March 2026 marked a significant period of progress in esophageal, gastric and gastroesophageal junction cancers, with advances spanning regulatory approvals, clinical trials, real-world evidence, guideline updates, and evolving biomarker-driven strategies.
A major milestone was the European Commission approval of perioperative durvalumab plus FLOT, introducing the first and only perioperative immunotherapy regimen in this setting in the EU. At the same time, emerging data from studies such as ILUSTRO, MOONLIGHT, and KEYNOTE-585 further refined the role of chemoimmunotherapy across disease stages. Novel treatment approaches, including zolbetuximab- and camrelizumab-based strategies, continue to expand options in advanced disease.
In parallel, updated clinical guidelines and expert insights from NCCN 2026 highlight the increasing importance of histology, tumor location, and biomarker status—including HER2, PD-L1, MSI/MMR, and CLDN18.2—in guiding treatment decisions. Real-world data on early-onset disease and meta-analyses evaluating surrogate endpoints such as pathologic complete response (pCR) provide additional context for clinical outcomes and trial design.
This article highlights 10 key updates from March 2026 that are shaping the current and future landscape of gastric and esophageal cancer management.
EU Approval: Durvalumab + FLOT in Resectable Gastric and GEJ Cancers
On March 16, 2026, the European Commission approved durvalumab (Imfinzi) in combination with perioperative FLOT chemotherapy for adult patients with resectable Stage II–IVA gastric and gastroesophageal junction (GEJ) cancers.
This is the first and only approved perioperative immunotherapy regimen in this setting in the EU, based on results from the phase III MATTERHORN trial (The New England Journal of Medicine).
MATTERHORN Trial
MATTERHORN is a randomized, double-blind phase III trial (n=948) evaluating perioperative durvalumab plus FLOT versus chemotherapy alone. The primary endpoint was event-free survival (EFS), with overall survival (OS) as a key secondary endpoint.
Results
The durvalumab-based regimen demonstrated statistically significant and clinically meaningful improvements in both endpoints:
- EFS: HR 0.71 (95% CI 0.58–0.86; p<0.001)
- OS: HR 0.78 (95% CI 0.63–0.96; p=0.021)
- 3-year OS: 69% vs 62%
- Benefit was observed regardless of PD-L1 status.
- Grade ≥3 adverse events were similar between arms (71.6% vs 71.2%), with no new safety signals.
Takeaway
Perioperative durvalumab plus FLOT significantly improved EFS and OS, establishing a new standard of care in resectable gastric and GEJ cancers.
Read about Imfinzi approval in the EU as First Perioperative Immunotherapy for Early Gastric and GEJ Cancers on OncoDaily.
Zolbetuximab + mFOLFOX6 + Nivolumab in CLDN18.2-Positive Gastric and GEJ Cancer
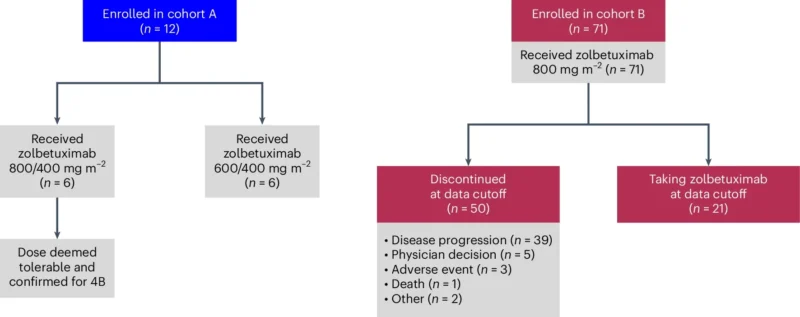
Published on March 16, 2026, in Nature Medicine, Shitara et al. reported results from cohort 4 of the global, open-label, phase II ILUSTRO trial evaluating first-line zolbetuximab plus mFOLFOX6 and nivolumab in patients with HER2-negative, locally advanced unresectable or metastatic CLDN18.2-positive gastric or gastroesophageal junction (GEJ) adenocarcinoma.
