In resectable gastric and gastroesophageal junction (GEJ) adenocarcinoma, prior studies in the neoadjuvant chemotherapy setting have reported associations between pathologic response or downstaging and survival outcomes. With immune checkpoint inhibitors now incorporated into perioperative strategies, it remains uncertain whether early pathologic endpoints can meaningfully reflect long-term survival outcomes in this setting.
A post hoc analysis of the phase III KEYNOTE-585 trial evaluated whether pathologic complete response (pCR), major pathologic response (mPR), and pathologic downstaging (pDS) after neoadjuvant/adjuvant pembrolizumab plus chemotherapy were associated with event-free survival (EFS) and overall survival (OS).
This study was published in March 2026 in ESMO Open (Volume 11, Issue 3; Article 106090).
Title: Surrogate endpoints for survival in KEYNOTE-585: neoadjuvant/adjuvant pembrolizumab plus chemotherapy versus placebo plus chemotherapy for gastric or gastroesophageal junction adenocarcinoma
Authors: K. Shitara, Y.-J. Bang, L.S. Wyrwicz, S.Y. Rha, T. Oshima, F. Pietrantonio, Y.-K. Park, S. Lonardi, P. Yañez, C.-J. Yen, J.-P. Metges, M. Garrido, M. Moehler, S. Pelles-Avraham, W.P. Yong, A. Spallanzani, E. Jensen, R. Krishnan, C.-S. Shih, and S.-E. Al-Batran.
Background
In the original randomized phase III KEYNOTE-585 trial, neoadjuvant and adjuvant pembrolizumab plus chemotherapy was not superior to placebo plus chemotherapy for EFS in participants with locally advanced gastric or GEJ adenocarcinoma. However, pembrolizumab plus chemotherapy significantly improved pCR compared with placebo plus chemotherapy (difference 10.9%, 95% CI 7.5–14.8; P < 0.00001).
In light of these findings, the present post hoc analysis examined whether early pathologic endpoints were associated with survival outcomes in the intention-to-treat (ITT) population.
Read about Immunotherapy Success Rate for Gastric Cancer on OncoDaily.
Methods
KEYNOTE-585 (NCT03221426) was a multicenter, double-blind, randomized, placebo-controlled phase III trial in untreated, locally advanced gastric or GEJ adenocarcinoma (including Siewert type 2 or 3) scheduled for surgery after preoperative chemotherapy; eligible participants had a T3 or greater primary lesion or any positive nodes, without evidence of metastatic disease.
Participants were randomized 1:1 to receive:
- Pembrolizumab plus chemotherapy
- Placebo plus chemotherapy
In the main cohort, chemotherapy consisted of cisplatin-based regimens (XP or FP). A smaller safety cohort received FLOT. For this post hoc analysis, outcomes are reported in the intention-to-treat (ITT) population, with results reflecting the main and FLOT cohorts combined.
The post hoc analysis evaluated associations between the following pathologic endpoints and survival outcomes:
- Pathologic complete response (pCR)
- Major pathologic response (mPR; ≤10% residual viable tumor, Mandard grade 1–2)
- Pathologic downstaging (pDS), including nodal downstaging to N0 and any tumor or nodal pDS
- Survival outcomes included EFS (per RECIST v1.1, investigator assessed) and OS.
The data cut-off date was February 9, 2023. Median time from random assignment to database cut-off was 46.3 months (range 24.2–63.5).
Results
A total of 1007 participants were randomized: 502 to pembrolizumab plus chemotherapy and 505 to placebo plus chemotherapy. Baseline characteristics were reported as largely balanced between treatment groups.
