunotherapy extends beyond traditional response categories.
Consistency Across Analyses
Across interim and final analyses, the efficacy results remained highly consistent:
- EFS hazard ratio: 0.71
- OS hazard ratio: 0.78
- Pathologic complete response: 19.2% vs 7.2%
Taken together, the data demonstrate that adding durvalumab to perioperative chemotherapy enhances tumor response, reduces recurrence risk, and improves survival outcomes in resectable gastric and GEJ cancers.
Notably, the overall survival benefit was observed regardless of tumour PD-L1 status, supporting the broad applicability of this treatment approach.
Perioperative Treatment Strategy
The approved regimen integrates immunotherapy across the full perioperative continuum. Patients receive durvalumab in combination with FLOT chemotherapy before surgery, followed by additional treatment after resection, and then continue with durvalumab monotherapy.
Specifically, the approach includes two cycles of durvalumab plus chemotherapy prior to surgery and two cycles after surgery, followed by durvalumab monotherapy. This strategy reflects a growing emphasis on enhancing systemic disease control both before and after surgery in order to reduce recurrence risk.
Safety and Surgical Feasibility
The safety profile of durvalumab in combination with FLOT chemotherapy was consistent with the known safety profiles of each agent. No new safety signals were identified.
Rates of grade 3 or higher adverse events were similar between the durvalumab and control arms (71.6% vs 71.2%), indicating that the addition of immunotherapy did not substantially increase severe toxicity. Importantly, the proportion of patients who were able to proceed to surgery was comparable between groups, supporting the feasibility of incorporating durvalumab into perioperative treatment without compromising surgical outcomes.
Clinical Context: Persistent Risk of Recurrence
Gastric cancer remains a major global health burden, ranking among the leading causes of cancer-related mortality worldwide, with nearly one million new cases diagnosed annually.
Even in patients with resectable disease, recurrence is frequent. Approximately one in four patients relapse within the first year after surgery, and long-term survival remains limited, with fewer than half of patients alive at five years. These challenges underscore the need for more effective perioperative strategies.
Expert Perspective
Josep Tabernero, MD, PhD, head of the Medical Oncology Department at Vall d’Hebron University Hospital and director of the Vall d’Hebron Institute of Oncology (VHIO) in Barcelona, Spain, and principal investigator of the trial, emphasized the clinical relevance of these findings:

“Despite curative-intent surgery and chemotherapy, patients with resectable gastric and gastroesophageal cancers still face high recurrence rates and an urgent need for improved long-term survival. In MATTERHORN, nearly 70 per cent of patients were still alive three years after treatment with the durvalumab-based perioperative regimen. This EU approval brings patients the first immunotherapy regimen to extend survival in this early setting and is poised to become the new standard of care.”
Conclusion
The EU approval of durvalumab in combination with perioperative FLOT chemotherapy establishes a new treatment paradigm for resectable gastric and GEJ cancers. By demonstrating improvements in both event-free survival and overall survival—without compromising safety or surgical feasibility—this regimen represents a significant advance in the early-stage setting.
More broadly, it reflects an ongoing shift toward integrating immunotherapy earlier in the course of disease, where the opportunity for durable benefit and potential cure is greatest.
The full announcement is available on the official AstraZeneca website.