Neoadjuvant chemoradiotherapy (nCRT) followed by total mesorectal excision (TME) has long been a standard strategy for locally advanced rectal cancer (LARC). However, pelvic radiotherapy can add treatment burden—both during therapy and long after it ends—through bowel and pelvic toxicities that may affect quality of life. This has fueled interest in radiation-sparing approaches for selected patients, especially those without high-risk MRI features. The CONVERT trial was designed to test whether neoadjuvant chemotherapy (nCT) with CAPOX alone could be an alternative to capecitabine-based nCRT in MRF-uninvolved LARC.
The final results of the phase III CONVERT trial were published as an Original Report in the Journal of Clinical Oncology on February 19, 2026. The paper reports long-term oncologic outcomes with a median follow-up of 48 months, alongside a detailed comparison of short- and long-term toxicities.
Title: Neoadjuvant Chemotherapy With CAPOX Versus Chemoradiation for Locally Advanced Rectal Cancer With Uninvolved Mesorectal Fascia (CONVERT): Final Results of a Phase III Trial
Authors: Wei-Jian Mei; Xiao-Zhong Wang; Xuan Zhang; Yue-Ming Sun; Chun-Kang Yang; Jun-Zhong Lin; Zu-Guang Wu; Rui Zhang; Wei Wang; Yong Li; Ye-Zhong Zhuang; Jian Lei; Xiang-Bin Wan; Ying-Kun Ren; Yong Cheng; Wen-Liang Li; Zi-Qiang Wang; Dong-Bo Xu; Xian-Wei Mo; Hai-Xing Ju; Sheng-Wei Ye; Jing-Lin Zhao; Hong Zhang; Yuan-Hong Gao; Zhi-Fan Zeng; Wei-Wei Xiao; Xiao-Peng Zhang; Yun-Feng Li; E Xie; Yi-Fei Feng; Jing-Hua Tang; Xiao-Jun Wu; Gong Chen; Li-Ren Li; Zhen-Hai Lu; De-Sen Wan; Jin-Xin Bei; Zhi-Zhong Pan; Jie-Hai Yu; Pei-Rong Ding.
Methods
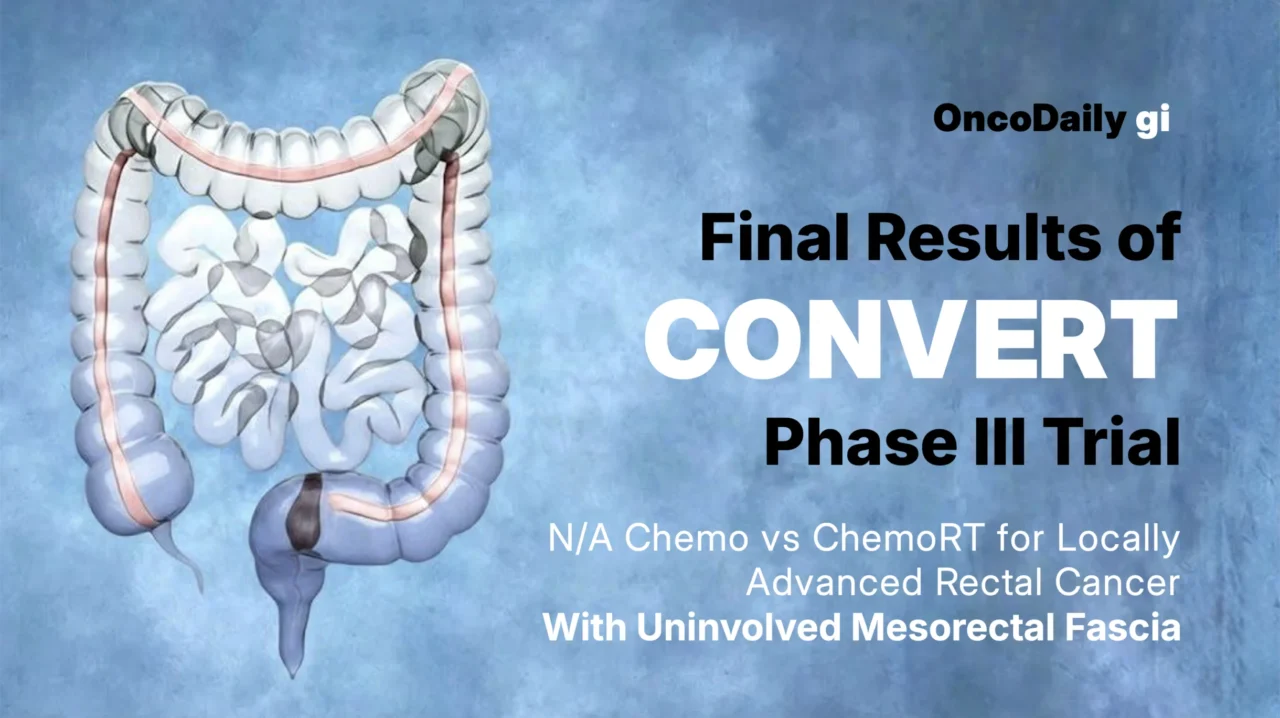
CONVERT was a phase III, open-label, multicenter, randomized noninferiority trial conducted across 21 hospitals in China (ClinicalTrials.gov: NCT02288195). Eligible patients were 18–75 years old with previously untreated, pathologically confirmed LARC located within 12 cm from the anal verge and with uninvolved MRF. Patients had to have ECOG performance status ≤1 and adequate organ function. Key exclusions included cT4b disease, tumors adjacent to the MRF, symptomatic bowel obstruction, prior pelvic radiotherapy or chemotherapy, or another invasive malignancy within 5 years.
