Tumor lysis syndrome (TLS) is one of oncology’s most urgent complications, arising when large numbers of cancer cells undergo rapid lysis and release their intracellular contents into the systemic circulation. Although effective cancer therapy depends on tumor cell destruction, abrupt large-scale cell breakdown may overwhelm the body’s homeostatic capacity.
Still considered a predictable and potentially preventable, TLS has become more variable in modern practice with the use of newer, highly effective therapies. In this context, the review highlights key aspects of TLS, from underlying mechanisms to present-day prevention and management.
What Increases the Risk of TLS ?
The risk is determined by a combination of tumor-related and patient-related factors. Malignancies characterized by high proliferation rates, substantial tumor burden and marked sensitivity to therapy carry the greatest risk. TLS most commonly occurs in patients with bulky, chemosensitive hematologic malignancies. Importantly, tumor-related risk does not necessarily reflect prognosis. While solid tumors are generally categorized as low risk, the occurrence of TLS in this setting is associated with poor prognosis.
Patient-specific factors such as pre-existing renal dysfunction, dehydration and elevated baseline uric acid or LDH levels raise the risk of clinically significant TLS.
The predisposition of hematologic malignancies to TLS is also explained by their unique biological setting. In leukemias and aggressive lymphomas, malignant cells circulate within the bloodstream and bone marrow, placing them in immediate and continuous contact with systemic therapy. Unlike solid tumors, where drug penetration may be heterogeneous, hematologic cancers are exposed rapidly and uniformly, allowing treatment to trigger a near-synchronous wave of cell death.
Of Note
TLS has also been reported in less predictable clinical settings, including spontaneous occurrence, as a presentation of occult malignancy, in association with glucocorticoids, in traditionally nonchemosensitive cancers, and following radiation therapy. Risk is not static, it evolves with treatment decisions.
The Chain Reaction of Systemic Failure
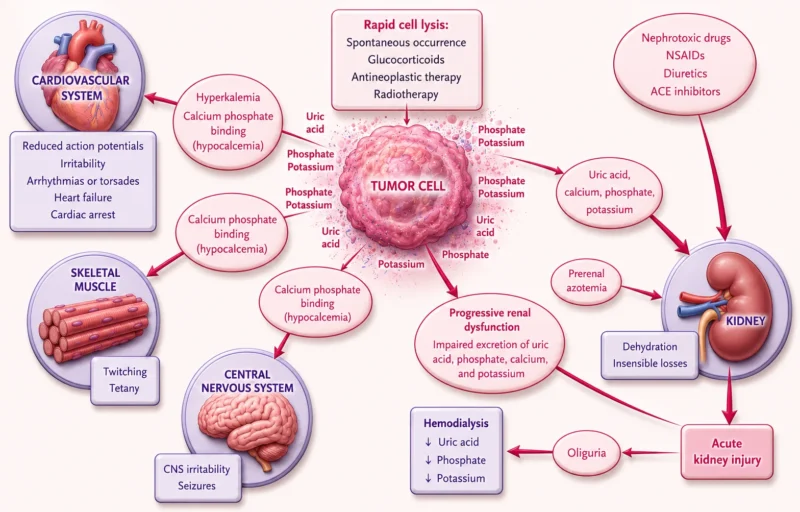
At its core, TLS unfolds as a rapid biological chain reaction. Following cell lysis, potassium surges first, increasing the risk of cardiac arrhythmias. At the same time, phosphate levels rise, binding circulating calcium and leading to functional hypocalcemia. Meanwhile, nucleic acids are metabolized into uric acid, which, at concentrations exceeding its solubility threshold, can crystallize within the renal tubules and disrupt kidney function.
Experimental data suggest that crystal-dependent mechanisms alone are insufficient to explain renal injury. Increased extracellular histones and proinflammatory signaling, including uric acid-mediated activation of cytokines and innate immunity, might be additional pathways. Xanthine, which is less soluble than uric acid, may also contribute to nephrotoxicity.
