The treatment landscape of EGFR-mutant NSCLC has been transformed by third-generation EGFR tyrosine kinase inhibitors (TKIs), particularly osimertinib. Patients with sensitizing EGFR mutations, such as exon 19 deletions, often experience significant and durable responses to first-line osimertinib-based therapy. However, despite these advances, acquired resistance remains inevitable in most cases, representing a critical barrier to long-term disease control (Kalofonou et al., 2026).
Resistance to osimertinib is biologically complex and heterogeneous. It may arise through “on-target” mechanisms, such as secondary EGFR mutations including C797X alterations that interfere with drug binding, or through “off-target” bypass pathways involving activation of alternative oncogenic drivers such as MET, HER2, or FGFR (Kalofonou et al., 2026). Understanding and identifying these mechanisms is increasingly essential, as it opens the possibility of tailoring subsequent therapies based on tumor evolution rather than applying empiric treatment strategies.
This case report provides a compelling example of how longitudinal tumor sampling and molecular profiling can guide a sequential precision medicine approach, enabling multiple lines of targeted therapy and extending disease control in a patient with metastatic EGFR-mutant NSCLC.
Initial Diagnosis and First-Line Treatment Strategy
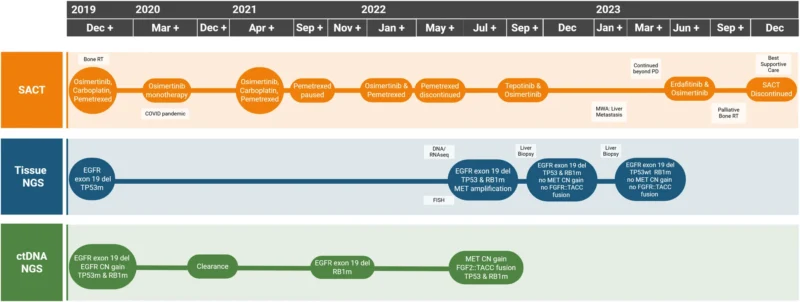
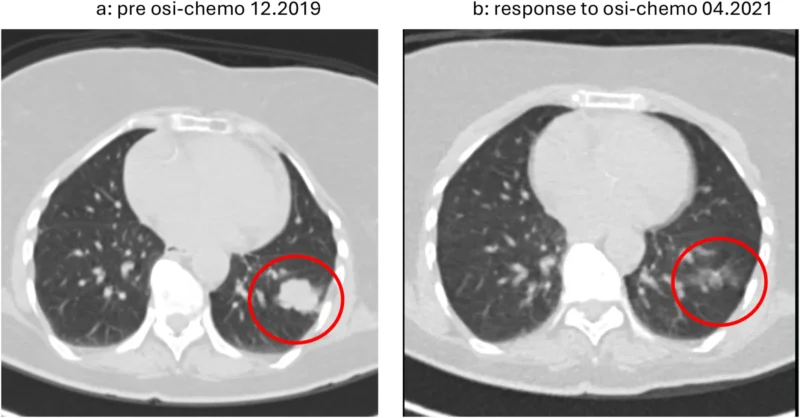
The patient described in this report had metastatic NSCLC harboring an EGFR exon 19 deletion, one of the most common and well-characterized sensitizing mutations. In line with emerging evidence supporting combination strategies, the patient received first-line treatment with osimertinib combined with carboplatin and pemetrexed, reflecting the regimen evaluated in the FLAURA2 trial (Kalofonou et al., 2026).
This approach aims to enhance initial disease control by combining targeted therapy with chemotherapy, potentially delaying the onset of resistance. As expected, the patient initially demonstrated a favorable response, highlighting the continued effectiveness of EGFR-targeted therapy in the frontline setting.
However, as is typical in EGFR-mutant NSCLC, disease progression eventually occurred, prompting further investigation into the underlying resistance mechanisms.
First Resistance Event: MET Amplification as a Targetable Driver
Upon disease progression, the patient underwent repeat tumor biopsy alongside circulating tumor DNA (ctDNA) analysis. This dual approach allowed for a comprehensive assessment of tumor biology across both tissue and plasma compartments.
Molecular profiling revealed MET amplification, which is recognized as the most common acquired resistance mechanism to osimertinib (Kalofonou et al., 2026). MET amplification activates bypass signaling pathways that sustain tumor growth independently of EGFR inhibition, thereby rendering osimertinib less effective.
