Advances in immunotherapy have reshaped the treatment landscape for advanced gastroesophageal adenocarcinoma (GEA). The addition of PD-1 blockade to platinum-based chemotherapy has improved survival in patients with HER2-negative metastatic gastric and gastroesophageal junction cancers, establishing chemoimmunotherapy as a modern first-line standard. However, long-term outcomes remain limited for many patients, and researchers continue to explore strategies that could further enhance treatment efficacy.
The MOONLIGHT trial was designed to investigate whether intensifying immune checkpoint blockade or modifying the chemotherapy backbone could improve outcomes in this disease.
The results of this study were published on February 27, 2026, in Nature Communications.
Title: First-line modified FOLFOX plus/minus nivolumab and ipilimumab or FLOT plus nivolumab in advanced gastroesophageal adenocarcinoma: a phase II multi-cohort trial
Authors: Sylvie Lorenzen, Thorsten Oliver Goetze, Peter C. Thuss-Patience, Jorge Riera-Knorrenschild, Eray Goekkurt, Tobias Nicolaas Dechow, Thomas Jens Ettrich, Ralf Dieter Hofheinz, Kim Barbara Luley, Daniel Pink, Udo Lindig, Gunnar Folprecht, Gunter Schuch, Michael Bitzer, Volker Heinemann, Stefan Angermeier, Claus Bolling, Maria Loose, Sabine Junge, Claudia Pauligk, Salah-Eddin Al-Batran.
Read more about the Immunotherapy Success Rate for Gastric Cancer on OncoDaily.
Study Design
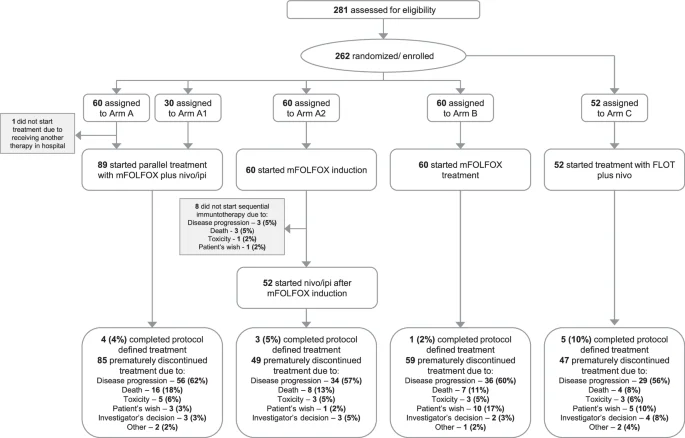
MOONLIGHT (NCT03647969) was an investigator-initiated, multicenter, open-label phase II trial with randomized cohorts followed by a non-randomized arm, conducted at 27 sites in Germany.
Eligible patients had previously untreated advanced or metastatic HER2-negative gastric or gastroesophageal junction adenocarcinoma, ECOG performance status ≤1, and adequate hematologic, hepatic, and renal function. Enrollment occurred independent of PD-L1 expression and microsatellite instability status, although PD-L1 expression was assessed centrally using immunohistochemistry with the 28-8 antibody clone.
Between November 2018 and February 2022, 281 patients were screened, and 262 patients were enrolled in the study.
Patients received one of four treatment strategies:
• Arm A/A1: mFOLFOX + nivolumab + ipilimumab administered concurrently
• Arm A2: three cycles of mFOLFOX induction followed by nivolumab + ipilimumab
• Arm B: mFOLFOX chemotherapy alone
• Arm C: FLOT chemotherapy combined with nivolumab (single-arm cohort)
The primary endpoint was progression-free survival (PFS). Secondary endpoints included overall survival (OS), objective response rate (ORR), and safety.
Baseline characteristics were generally balanced across treatment arms. Median age ranged between 58 and 63 years, approximately two-thirds of patients were male, and about one-third had undergone prior resection of the primary tumor. PD-L1 CPS ≥1 was reported in 38–47% of patients in the mFOLFOX-based arms and 60% in the FLOT plus nivolumab cohort.
Results
Dual checkpoint blockade plus chemotherapy
A central question of the MOONLIGHT trial was whether adding CTLA-4 inhibition (ipilimumab) to nivolumab and chemotherapy could improve treatment outcomes.
