The treatment landscape of EGFR-mutant NSCLC has evolved significantly with the introduction of third-generation EGFR tyrosine kinase inhibitors such as osimertinib. However, not all patients derive the same benefit. A growing body of evidence suggests that EGFR/TP53 mutant NSCLC represents a biologically aggressive subgroup, associated with shorter progression-free survival and reduced responsiveness to EGFR-TKI monotherapy.
This clinical challenge has raised an important question: can intensifying first-line therapy improve outcomes in this high-risk population?
At the European Lung Cancer Congress 2026, results from the phase III TOP study provide compelling data addressing this question, evaluating osimertinib with or without chemotherapy as first-line treatment in patients with EGFR/TP53 mutant NSCLC.

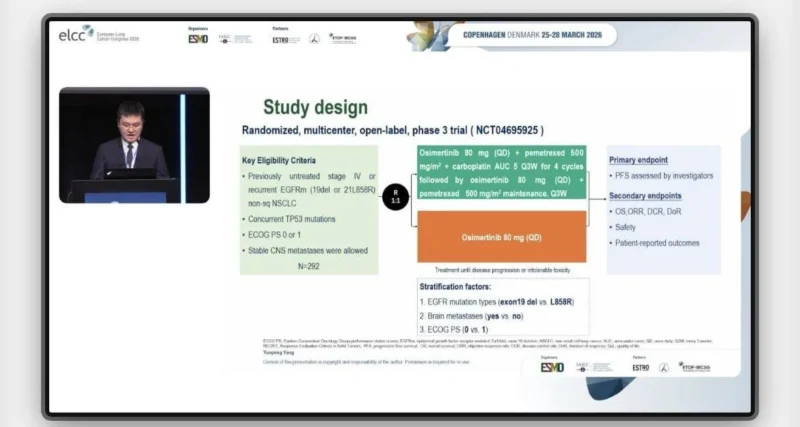
Study Design and Patient Population
The TOP study is a randomized, multicenter phase III trial specifically designed to address the unmet need in patients with EGFR-mutant NSCLC and concurrent TP53 mutations. Eligible patients had previously untreated advanced non-squamous NSCLC with sensitizing EGFR mutations, including exon 19 deletions or L858R substitutions, along with confirmed TP53 alterations.
Participants were randomized in a 1:1 ratio to receive either standard osimertinib monotherapy at 80 mg once daily or a combination regimen consisting of osimertinib plus platinum-based chemotherapy with pemetrexed for four cycles, followed by maintenance osimertinib and pemetrexed.
A total of 294 patients were enrolled, with 146 assigned to the combination arm and 148 to osimertinib alone. Baseline characteristics were well balanced between groups. The median age was 57 years, and approximately half of the patients were female. Notably, nearly 50% of patients in both arms had central nervous system metastases at baseline, underscoring the aggressive nature of this disease subset.
Key Efficacy Results
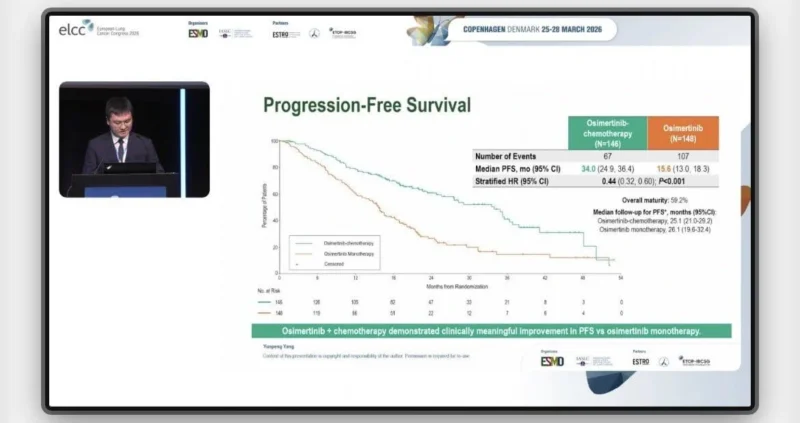
The addition of chemotherapy to osimertinib resulted in a marked and statistically significant improvement in progression-free survival, the primary endpoint of the study.