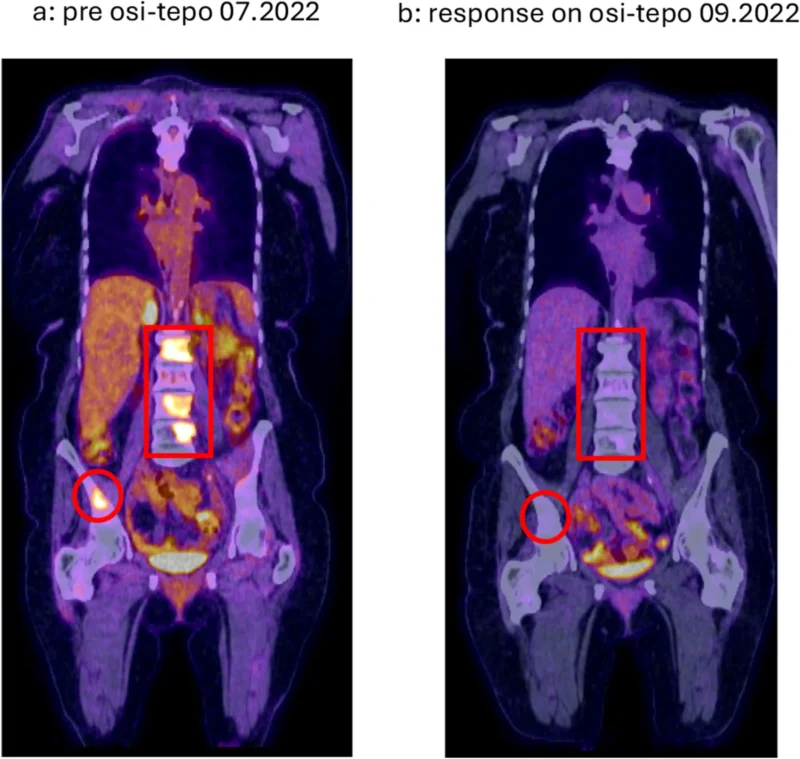
Importantly, MET amplification represents a therapeutically actionable alteration. Based on this finding, the patient was treated with a combination of osimertinib and the selective MET inhibitor tepotinib. This strategy reflects an increasingly adopted approach in clinical practice, where the original EGFR inhibition is maintained while targeting the newly emerged resistance pathway.
The patient achieved a durable partial response with this combination, demonstrating the clinical benefit of mechanism-driven treatment selection.
Tumor Evolution Continues: Emergence of FGFR Fusion
Despite the success of MET-targeted therapy, further disease progression occurred, underscoring the dynamic and adaptive nature of tumor evolution. Once again, repeat molecular profiling was performed to identify new resistance mechanisms.
This time, analysis revealed the emergence of an FGFR fusion, representing a distinct bypass pathway driving tumor growth. FGFR alterations are less common compared to MET amplification but are increasingly recognized as contributors to resistance in EGFR-mutant NSCLC (Kalofonou et al., 2026).
The identification of this new molecular driver prompted a change in therapeutic strategy. The patient was switched to a combination of osimertinib and erdafitinib, an FGFR inhibitor. This sequential targeting of evolving resistance mechanisms led to another favorable clinical response.
This phase of treatment highlights a critical principle in modern oncology: resistance is not a single event but a continuous process of clonal selection and evolution. Each new resistance mechanism may represent a new therapeutic opportunity if appropriately identified.
The Role of Longitudinal Tumor Sampling
A central theme of this case is the importance of repeated and longitudinal tumor sampling. At each point of disease progression, both tissue biopsy and ctDNA analysis were used to reassess tumor biology.
This approach offers several advantages. Tissue biopsy provides detailed histologic and genomic information from specific disease sites, while ctDNA allows for a broader, systemic view of tumor heterogeneity. Together, they enable a more complete understanding of evolving resistance mechanisms.
In this case, the integration of these diagnostic modalities was essential in identifying sequential resistance drivers, first MET amplification and later FGFR fusion, each of which directly informed treatment decisions.
Without such repeated molecular evaluation, these actionable alterations could have been missed, and the patient may have been transitioned prematurely to non-targeted therapies such as chemotherapy.
Sequential Precision Medicine: Concept and Clinical Implications
The concept of sequential precision medicine is illustrated clearly in this case. Rather than viewing resistance as the endpoint of targeted therapy, it is approached as an opportunity to re-characterize the tumor and identify new vulnerabilities.
This strategy involves several key steps. First, recognition of disease progression prompts re-biopsy and molecular profiling. Second, identified resistance mechanisms are matched with targeted therapies when available. Third, treatment is adapted dynamically as new alterations emerge.