Study Design
Cohort 4 included a safety lead-in phase (4A) and an expansion phase (4B). A total of 77 patients were treated across cohorts 4A and 4B, while primary efficacy analyses were conducted in cohort 4B. The main efficacy endpoint for cohort 4 was investigator-assessed progression-free survival (PFS) per RECIST v1.1.
Results
In cohort 4B:
- Median PFS: 14.8 months (95% CI, 8.3–not estimable)
- Median PFS in CLDN18.2-high tumors: 18.0 months (95% CI, 11.1–not estimable)
- ORR in patients with measurable disease: 62.1% (95% CI, 48.4–74.5)
- ORR in CLDN18.2-high tumors: 68.1% (95% CI, 52.9–80.9)
Median OS was 18.0 months, but OS data were immature at the time of analysis.
Safety
Across cohorts 4A and 4B, grade 3 or higher treatment-emergent adverse events occurred in 66.2% of patients. The most common treatment-emergent adverse events were nausea (80.5%) and decreased appetite (72.7%).
Takeaway
First-line zolbetuximab plus mFOLFOX6 and nivolumab showed promising activity with manageable toxicity in CLDN18.2-positive, HER2-negative gastric and GEJ adenocarcinoma, supporting further evaluation in the ongoing phase III LUCERNA trial.
Metastatic Gastric Cancer: Evolving Toward Biomarker-Informed Treatment
Published on March 10, 2026, in Nature Reviews Clinical Oncology, Joan Choo et al. reviewed current strategies and emerging therapies in metastatic gastric cancer.
First-line treatment remains based on fluoropyrimidine–platinum chemotherapy, with therapeutic benefit increasingly shaped by biomarker-informed use of immune checkpoint inhibitors and targeted therapies in molecularly defined subgroups, including MSI, PD-L1-expressing, HER2-positive, and CLDN18.2-positive disease.
Key Highlights
- Treatment is increasingly guided by biomarkers
- Fluoropyrimidine–platinum chemotherapy remains the first-line backbone
- Durable responses remain uncommon, and resistance is frequent
- Emerging strategies include ADCs, bispecific antibodies, and cellular therapies
- ctDNA and theranostics may improve patient selection and disease monitoring
Takeaway
Metastatic gastric cancer care is becoming increasingly biomarker-informed, but resistance to current first-line therapies remains a major challenge, highlighting the need for next-generation treatment strategies.
MOONLIGHT Trial: Chemoimmunotherapy Strategies in Advanced Gastroesophageal Adenocarcinoma
Published on February 27, 2026, in Nature Communications, Lorenzen et al. reported results from the phase II MOONLIGHT trial evaluating different chemoimmunotherapy strategies in previously untreated advanced or metastatic HER2-negative gastric and gastroesophageal junction (GEJ) adenocarcinoma.
Study Design
MOONLIGHT (NCT03647969) is an investigator-initiated, multicenter, open-label phase II trial (n=262) with randomized mFOLFOX-based cohorts and a non-randomized cohort evaluating FLOT plus nivolumab. The primary endpoint was progression-free survival (PFS).
Key Highlights
- Adding ipilimumab to nivolumab + mFOLFOX did not improve outcomes vs mFOLFOX alone
- Median PFS: 5.8 vs 6.6 months; median OS: 10.1 vs 12.5 months
- Sequential chemotherapy followed by dual immunotherapy showed inferior outcomes (PFS 4.0 months; OS 7.6 months)
- Non-randomized FLOT + nivolumab cohort showed promising activity (PFS 7.0 months; OS 14.6 months; ORR 56%)
- Higher toxicity observed with dual checkpoint blockade (grade ≥3 AEs: 74%)
Takeaway
Adding CTLA-4 inhibition to nivolumab-based chemotherapy increased toxicity without improving efficacy, while FLOT plus nivolumab demonstrated encouraging activity, requiring confirmation in randomized trials.