Importantly, hyperphosphatemia plays a central role, both directly and indirectly, by calcium-phosphate deposition in renal tubules. As renal clearance declines, these metabolic abnormalities intensify, amplifying one another in a self-perpetuating cycle.
Viewed as a whole TLS is the mismatch between release and clearance. The kidneys become both targets and amplifiers of injury – even modest ongoing cellular breakdown can produce disproportionate systemic effects. What begins as effective tumor destruction can therefore escalate into a systemic emergency – illustrating how, in TLS, each step fuels the next stage of clinical decompensation.
nejm.org
TLS Across the Spectrum
TLS, according to the Cairo-Bishop criteria, is categorized into two main groups: Laboratory TLS (LTLS) and Clinical TLS (CTLS).
LTLS requires at least two metabolic abnormalities within the same 24-hour period: hyperuricemia, hyperkalemia, hyperphosphatemia (≥25% increase from baseline), and hypocalcemia ≥25% decrease. CTLS is diagnosed when LTLS is accompanied by end-organ complications.
The most significant drawback is that the definition requires the initiation of chemotherapy, whereas, spontaneous TLS is also possible. Another one is the use of a creatinine threshold >1.5 times the upper limit for age and sex, as patients with chronic kidney disease may have elevated baseline levels independent of acute kidney injury.
The exact incidence of TLS remains undetermined. A study of 788 adult and pediatric patients with acute leukemia or non-Hodgkin lymphoma across 4 European countries showed that LTLS was more common than CTLS, occurring in 18.9% and 5% of patients, respectively. By disease, rates were highest in ALL (21.4% laboratory, 5.2% clinical), followed by non-Hodgkin lymphoma (19.6% and 6.1%), and AML (14.7% and 3.4%).
Clinical Profile and Mimics
Early symptoms may include abdominal pain, distension and dysuria. As it progresses, renal involvement may appear as acute kidney injury and uremia, with nausea, vomiting, pruritus and volume overload (rales, rhonchi, peripheral edema). Hypocalcemia may present with neuromuscular irritability and altered mental status, with bronchospasm or seizures in more severe cases.
Hyperkalemia may lead to weakness or paralysis. As the syndrome progresses, patients may develop lethargy with potential progression to arrhythmias, syncope or sudden cardiac death.
Importantly, TLS must be distinguished from other conditions with similar electrolyte disturbances.
- Hyperkalemia may arise from rhabdomyolysis, acute tubular necrosis, metabolic acidosis or endocrine disorders.
- Hyperphosphatemia can be seen in plasma cell dyscrasias, vitamin D intoxication or pseudohyperphosphatemia.
- Hyperuricemia may be associated with gout, hemolytic anemia, diabetic ketoacidosis or lymphoproliferative disorders themselves.
Important Note: Diagnostic Confounders
In patients with lymphoid malignancies and marked leukocytosis (>100,000/µL), pseudohyperkalemia may occur, most commonly in CLL. It results from ex vivo lysis of fragile tumor cells during blood collection, transport or processing. Isolated hyperkalemia without hyperphosphatemia or hyperuricemia after therapy suggests pseudohyperkalemia, an ECG may help clarify the diagnosis.
Spontaneous TLS is not associated with hyperphosphatemia. This distinction may be attributed to the high cell turnover rate in tumors, which increases uric acid levels while the tumor reutilizes the released phosphate to grow new cells. In contrast, postchemotherapy TLS involves the destruction of tumor cells without the synthesis of new cells.
