February 2026 brought important developments across the colorectal cancer spectrum. New data included long-term results from the phase III CONVERT trial in MRI-selected locally advanced rectal cancer, real-world evidence supporting trifluridine/tipiracil plus bevacizumab in metastatic CRC, and screening outcomes from the large Swedish SCREESCO randomized trial.
In metastatic disease, liver metastases were identified as a key negative factor for immune checkpoint inhibitor outcomes in MSS mCRC. The U.S. FDA granted traditional approval to encorafenib-based triplet therapy in first-line BRAF V600E–mutant mCRC and Fast Track designation to pelareorep in second-line KRAS-mutant MSS disease.
Additional updates addressed subsequent primary cancer risks in non-hereditary CRC survivors, initiation of the phase III JSKN003 trial in HER2-positive advanced CRC, phase II results of cabozantinib in refractory MSS mCRC (ABACO), and inferior post-metastasectomy outcomes in early-onset CRC compared with average-onset disease.
Together, these updates reflect steady progress toward more precise, biomarker-driven colorectal cancer care.
CONVERT Trial: CAPOX Alone vs ChemoRT in MRI-Selected Locally Advanced Rectal Cancer
Published on February 19, 2026, in the Journal of Clinical Oncology, the phase III CONVERT trial compared neoadjuvant CAPOX (nCT) versus capecitabine-based chemoradiotherapy (nCRT) in locally advanced rectal cancer with uninvolved mesorectal fascia on MRI.
A total of 589 treated patients were analyzed (nCT, n = 300; nCRT, n = 289), with a median follow-up of 48 months. The primary endpoint was 3-year locoregional recurrence-free survival (LRRFS).
Results (3-year)
- LRRFS: 97.4% (nCRT) vs 96.3% (nCT); HR 1.40 (95% CI, 0.53–3.68)
- DFS: 87.9% vs 89.2%; HR 0.88 (95% CI, 0.54–1.44)
- OS: 94.1% vs 95.0%; HR 0.86 (95% CI, 0.42–1.76)
Noninferiority of CAPOX was not confirmed, as the upper bound of the CI exceeded the prespecified margin, although local recurrence rates were very low in both arms.
Safety
- Grade 2–4 long-term adverse events were significantly lower with nCT (16.0% vs 26.3%, P = 0.002).
- Proctitis was also less frequent with nCT (33.6% vs 41.7%, P = 0.049).
CONVERT suggests comparable survival with less long-term toxicity using CAPOX alone in MRI-selected LARC, but formal noninferiority was not established.
Read about Final Phase 3 CONVERT Trial Results on OncoDaily.
Trifluridine/Tipiracil ± Bevacizumab: Real-World Evidence
Published on February 24, 2026, in NEJM Evidence, this U.S.-based real-world study evaluated trifluridine–tipiracil (FTD–TPI) with or without bevacizumab in metastatic colorectal cancer (mCRC), following the survival benefit observed in the phase III SUNLIGHT trial.
Using the ConcertAI RWD360 dataset, adults with mCRC were grouped by first exposure to FTD–TPI alone or FTD–TPI plus bevacizumab. After propensity score matching, 472 patients were included in each cohort with balanced baseline characteristics.
Results
- Median real-world overall survival (rwOS):
8.9 months with FTD–TPI + bevacizumab vs 5.8 months with FTD–TPI (P < 0.001) - Median real-world time to treatment discontinuation (rwTTD):
3.5 vs 2.2 months - Median real-world time to next treatment or death (rwTTNTD):
4.9 vs 3.5 months
Takeaway
In routine U.S. clinical practice, FTD–TPI combined with bevacizumab was associated with significantly longer real-world survival compared with FTD–TPI alone, supporting and extending the survival benefit previously demonstrated in the SUNLIGHT trial.
SCREESCO Trial Updates: Colonoscopy and FIT vs Usual Care in CRC Screening
Published February 20, 2026, in Nature Medicine, the Swedish SCREESCO randomized controlled trial compared once-only primary colonoscopy or two rounds of low-cutoff fecal immunochemical testing (FIT) versus usual care (no screening invitation) in 278,280 60-year-olds in a screening-naïve setting. Median follow-up during the diagnostic phase was 4.8 years.
Overall CRC incidence
- Colonoscopy IRR 1.08 (95% CI, 0.91–1.28);
- FIT IRR 0.92 (95% CI, 0.81–1.05).
Stage shift
- Stage I–II CRC was more frequently detected with colonoscopy (IRR 1.38, 95% CI 1.09–1.74) and numerically higher with FIT (IRR 1.19, 95% CI 0.99–1.43).
- Stage III–IV CRC was lower with FIT (IRR 0.71, 95% CI 0.58–0.86) and directionally lower with colonoscopy (IRR 0.86, 95% CI 0.67–1.11).
Safety
Gastrointestinal and cardiovascular events were slightly increased during the first year after randomization, then converged toward control rates. FIT×2 was associated with higher venous thromboembolism and bleeding-related GI events. All-cause mortality was similar across groups.