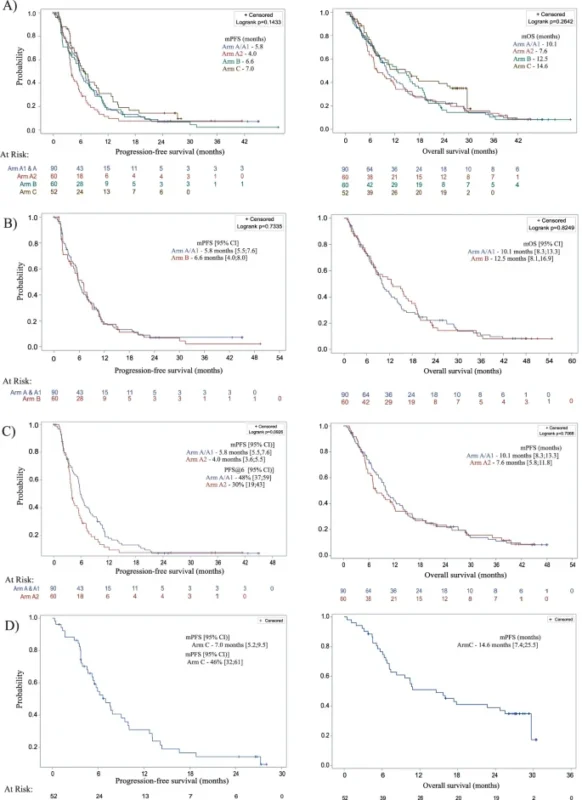
However, outcomes with mFOLFOX combined with nivolumab and ipilimumab were largely comparable to those observed with mFOLFOX alone. The 6-month PFS rate was 48% with chemo-immunotherapy versus 47% with chemotherapy alone. Median progression-free survival was 5.8 months in the chemo-immunotherapy arm compared with 6.6 months in the chemotherapy arm, while median overall survival was 10.1 months versus 12.5 months, respectively.
Objective response rates were also similar between the two groups (46% vs 47%), with complete responses observed in six patients in Arm A/A1 and three patients in Arm B. Analyses according to PD-L1 expression did not demonstrate a clear advantage for adding CTLA-4 blockade.
Sequential chemotherapy followed by immunotherapy
The study also examined whether a strategy of short induction chemotherapy followed by dual checkpoint blockade could maintain disease control while limiting chemotherapy exposure.
In Arm A2, patients received three cycles of mFOLFOX before transitioning to nivolumab plus ipilimumab. Outcomes in this sequential arm were less favorable compared with concurrent chemo-immunotherapy. Median PFS was 4.0 months, median overall survival was 7.6 months, and the objective response rate was 32%.
Importantly, 13% of patients discontinued treatment during or shortly after the induction chemotherapy phase, meaning they never initiated immunotherapy.
FLOT plus nivolumab
A separate cohort explored the combination of triplet FLOT chemotherapy with nivolumab.
Although this arm was non-randomized, the regimen was feasible and showed promising efficacy in this phase II study. Patients treated with FLOT plus nivolumab achieved a median PFS of 7.0 months and a median overall survival of 14.6 months.
Key efficacy outcomes included:
• Objective response rate: 56%
• Complete response rate: 10%
Outcomes appeared more favorable among patients with PD-L1 CPS ≥1, where median OS reached 16.7 months, compared with 10.5 months in CPS-negative tumors. Because this cohort was small and non-randomized, comparisons across treatment arms should be interpreted descriptively.
Safety
Treatment-related adverse events were observed across all treatment groups, although toxicity differed between strategies.
The highest toxicity occurred in the dual checkpoint blockade plus chemotherapy arm, where 74% of patients experienced grade ≥3 treatment-related adverse events, compared with 45% in both the sequential treatment arm and the chemotherapy-alone arm.
Across the study, the most commonly reported adverse events included anorexia, constipation, diarrhea, fatigue, nausea, peripheral sensory neuropathy, and vomiting.
In the FLOT plus nivolumab cohort, 67% of patients experienced grade ≥3 treatment-related adverse events.
Conclusion
Adding ipilimumab to nivolumab-based chemotherapy increased toxicity but did not improve efficacy compared with mFOLFOX chemotherapy alone. The strategy of short induction chemotherapy followed by dual checkpoint blockade was associated with inferior disease control.
In contrast, the exploratory cohort combining FLOT chemotherapy with nivolumab was feasible and showed promising activity, although confirmation in randomized trials is needed.
The full article is available in Nature Communications.