Read more about MOONLIGHT Trial on OncoDaily.
Biomarker-Guided Immunotherapy in Gastric Cancer: Current Insights and Future Perspectives
Published in April 2026 in Cancer Treatment Reviews, Mulet et al. reviewed the current immunotherapeutic landscape in gastric cancer, established and emerging predictive biomarkers, and future perspectives for biomarker-guided treatment.
Immunotherapy has improved outcomes in selected patients, particularly in biomarker-defined subgroups such as MSI-H/dMMR and PD-L1-positive disease, but responses remain highly heterogeneous.
Key Highlights
- Chemo-immunotherapy has become standard of care for selected advanced gastric cancer patients
- MSI-H/dMMR is the most robust predictive biomarker for immunotherapy benefit
- PD-L1 expression remains a suboptimal biomarker
- Most patients derive limited long-term benefit from ICIs
- Emerging approaches include gene expression and epigenetic profiling, liquid biopsy, gut microbiome characterization, and AI-driven multimodal algorithms
Takeaway
Biomarker-guided immunotherapy is central to modern gastric cancer care, but more robust predictive biomarkers are needed to improve patient selection and outcomes.
French Guidelines 2026: Updated Management of Gastric and GEJ Adenocarcinoma
Published on March 24, 2026, in the European Journal of Cancer, Zaanan et al. presented updated French Intergroup clinical practice guidelines for the diagnosis, staging, treatment, and follow-up of gastric and gastroesophageal junction (GEJ) adenocarcinoma.
These recommendations are based on multidisciplinary consensus and evidence available up to January 2026.
Key Highlights
- Initial staging should include CT imaging, endoscopy, and selective use of EUS and laparoscopy
- Perioperative FLOT remains the standard of care for ≥cT2 and/or cN+ disease
- Addition of durvalumab to perioperative FLOT was associated with improved survival, with early access granted in France
- First-line metastatic treatment is based on platinum–fluoropyrimidine chemotherapy
- Treatment selection is guided by biomarkers, including HER2, PD-L1, MSI, and CLDN18.2
Takeaway
The updated French guidelines reinforce perioperative FLOT as standard in localized disease and highlight the expanding role of immunotherapy and biomarker-driven strategies in advanced gastric and GEJ cancer management.
NCCN 2026: Evolving Treatment Landscape in Gastric, Esophageal, and EGJ Cancers
Presented at the NCCN Annual Conference 2026, Mary F. Mulcahy outlined the evolving management of gastric, esophageal, and esophagogastric junction (EGJ) cancers, emphasizing the growing role of histology, tumor location, and biomarker status in treatment selection.
Key Highlights
- Treatment decisions are guided by HER2, PD-L1, MSI/MMR, and CLDN18.2
- Approximately 20% HER2 discordance between primary and metastatic tumors supports repeat biopsy
- Perioperative FLOT (± durvalumab) is central in localized adenocarcinoma
- Preoperative chemoradiation remains standard for esophageal squamous cell carcinoma
- Adjuvant nivolumab improves disease-free survival, without statistically significant overall survival benefit
- Immunotherapy benefit increases with higher PD-L1 expression in advanced disease
- Zolbetuximab-based therapy expands options in CLDN18.2-positive tumors
Takeaway
Management of gastroesophageal cancers is increasingly driven by tumor biology and biomarkers, with treatment strategies tailored by histology, molecular profile, and disease setting.
Read more about NCCN Annual Conference 2026 on OncoDaily.
Early- versus Late-Onset Gastroesophageal Cancer: Real-World Outcomes from a Central European Cohort
Published on March 27, 2026, in Gastric Cancer, Sokop et al. reported results from a retrospective real-world study comparing clinicopathological features and outcomes in early-onset (EO, <50 years) versus late-onset (LO, ≥50 years) gastroesophageal cancer.
Study Design
This single-center cohort study included 1,377 patients treated between 2010 and 2022, of whom 161 (11.7%) had EO disease. Treatment patterns and survival outcomes were evaluated in patients with adenocarcinoma.