Read more about SCREESCO Trial on OncoDaily.
Liver Metastases and ICI Outcomes in MSS mCRC
A study published on February 18, 2026 in Cancer Research Communications evaluated the impact of liver metastases on outcomes in patients with microsatellite-stable/proficient MMR (MSS/pMMR) metastatic colorectal cancer treated with immune checkpoint inhibitor (ICI)–based therapies.
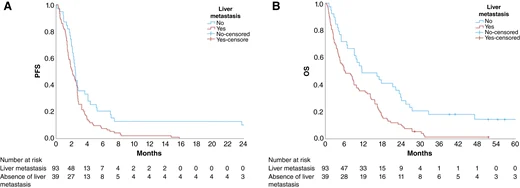
This retrospective cohort from Mass General Brigham included 132 MSS/pMMR mCRC patients; 93 (70.5%) had liver metastases at ICI initiation. The primary endpoint was progression-free survival (PFS).
Key results (no liver mets vs liver mets at ICI start):
- Clinical benefit rate: 46.2% vs 16.1% (P = 0.001)
- Median PFS: 2.5 vs 2.1 months (HR 1.68, P = 0.009)
- 12-month PFS: 12.8% vs 1.1% (P = 0.034)
- Median OS: 11.5 vs 6.2 months (HR 2.03, P < 0.001)
In univariable and multivariable analyses, no history of liver metastases was an independent favorable factor for both PFS and OS.
Takeaway
Liver metastases are associated with inferior outcomes to ICI-based regimens in MSS/pMMR mCRC, supporting liver metastasis status as a key stratification factor in this setting.
FDA Approval: BRAFTOVI Combination Regimen in 1L Metastatic Colorectal Cancer
On February 24, 2026, the U.S. FDA granted traditional approval to encorafenib (Braftovi) in combination with cetuximab and fluorouracil-based chemotherapy for adult patients with metastatic colorectal cancer with a BRAF V600E mutation, as detected by an FDA-authorized test. (Encorafenib previously received accelerated approval in 2024 in combination with cetuximab and mFOLFOX6 for this setting.)
Approval was based on the phase III BREAKWATER trial (NCT04607421) in previously untreated BRAF V600E–mutant mCRC.
Phase III BREAKWATER (Arm B vs Arm C)
Encorafenib + cetuximab + mFOLFOX6 was compared with standard chemotherapy (mFOLFOX6/FOLFOXIRI/CAPOX ± bevacizumab).
- Median PFS: 12.8 vs 7.1 months, HR 0.53 (95% CI, 0.41–0.68); p < 0.0001
- Median OS: 30.3 vs 15.1 months, HR 0.49 (95% CI, 0.38–0.63); p < 0.0001
- ORR: 61% vs 40% (p = 0.0008)
Cohort 3 (FOLFIRI Backbone)
Encorafenib + cetuximab + FOLFIRI demonstrated:
- ORR: 64% vs 39% with FOLFIRI ± bevacizumab, p = 0.0011
Takeaway
BREAKWATER confirms substantial improvements in PFS, OS, and response rate with encorafenib-based triplet therapy in first-line BRAF V600E–mutant mCRC, establishing a new standard of care in this historically poor-prognosis molecular subset.
Read about Full FDA Approval of BRAFTOVI Combination Regimen on OncoDaily.
FDA Grants Fast Track to Pelareorep in 2L KRAS-Mutant MSS mCRC
On February 4, 2026, Oncolytics Biotech announced that the U.S. FDA granted Fast Track Designation to pelareorep in combination with bevacizumab and FOLFIRI for the treatment of patients with KRAS-mutant, microsatellite-stable (MSS) metastatic colorectal cancer (mCRC) in the second-line setting.
This designation addresses a population with significant unmet need, as MSS mCRC derives minimal benefit from immunotherapy and outcomes after first-line progression remain poor.
Clinical Data Supporting Fast Track
Pelareorep plus standard-of-care demonstrated:
- ORR: 33% (vs ~10% historically)
- Median PFS: 16.6 months (vs ~5.7 months historically)
- Median OS: 27 months (vs ~11.2 months historically)
Pelareorep is an investigational intravenously administered double-stranded RNA immunotherapeutic agent designed to stimulate innate and adaptive immune responses.
A controlled study in second-line KRAS-mutant MSS mCRC comparing standard-of-care ± pelareorep is planned, with first site activation expected in March 2026.
Takeaway
Fast Track Designation reflects encouraging early activity in a difficult-to-treat MSS mCRC subgroup. Randomized data will determine its ultimate clinical role.
Subsequent primary cancer risks for non-hereditary colorectal cancer survivors
A study published in eClinicalMedicine (February 2026, Volume 92) evaluated risks of subsequent primary cancers (SPCs) among colorectal cancer (CRC) survivors without a known hereditary predisposition, excluding individuals with pathogenic germline variants in APC, MUTYH, DNA mismatch repair genes (MLH1, MSH2, MSH6, PMS2), or EPCAM.
