SCREESCO trial evaluated two real-world colorectal cancer screening strategies—once-only primary colonoscopy and repeated fecal immunochemical testing (FIT) with colonoscopy reserved for positive results—against usual care with no screening invitation, aiming to clarify both the potential benefits of earlier cancer detection and the short-term harms linked to the diagnostic workup in a population-based setting in Sweden.
Title: Colonoscopy and fecal immunochemical testing versus usual care in diagnostic colorectal cancer screening: the SCREESCO randomized controlled trial
Authors: Marcus Westerberg, Jonas F. Ludvigsson, Chris Metcalfe, Ulf Strömberg, Johannes Blom, Lars Engstrand, Mikael Hellström, Christian Löwbeer, Robert Steele, Lars Holmberg & Anna Forsberg
Background
Colorectal cancer (CRC) screening is recommended in many guidelines because it can detect cancer earlier and remove precancerous lesions, but every screening strategy also has tradeoffs—especially procedure-related harms. While colonoscopy is often viewed as the most “definitive” test, many organized programs rely on fecal immunochemical testing (FIT) repeated every 1–2 years, followed by colonoscopy only after a positive result.
A key evidence gap has been how these approaches compare with usual care (no screening invitation) in a randomized setting, not only for cancer detection patterns but also for real-world short- and medium-term adverse events. The Swedish SCREESCO randomized controlled trial was designed to quantify both benefits and harms by comparing once-only primary colonoscopy and two rounds of low-cutoff FIT against usual care during the “diagnostic phase,” when screening procedures and downstream workups occur.
Methods
SCREESCO (Screening of Swedish Colons; ClinicalTrials.gov NCT02078804) used Swedish population and healthcare registers to track outcomes for all randomized individuals, regardless of whether they actually participated in screening (an intention-to-screen approach). The analysis focused on the diagnostic phase (2014–2020), evaluating:
- CRC incidence (overall and by stage), including cancers detected by screening and those diagnosed outside the trial through routine clinical care
- Adverse events—specifically gastrointestinal (GI) and cardiovascular events recorded in healthcare registers
- All-cause mortality during follow-up, as a broad safety signal.
CRC staging was categorized primarily as stage I–II versus stage III–IV, using data from national cancer and quality registers (with chart review for trial-detected cases to confirm diagnosis/stage when needed). Comparative effects were expressed as incidence rates per 100,000 person-years and incidence rate ratios (IRRs) with 95% confidence intervals (CIs).
Study Design
This was a large, pragmatic, population-based randomized trial in Sweden across 18 of 21 regions (screening-naïve setting for the study regions). A randomized block method (unmasked, due to the nature of screening) allocated 278,280 individuals aged 60 years to:
- Primary colonoscopy (once-only invitation)
- FIT×2: two rounds of two-stool FIT performed 2 years apart, using an unusually low positivity cutoff of 10 μg hemoglobin per gram feces (positive if either sample met cutoff), with colonoscopy after a positive FIT
- Usual care control (no screening invitation; routine symptom-driven evaluation and existing surveillance pathways).
Final analytic population (after excluding individuals discovered post-randomization to have died before allocation or to have prevalent CRC, and two unidentifiable controls) included 278,051 unique participants:
- 31,113 in the colonoscopy arm
- 60,267 in the FIT×2 arm
- 186,671 colonoscopy-controls, of whom 120,521 served as controls for the FIT×2 comparison (randomized in the same period as FIT×2).
Participation reflected real-world uptake: 35% underwent colonoscopy in the colonoscopy arm (median time to colonoscopy 349 days, IQR 253–446), and 55% returned at least one FIT in the FIT×2 arm (median time to first FIT 338 days, IQR 229–395). Baseline demographics and comorbidities were well balanced between each intervention arm and its control group; notably, 92% had no prior GI or cardiovascular event history.
Results of SCREESCO Trial
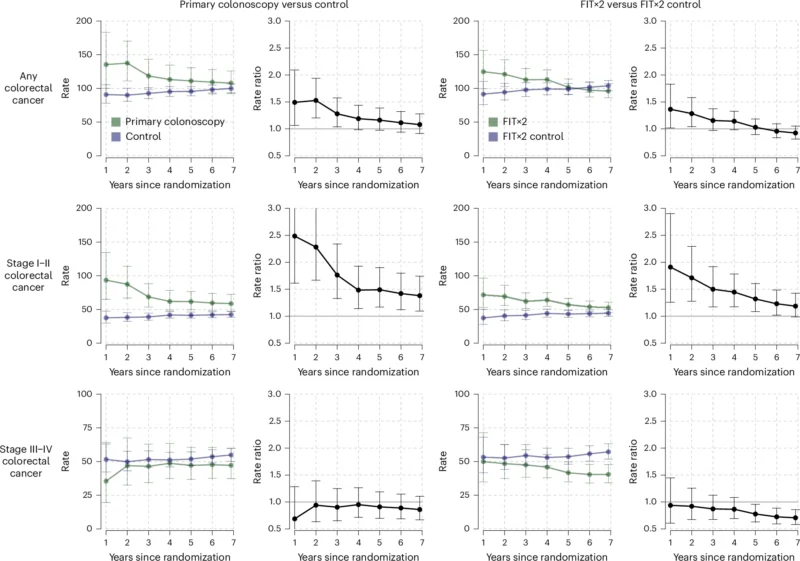
After a median follow-up of 4.8 years (maximum 6.9 years), overall CRC incidence did not show a clear reduction versus usual care during this early diagnostic window, but there were important shifts in when and which stage cancers were diagnosed.
Overall CRC incidence (per 100,000 person-years):
- Colonoscopy vs control: 107.9 vs 99.9; IRR 1.08 (95% CI 0.91–1.28).