Key Highlights
- EO patients more frequently presented with gastric primaries (63%), poorly cohesive adenocarcinoma (45%), and metastatic disease at diagnosis (55%)
- EO disease was associated with ECOG performance status 0 more often, lower BMI, and higher rates of current smoking (44%)
- In the first-line setting, triplet chemotherapy was more frequently used in EO adenocarcinoma patients (29% vs 11%; p<0.001)
- In inoperable or metastatic disease, no significant survival difference was observed between EO and LO patients (median OS 9.1 vs 9.6 months; p=0.913)
- Hereditary cancer syndrome was confirmed in 4.3% of EO cases
Takeaway
Early-onset gastroesophageal cancer was associated with distinct and more aggressive clinicopathological features, but without a significant survival difference compared with late-onset disease.
KEYNOTE-585: Pathologic Endpoints and Survival in Perioperative Pembrolizumab
Published in March 2026 in ESMO Open, Shitara et al. reported a post hoc analysis of the phase III KEYNOTE-585 trial evaluating the association between early pathologic endpoints and survival outcomes in resectable gastric and gastroesophageal junction (GEJ) adenocarcinoma.
Study Design
KEYNOTE-585 is a randomized, double-blind, phase III trial (n=1007) comparing perioperative pembrolizumab plus chemotherapy versus placebo plus chemotherapy in locally advanced gastric or GEJ adenocarcinoma. This post hoc analysis assessed associations between pathologic complete response (pCR), major pathologic response (mPR), and pathologic downstaging (pDS) with event-free survival (EFS) and overall survival (OS).
Key Highlights
- Pembrolizumab plus chemotherapy increased pathologic response rates vs chemotherapy alone (pCR 13.9% vs 2.8%)
- Higher rates of mPR (31.5% vs 22.2%) and pathologic downstaging were observed
- Patients achieving pCR, mPR, or pDS had numerically longer EFS and OS
- Among patients with mPR: EFS HR 0.6 (95% CI 0.4–1.0), OS HR 0.7 (95% CI 0.4–1.2)
- The parent KEYNOTE-585 trial did not demonstrate superiority for EFS
Takeaway
Early pathologic responses were associated with numerically improved survival outcomes; however, these findings are exploratory and do not establish validated surrogate endpoints for survival in perioperative immunotherapy for gastric and GEJ adenocarcinoma.
Read about KEYNOTE-585 Trial on OncoDaily.
Camrelizumab + CAPOX in Advanced Gastric and GEJ Cancer: Phase III Results
Published on March 12, 2026, in The BMJ, Peng et al. reported results from a randomized phase III trial evaluating camrelizumab plus CAPOX followed by camrelizumab-based maintenance versus CAPOX alone in HER2-negative, unresectable, locally advanced or metastatic gastric or gastroesophageal junction (GEJ) adenocarcinoma.
Study Design
A total of 885 patients were randomized (2:2:1) to camrelizumab + CAPOX → camrelizumab + apatinib, CAPOX alone, or camrelizumab + CAPOX → camrelizumab. The primary endpoint was overall survival (OS) for camrelizumab + CAPOX → camrelizumab + apatinib versus CAPOX.
Key Highlights
- OS improved with camrelizumab + CAPOX → camrelizumab + apatinib vs CAPOX
- PD-L1–positive: 15.0 vs 12.5 months (HR 0.80)
- Overall population: 13.5 vs 12.1 months (HR 0.80)
- Camrelizumab maintenance also showed longer OS vs CAPOX (descriptive)
- No added benefit with apatinib
Safety
Grade ≥3 adverse events were higher with apatinib-containing maintenance (67.9% vs 45–47%).
Takeaway
Camrelizumab plus CAPOX followed by camrelizumab-based maintenance improved overall survival versus chemotherapy alone, while adding apatinib increased toxicity without additional survival benefit.
Find out Top 10 Colorectal Cancer Updates – February 2026 on OncoDaily.