In this patient, this approach allowed for multiple lines of targeted therapy, each tailored to the evolving genomic landscape of the tumor. The result was prolonged disease control and an extended chemotherapy-free interval, which is particularly meaningful for patient quality of life.
This paradigm represents a shift from static to dynamic cancer treatment, where therapy evolves in parallel with tumor biology.
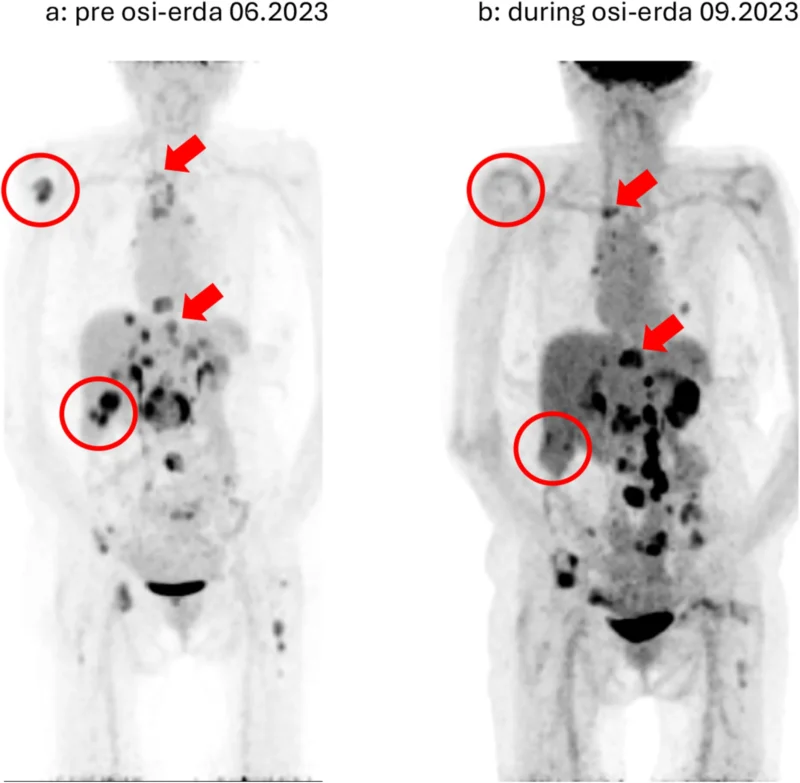
Limitations and Challenges
While this case highlights the promise of sequential precision medicine, it also underscores its limitations.
Not all resistance mechanisms are targetable, and access to appropriate targeted therapies may be limited by regulatory, financial, or logistical constraints. Additionally, tumor heterogeneity can complicate interpretation of molecular findings, as different metastatic sites may harbor distinct resistance mechanisms.
There are also practical challenges associated with repeated biopsies, including procedural risks and patient burden. Although ctDNA offers a less invasive alternative, it may not always capture all relevant alterations.
Furthermore, evidence supporting specific combination strategies, such as osimertinib with MET or FGFR inhibitors, is still evolving, and optimal sequencing remains an area of active research.
A Glimpse Into the Future of EGFR-Mutant NSCLC Management
This case exemplifies the direction in which lung cancer treatment is heading. The integration of molecular diagnostics, targeted therapies, and longitudinal monitoring is enabling a more personalized and adaptive approach to care.
As our understanding of resistance mechanisms deepens, and as new targeted agents become available, the ability to extend disease control through sequential therapy is likely to improve further.
Future strategies may include earlier detection of resistance through ctDNA monitoring, combination therapies designed to prevent resistance from emerging, and more comprehensive profiling techniques to capture tumor heterogeneity.
Ultimately, the goal is to transform metastatic EGFR-mutant NSCLC into a chronic, manageable disease, where treatment is continuously adapted based on tumor evolution.

Read About The TOP Study on OncoDaily lung
Conclusion
This case demonstrates that acquired resistance to osimertinib in EGFR-mutant NSCLC is not a therapeutic dead end but a biologically informative process that can guide further treatment.
Through longitudinal tumor sampling and molecular profiling, sequential resistance mechanisms including MET amplification and FGFR fusion were identified and successfully targeted. This enabled multiple lines of effective therapy and prolonged disease control.
The findings highlight the importance of integrating precision diagnostics into routine clinical practice and adopting a dynamic, evolution-based approach to cancer treatment.
As precision oncology continues to advance, such strategies are likely to play an increasingly central role in improving outcomes for patients with EGFR-mutant NSCLC.
You Can Read Full Article Here