The analysis included 7,202 CRC survivors recruited (1998–2012) via cancer registries in Australia, Canada, and the United States, followed to December 2022 (mean follow-up 10.6 years).
Key results (SIR, 95% CI)
- Any SPC (excluding skin cancer): 1.04 (0.98–1.11) — no overall increase
- Subsequent primary CRC: 1.34 (1.14–1.57)
- Hematopoietic cancers: 2.49 (1.92–3.21)
- Liver cancer: 2.25 (1.51–3.36)
- Thyroid cancer: 1.90 (1.20–3.02)
- Age effect: CRC diagnosed <50 had higher SPC risk (1.43, 1.25–1.64), whereas those diagnosed ≥50 did not (p < 0.001 for difference).
Takeaway
In CRC survivors without an identified hereditary syndrome, overall SPC risk is not elevated, but risks are higher for metachronous CRC and selected extracolonic sites (blood/hematopoietic, liver, thyroid). Early-onset CRC survivorsshow higher overall risk and may warrant targeted surveillance.
JSKN003 Phase III Trial in HER2-Positive CRC
On February 14, 2026, Alphamab Oncology reported that the first patient was dosed in JSKN003-005, a randomized, open-label, controlled, multicenter Phase III study in HER2-positive advanced colorectal cancer (CRC).
JSKN003 is described by the company as a biparatopic HER2 antibody–drug conjugate (ADC). The trial compares JSKN003 versus investigator’s choice of later-line therapies: regorafenib, fruquintinib, or trifluridine–tipiracil.
The primary endpoint is progression-free survival (PFS) assessed by blinded independent central review (BICR) using RECIST v1.1.
Alphamab also cited a company-reported dataset presented at ESMO 2025: in 32 patients with HER2-positive advanced CRC, JSKN003 achieved an objective response rate (ORR) of 68.8% and a median PFS of 11.04 months.
Why it matters
In advanced CRC, later-line options such as regorafenib, fruquintinib, and trifluridine–tipiracil are widely used, but median PFS is generally measured in months and objective responses are typically limited, supporting evaluation of more active HER2-directed approaches in a biomarker-selected setting.
Phase 2 ABACO trial: Cabozantinib in pretreated metastatic colorectal cancer
Published in ESMO Gastrointestinal Oncology (March 2026), the single-center, open-label, single-arm Phase II ABACO trial (Italy) evaluated cabozantinib in patients with microsatellite-stable (MSS) metastatic colorectal cancer who had progressed after fluoropyrimidines, oxaliplatin, irinotecan, anti-VEGF therapy, and anti-EGFR agents when appropriate.
Patients received cabozantinib 60 mg orally once daily. The trial followed a Simon two-stage design, with ≥30% PFS at 16 weeks predefined as clinically meaningful.
Efficacy (n = 33):
- 16-week PFS rate: 33% (11/33) — primary endpoint met
- Median PFS: 2.27 months (95% CI 1.71–3.65)
- Median OS: 6.25 months (95% CI 3.81–10.26)
- ORR: 3% (1 partial response); disease control rate: 45.5%
No significant correlation was observed between PFS and RAS/BRAF status, sidedness, or clinical features.
Grade ≥3 adverse events occurred in 17% of patients; dose reductions were required in ~40%, with no unexpected safety signals.
Exploratory analyses showed that absence of TP53 mutations and tumor mutational burden ≥4 mut/Mb were associated with improved PFS. RNA sequencing demonstrated a predominance of CMS4 biology, and tumors with prolonged benefit showed higher EMT and angiogenesis pathway activation.
Takeaway
In heavily pretreated MSS mCRC, cabozantinib achieved durable disease control in approximately one-third of patients at 16 weeks, with exploratory molecular signals suggesting potential biologic enrichment strategies.
Early-Onset CRC and Liver Metastasectomy Outcomes
Published February 16, 2026, in Clinical Colorectal Cancer, this multicenter Italian study examined outcomes of liver metastasectomy in early-onset colorectal cancer (EO-CRC, ≤50 years) versus average-onset CRC (AO-CRC, >50 years) with liver-limited metastatic disease.
Among 782 patients with liver metastases, EO-CRC had significantly worse outcomes after resection.
Key results:
- Median OS: 44.0 vs 64.0 months (p < 0.0001)
- Median PFS: 13.0 vs 17.0 months (p < 0.0001)
- Survival gap persisted in RAS-mutated and RAS/BRAF wild-type subgroups
Multivariable analysis confirmed early-onset disease as an independent adverse prognostic factor. Molecular profiling showed higher TP53 alterations and lower APC mutation rates in EO-CRC, with increased ARID1A and CTNNB1 alterations.
Despite aggressive treatment and higher resection rates, EO-CRC patients experienced inferior outcomes, supporting a distinct and more aggressive biological phenotype.
Find out Top 10 Pancreatic Cancer Updates – January 2026 on OncoDaily.