Baseline staging included contrast-enhanced CT of the chest/abdomen and pelvic MRI (mandatory unless contraindicated), with endoscopic ultrasound (EUS) used for staging support. Patients were randomized 1:1 with stratification by tumor location and clinical nodal stage.
Treatment arms
- Neoadjuvant chemotherapy (nCT) arm: four cycles of CAPOX (oxaliplatin 130 mg/m² day 1 plus capecitabine 1,000 mg/m² twice daily days 1–14, every 3 weeks), followed by restaging. Patients without progression proceeded to TME 2–4 weeks later. Patients with local progression could receive chemoradiation before surgery. Postoperative chemoradiation was recommended if surgical margins contained microscopic or macroscopic disease. Four cycles of adjuvant CAPOX were recommended.
- Neoadjuvant chemoradiotherapy (nCRT) arm: capecitabine 825 mg/m² twice daily concurrent with radiotherapy (5 days/week for 5 weeks). Radiotherapy was delivered via IMRT with 50 Gy/25 fractions to gross tumor volume and 45 Gy/25 fractions to clinical target volume. Restaging was performed 5 weeks after CRT, with TME planned 6–10 weeks after CRT. Six cycles of adjuvant CAPOX were recommended.
Key endpoints
The primary endpoint was 3-year locoregional recurrence-free survival (LRRFS), defined as time from random assignment to local or regional progression/relapse. Secondary endpoints reported in this final analysis included 3-year disease-free survival (DFS), 3-year overall survival (OS), and adverse events (AEs). AEs were categorized as short-term (during neoadjuvant/adjuvant therapy) and long-term (persisting 1 year after trial start), graded by CTCAE v4.
Read about Rectal Cancer Cure Rate on OncoDaily.
Results
From June 1, 2014, to October 1, 2020, 663 patients were recruited and randomized (331 to nCT and 332 to nCRT). Seventy-four patients did not receive protocol treatment after randomization (31 in nCT; 43 in nCRT). The modified intention-to-treat (mITT) population therefore included 589 treated patients (nCT 300; nCRT 289). Baseline characteristics were well balanced between arms. Median follow-up was 48 months.
Primary endpoint
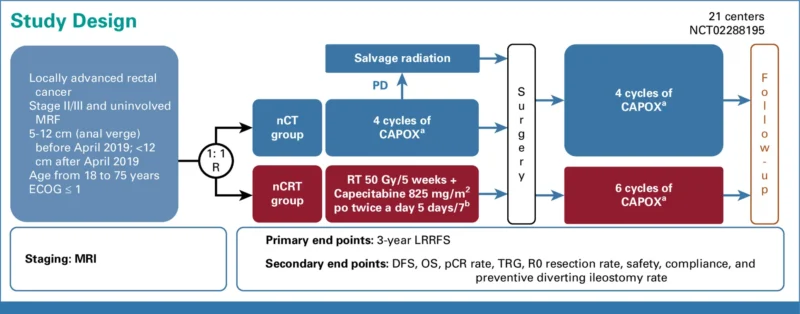
Locoregional progression or relapse within 3 years was rare, occurring in 17 patients total (10 in the nCT arm and 7 in the nCRT arm). Reported sites of local recurrence included the presacral area, mesorectal space, anastomotic site, lateral area, and anterior area.
At 3 years, LRRFS was 97.4% (95% CI, 95.5–99.3) with nCRT and 96.3% (95% CI, 94.0–98.6) with nCT, corresponding to a hazard ratio of 1.40 (95% CI, 0.53–3.68). Because the upper bound of the 95% confidence interval for the hazard ratio exceeded the prespecified noninferiority margin of 1.6, noninferiority of neoadjuvant CAPOX alone was not confirmed, despite very low local recurrence rates in both groups.