Prevention Is Part of Treatment
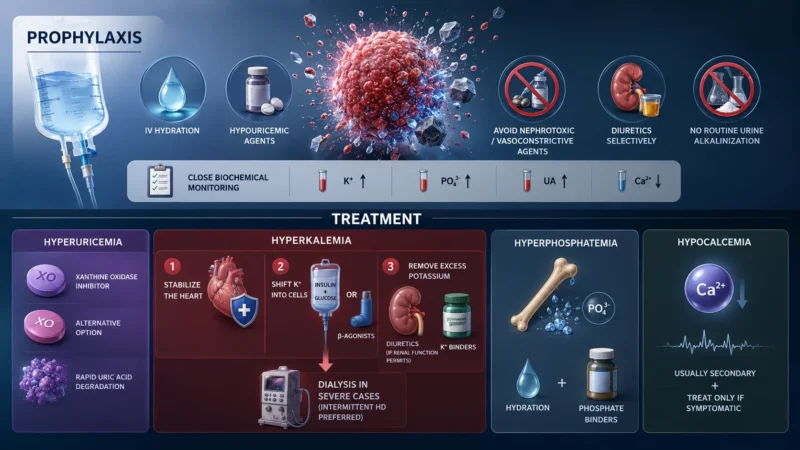
Prophylaxis is primarily achieved through aggressive intravenous hydration and the use of hypouricemic agents. Nephrotoxic or vasoconstrictive agents (NSAIDs, iodinated contrast) should be avoided.
Diuretics may be used selectively, provided there is no hypovolemia or obstruction. Routine urine alkalinization with sodium bicarbonate is no longer recommended due to concerns about calcium-phosphate deposition. Its use is limited to selected cases with metabolic acidosis when other options are unavailable. Close biochemical monitoring is essential, with frequency guided by risk stratification.
Among pharmacologic strategies, allopurinol inhibits xanthine oxidase and prevents new uric acid formation. Yet, it does not reduce existing hyperuricemia and may lead to xanthine accumulation. Febuxostat offers an alternative with fewer hypersensitivity reactions and no need for renal dose adjustment, though it should be avoided with certain drugs such as azathioprine.
Rasburicase, a recombinant urate oxidase, is preferred in high-risk patients because it rapidly degrades uric acid into soluble allantoin. This reaction generates hydrogen peroxide, so caution is required in patients with impaired antioxidant capacity.
Preemptive Approaches: Pros and Cons
The American Society for Apheresis supports leukapheresis in patients with leukostasis and recommends prophylactic cytoreduction in acute leukemias. However, the extent to which these measures reduce tumor lysis risk or improve outcomes remains uncertain. In addition, procedures such as apheresis catheter placement carry bleeding risks in patients with thrombocytopenia or coagulopathy. Thus, the central principle remains rapid but safe reduction.
Although direct evidence for reducing TLS risk is limited, prephase steroids are often used in patients with high tumor volume or poor performance status, with additional benefits including improved functional status, reduced early toxicity and better tolerance of full-dose therapy. Similarly, short delays in administering agents such as Rituximab may reduce TLS and infusion-related reactions without compromising outcomes.
In very high-risk patients, early nephrology involvement is often warranted, and in select cases preemptive dialysis access may be considered.
Approaches to Established TLS
Acute hyperkalemia management centers on stabilizing the heart, shifting potassium into cells, and removing excess potassium. Calcium is given to stabilize myocardial cell membranes, while insulin with glucose or β-agonists temporarily shift potassium intracellularly.
Elimination can be supported with diuretics (if renal function permits) or potassium binders such as Sodium Zirconium Cyclosilicate. In severe cases, dialysis is required, with intermittent hemodialysis providing more rapid correction. Hyperphosphatemia is treated with hydration and phosphate binders. Hypocalcemia is usually secondary and should be treated only if symptomatic.
Special Management Considerations
Management of TLS can be challenging in Multiple Myeloma due to baseline renal dysfunction. Also, Bortezomib and Daratumumab have been associated with TLS in retrospective and pharmacovigilance studies, especially in combination regimens. Renal dysfunction lowers the threshold for more aggressive prophylaxis (often rasburicase over allopurinol).
Venetoclax, a selective BCL-2 inhibitor, carries a more substantial TLS risk due to its rapid debulking effect. Early clinical studies reported TLS fatalities, prompting the adoption of a stepwise dose ramp-up alongside intensive prophylaxis. With these measures, TLS incidence has markedly decreased, with only rare laboratory cases and no clinical events in subsequent cohorts. Risk stratification is primarily based on tumor burden and renal function.