Median progression-free survival was 34.0 months in the combination arm compared to 15.6 months with osimertinib alone, corresponding to a hazard ratio of 0.44 (95% CI 0.32–0.61; p<0.001). This translates into a 56% reduction in the risk of disease progression or death, representing a clinically meaningful benefit in this high-risk population.
Importantly, the benefit of combination therapy was consistent across all prespecified subgroups. Patients with central nervous system metastases derived similar benefit, highlighting the ability of this strategy to address both systemic and intracranial disease. Likewise, efficacy was maintained across different EGFR mutation subtypes, including both exon 19 deletions and L858R mutations.
The objective response rate was also improved with combination therapy, reaching 82.9% compared to 72.0% with osimertinib monotherapy. In addition to higher response rates, responses were more durable, with a median duration of response of 32.7 months in the combination arm versus 15.3 months in the monotherapy arm.
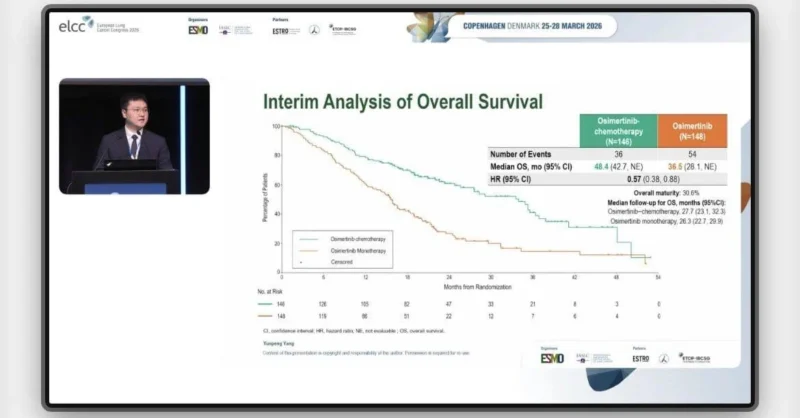
Although overall survival data remain immature, early signals suggest a potential survival advantage. At the time of analysis, the hazard ratio for overall survival was 0.57 (95% CI 0.37–0.87), indicating a favorable trend that will require longer follow-up for confirmation.
Safety and Tolerability
As expected, the addition of chemotherapy resulted in increased toxicity. Grade 3 or higher treatment-related adverse events were reported in 62.4% of patients receiving osimertinib plus chemotherapy, compared with 14.9% in the osimertinib monotherapy arm.
Despite this higher incidence of adverse events, no new safety signals were identified, and the toxicity profile was consistent with known effects of platinum-based chemotherapy combined with EGFR-targeted therapy. The safety findings suggest that, while more intensive, the combination regimen remains clinically manageable in appropriately selected patients.
Clinical Implications
The findings of the TOP study have important implications for clinical practice. Patients with EGFR-mutant NSCLC and concurrent TP53 mutations represent a subgroup with historically poor outcomes, often experiencing early progression on EGFR-TKI monotherapy.
By demonstrating a significant and durable improvement in progression-free survival, this study supports the use of upfront combination therapy as a strategy to overcome intrinsic resistance mechanisms associated with TP53 alterations.
These results are consistent with and extend findings from previous studies such as FLAURA2, reinforcing the concept that combining targeted therapy with chemotherapy may enhance treatment efficacy in selected populations. Importantly, the magnitude of benefit observed in the TOP study suggests that TP53 co-mutation status may serve as a key biomarker for treatment intensification.
What Are Experts Saying About Trial?
Roberto Ferrara- Medical Oncologist, Researcher in Thoracic Oncology and Molecular Immunology writes in X
TOP trial shows benefit of osi+chemio in EGFR mut/P53 mut NSCLC with early separation of OS curve (not occurring in Flaura2).
P53 mut could be DISRUPTIVE and non-DISRUPTIVE which are functionally different categories. Trials should catch the functional P53 heterogeneity #ELCC2026
Key Takeaway
The phase III TOP study demonstrates that osimertinib combined with chemotherapy significantly improves progression-free survival, response rate, and durability of response compared with osimertinib alone in patients with EGFR/TP53 mutant NSCLC.
These findings suggest that combination therapy may represent a more effective first-line strategy for this high-risk subgroup, pending further maturation of overall survival data.
You can read full abstract here