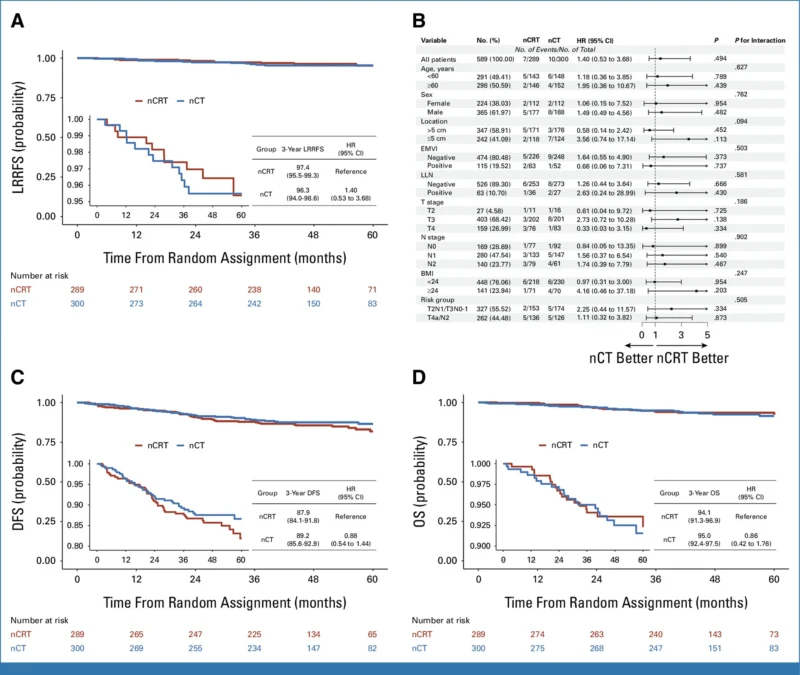
Subgroup analyses showed no significant interaction effects at a two-sided significance level of 0.05. Within the nCT arm, univariate and multivariate analyses did not identify factors clearly associated with worse LRRFS, although an exploratory signal was noted for tumors <5 cm from the anal verge (HR 3.60, P = .063), which did not reach statistical significance.
A small number of patients achieved a complete clinical response and pursued a watch-and-wait strategy: 2 patients in the nCT arm and 5 in the nCRT arm. All seven remained tumor-free during the 3-year postoperative follow-up.
Secondary endpoints
Events contributing to DFS (locoregional recurrence, metastasis, or death) occurred in 30 patients in the nCT arm and 33 in the nCRT arm within 3 years. The 3-year DFS rate was 89.2% (95% CI, 85.6–92.9) with nCT and 87.9% (95% CI, 84.1–91.8) with nCRT (HR 0.88, 95% CI 0.54–1.44).
Overall, 30 deaths were reported within 3 years (14 in the nCT arm and 16 in the nCRT arm). The 3-year OS rate was 95.0% (95% CI, 92.4–97.5) with nCT and 94.1% (95% CI, 91.3–96.9) with nCRT (HR 0.86, 95% CI 0.42–1.76). Subgroup analyses similarly reported no significant interaction effects for DFS or OS.
Safety Profile
During neoadjuvant and adjuvant therapy, the overall burden of toxicity was broadly comparable between strategies. The incidence of any-grade short-term AEs was 78.0% with nCT versus 73.3% with nCRT (P = .189), and grade 3–4 short-term AEs occurred in 15.3% versus 13.1% (P = .449). Selected differences were observed: neutropenia was less frequent with nCT (39.3% vs 48.1%, P = .032), while thrombocytopenia was more frequent with nCT (28.7% vs 20.1%, P = .015). Neurologic AEs were reported in 32.0% of nCT patients versus 24.6% of nCRT patients (P = .045). The incidence of grade ≥3 long-term AEs was low in both groups (4.5% with nCRT vs 3.0% with nCT), without a statistically significant difference.
Long-term toxicity patterns favored omission of radiotherapy. Although any-grade long-term AEs did not differ significantly (40.7% with nCT vs 48.1% with nCRT; P = .070), grade 2–4 long-term AEs were significantly lower with nCT (16.0% vs 26.3%, P = .002). Proctitis was also lower in the nCT arm (33.6% vs 41.7%, P = .049). Among patients receiving radiotherapy, 12.5% had radiodermatitis persisting at 1 year.
Read about Radiotherapy for Rectal Cancer: Types, Success Rate, Side Effects on OncoDaily.
Conclusion
The final CONVERT analysis shows that in MRF-uninvolved locally advanced rectal cancer, both strategies achieved excellent locoregional control at 3 years. Although the trial did not confirm noninferiority of CAPOX-only neoadjuvant therapy for the primary LRRFS endpoint—because the confidence interval crossed the prespecified margin—DFS and OS were similar between arms. Importantly, chemotherapy alone reduced the burden of late toxicity, with significantly fewer grade 2–4 long-term adverse events and less proctitis compared with chemoradiation.
Taken together, these findings suggest that radiation omission may be feasible in carefully selected patients with uninvolved MRF, while highlighting the importance of careful patient selection, particularly for tumors located close to the anal verge.
The full article is available on Journal of Clinical Oncology.