- FIT×2 vs control: 96.0 vs 103.9; IRR 0.92 (95% CI 0.81–1.05).
Cumulative incidence proportions at end of follow-up were:
- Colonoscopy: 0.69% (95% CI 0.66–0.71) vs control 0.72% (0.71–0.73).
- FIT×2: 0.61% (0.60–0.63) vs control 0.73% (0.72–0.75).
Stage I–II CRC (earlier-stage disease):
- Colonoscopy: 58.7 vs 42.5 per 100,000 person-years; IRR 1.38 (95% CI 1.09–1.74).
- FIT×2: 52.7 vs 44.4; IRR 1.19 (95% CI 0.99–1.43).
This pattern indicates a stage shift toward earlier detection, particularly strong for colonoscopy invitation. Importantly, incidence differences were most pronounced around year 1 after randomization, aligning with the timing of most screening procedures.
Stage III–IV CRC (later-stage disease):
- Colonoscopy: IRR 0.86 (95% CI 0.67–1.11) vs control (directionally lower).
- FIT×2: IRR 0.71 (95% CI 0.58–0.86) vs control (significantly lower).
Cumulative curves suggested later-stage cancers became less frequent in intervention arms after roughly 4 years, especially in the FIT×2 group—consistent with earlier detection and possible prevention effects beginning to emerge, even if a net incidence reduction is not yet visible at <5 years.
How many cancers were screen-detected?
Among CRCs diagnosed in the intervention arms, 32% (colonoscopy arm) and 38% (FIT×2 arm) were classified as screen-detected. Very few CRCs were diagnosed outside the trial among those who had completed screening procedures: 9 individuals (0.03%) after screening colonoscopy in the colonoscopy arm and 8 (0.01%) in the FIT×2 arm.
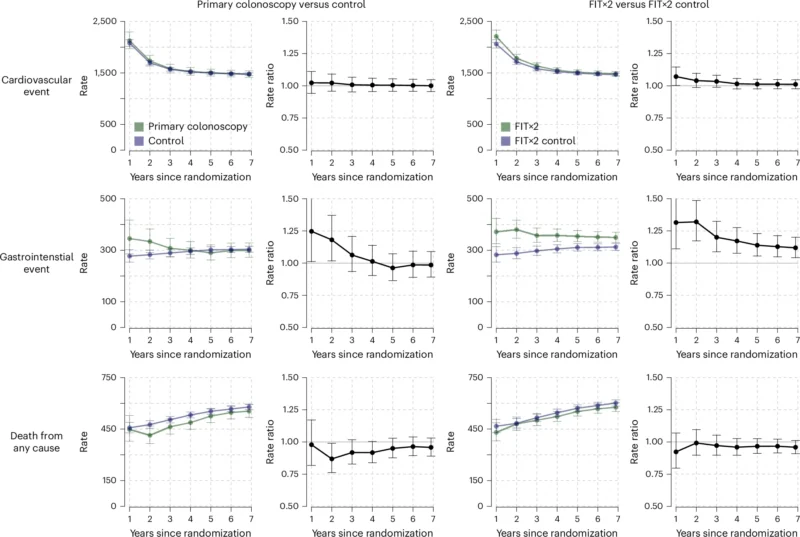
Safety and adverse events
Across both intervention strategies, GI and cardiovascular event rates were slightly higher during the first year and then became more similar to controls later on. At the end of follow-up:
- Cardiovascular events: essentially identical in colonoscopy vs control (1,475.8 per 100,000 person-years in both; IRR 1.00, 95% CI 0.96–1.05).
- In the FIT×2 arm, venous thromboembolism was higher than controls: 60.1 vs 43.3; IRR 1.39 (95% CI 1.16–1.66).
- GI events remained somewhat higher in the FIT×2 arm, mainly bleeding outcomes: iatrogenic bleeding IRR 1.18 (95% CI 1.05–1.32) and unspecified GI bleeding IRR 1.14 (95% CI 1.04–1.26).
A previously reported key safety anchor from SCREESCO was that serious adverse events directly linked to screening colonoscopies were 0.2%, including 2 bowel perforations and 15 major bleedings.
All-cause mortality:
- Colonoscopy: 554.6 vs 579.0 per 100,000 person-years; IRR 0.96 (95% CI 0.89–1.03).
- FIT×2: 577.1 vs 601.8; IRR 0.96 (95% CI 0.91–1.01).
Key findings
- Invitation to screening increased early-stage CRC diagnoses, especially with primary colonoscopy (stage I–II IRR 1.38), showing a clear stage shift versus usual care.
- Later-stage CRC became less common over time, with the strongest signal in the FIT×2 arm (stage III–IV IRR 0.71).
- Harms were concentrated early: GI and cardiovascular events rose slightly during the first year, then aligned more closely with controls.
- FIT×2 showed specific safety signals, including higher venous thromboembolism and bleeding-related GI events compared with controls.
No excess all-cause mortality was observed in either screening strategy during this diagnostic-phase follow-up.
Conclusion
During the diagnostic phase of the Swedish SCREESCO randomized trial, invitation to primary colonoscopy or two-round, low-cutoff FIT increased detection of earlier-stage CRC compared with usual care and was accompanied by slightly higher adverse event rates in the first year, followed by convergence toward control rates over time.
The emerging reduction in stage III–IV CRC, particularly with FIT×2, suggests potential longer-term benefit that may become clearer with extended follow-up. The trial’s definitive endpoint—CRC mortality—will be reported in the planned final analysis with follow-up through 31 December 2030, which will ultimately determine the net life-saving impact of these screening strategies.