Shifting Paradigms in TLS
The contemporary understanding of TLS extends beyond traditional chemotherapy. With the advent of highly effective targeted agents and immunotherapies, rapid tumor cell death can now occur across a broader spectrum of malignancies and clinical settings, including outpatient care.
CDK inhibitor Alvocidib has been associated with a high rate of TLS (≈46%). Identified risk factors include advanced disease stage, elevated β2-microglobulin and hypoalbuminemia. By comparison, Ibrutinib and Idelalisib demonstrate a very low incidence of TLS despite rapid nodal responses.
Over 150 cases have been reported to the FDA Adverse Event Reporting System, involving PD-1, PD-L1, and CTLA-4 inhibitors, with significant outcomes including hospitalization (29%), life-threatening events (5%), and death (44%), particularly when used in combination.
With BsAbs, continuous T-cell engagement may produce sustained tumor killing and evolving metabolic abnormalities during early treatment cycles. CAR T-cell therapies can trigger abrupt cellular breakdown following robust immune activation, and TLS may coexist with cytokine release syndrome, complicating the clinical picture.
Current risk stratification still relies largely on static biochemical thresholds, which may not fully capture the early dynamic changes preceding overt kidney injury. Emerging evidence suggests that rapidly rising serum phosphate levels may represent an earlier and more sensitive indicator of TLS-associated acute kidney injury.
TLS surveillance is evolving toward outpatient and longitudinal monitoring, particularly during early treatment initiation and dose-escalation phases. This shift underscores a broader principle: as therapies become more effective, the management of treatment-related complications must evolve in parallel.

You can also read: Acute Myeloid Leukemia (AML) in 2026: The Current Landscape and Future Perspectives
Written by Susanna Mikayelyan, MD
FAQ
Can Tumor Lysis Syndrome Happen Before Cancer Treatment Starts?
Yes. Although TLS is classically associated with chemotherapy, spontaneous TLS can occur before any treatment is initiated. This is most often seen in rapidly proliferating hematologic malignancies with high tumor turnover. In some cases, TLS may even become the first clue leading to a cancer diagnosis.
Why Are the Kidneys So Vulnerable in Tumor Lysis Syndrome?
The kidneys act as the body’s filtration system, suddenly forced to clear massive amounts of potassium, phosphate, and uric acid released from dying tumor cells. Crystal formation, inflammatory signaling, and reduced renal perfusion can together trigger acute kidney injury, turning the kidneys into both victims and amplifiers of TLS.
Could Immunotherapy Increase the Risk of Tumor Lysis Syndrome?
Potentially, yes. Modern immunotherapies such as CAR T-cell therapy and bispecific antibodies can induce extremely rapid tumor destruction. As cancer treatments become more effective, clinicians are increasingly encountering TLS in settings previously considered low risk.
Why Is Hyperkalemia Considered the Most Urgent Electrolyte Abnormality in TLS?
Because elevated potassium can rapidly destabilize cardiac electrical activity. Severe hyperkalemia may trigger arrhythmias or sudden cardiac death even before other metabolic complications fully evolve, making immediate recognition and treatment critical.
Can Low-Risk Tumors Still Cause Tumor Lysis Syndrome?
Surprisingly, yes. Although TLS is far more common in aggressive hematologic cancers, rare cases have been reported in solid tumors, especially after highly effective targeted therapies. When TLS develops in traditionally low-risk cancers, it is often associated with poor prognosis.
Could Future Biomarkers Detect TLS Earlier Than Current Criteria?
Possibly. Emerging evidence suggests that rapidly rising phosphate levels and dynamic metabolic trends may predict kidney injury earlier than traditional static laboratory thresholds. Researchers are now exploring more sensitive and real-time approaches to TLS surveillance.
Why Is Tumor Lysis Syndrome Becoming More Relevant Outside the Hospital?
Many modern cancer therapies are now initiated in outpatient settings, including targeted agents and immunotherapies. This shift means TLS monitoring increasingly extends beyond inpatient oncology wards, requiring patients and clinicians to recognize warning signs earlier and more proactively.