Pathologic outcomes were as follows:
- pCR: 13.9% with pembrolizumab plus chemotherapy vs 2.8% with placebo plus chemotherapy
- mPR: 31.5% vs 22.2%
- Tumor pDS: 46.6% vs 34.9%
- Nodal pDS: 40.4% vs 28.9%
- Any tumor or nodal pDS: 58.0% vs 47.7%
Pathologic nodal stage N0 was observed in 221/502 (44.0%) in the pembrolizumab arm and 172/505 (34.1%) in the placebo arm. Across the ITT population, participants who achieved pCR, mPR, nodal pDS to N0, or any tumor or nodal pDS were observed to have numerically longer EFS and OS compared with participants who did not achieve these outcomes. Sample sizes were inadequate for definitive findings, and these analyses were exploratory.
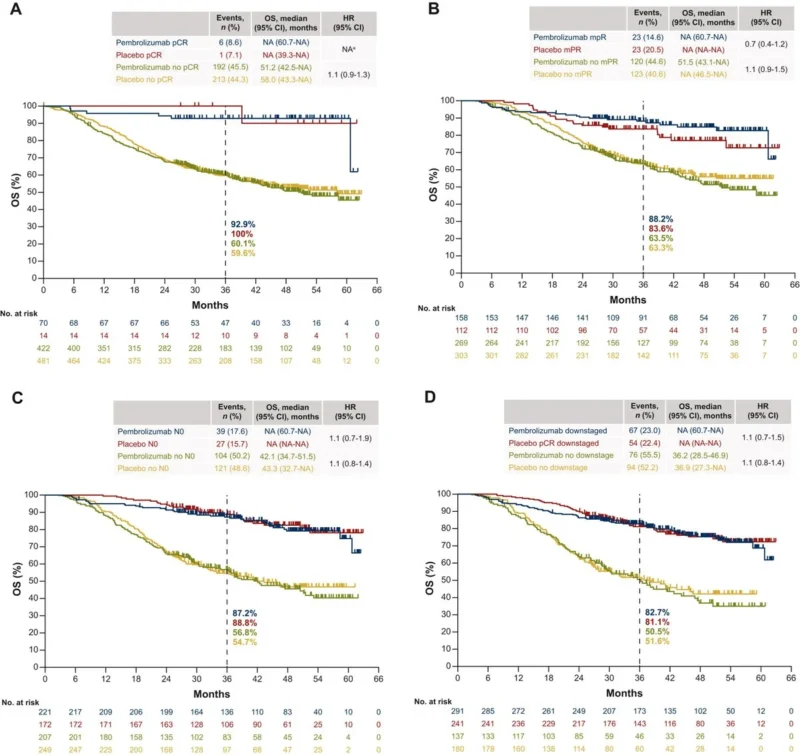
Among participants who achieved mPR, the hazard ratio comparing pembrolizumab plus chemotherapy versus placebo plus chemotherapy was:
- EFS HR: 0.6 (95% CI 0.4–1.0)
- OS HR: 0.7 (95% CI 0.4–1.2)
These analyses were exploratory and were not adjusted for multiplicity.
Interpretation
This post hoc analysis suggests a potential association between achieving pCR, mPR, or pDS and numerically longer event-free survival (EFS) and overall survival (OS) in patients with locally advanced resectable gastric or GEJ adenocarcinoma. Pembrolizumab plus chemotherapy was associated with higher rates of these pathologic endpoints compared with placebo plus chemotherapy.
However, the analysis was exploratory in nature, and the parent KEYNOTE-585 trial was not superior for EFS. Therefore, these findings do not establish validated surrogate endpoints for survival.
Conclusion
In this post hoc analysis of KEYNOTE-585, participants with locally advanced resectable gastric or GEJ adenocarcinoma who achieved pCR, mPR, or pathologic downstaging after neoadjuvant/adjuvant therapy were observed to have numerically prolonged event-free and overall survival compared with those who did not achieve these endpoints.
Pembrolizumab plus chemotherapy was associated with higher rates of these pathologic responses compared with placebo plus chemotherapy. However, these findings are exploratory and do not establish validated surrogate endpoints for survival.
Further validation through additional analyses and future studies will be required to clarify the potential role of early pathologic endpoints in predicting long-term outcomes in perioperative immunotherapy for gastric and GEJ adenocarcinoma.
Find out the full article on ESMO Open.