Acute Myeloid Leukemia (AML) is an aggressive hematologic malignancy characterized by clonal proliferation and maturation blockade of myeloid progenitor cells, resulting in the accumulation of leukemic blasts in the bone marrow, peripheral blood and occasionally extramedullary tissues, with consequent suppression of normal hematopoiesis.
Today, on AML World Awareness Day, it is an important opportunity to reflect on the progress achieved in AML management and how future care is taking shape.
AML Epidemiology and Risk Factors
According to the American Cancer Society, the incidence of AML is 6.6 cases per 100,000 people per year, with rates slightly higher in men. AML remains primarily a disease of older adults, with most cases occurring after age 60. Although AML accounts for only about 1% of all cancers, it represents nearly one-third of adult leukemias, making it the most common acute leukemia in adults, with an estimated 5-year overall survival of 32%.
Most cases of AML arise sporadically without a single identifiable cause, yet several factors are associated with increased risk. Advancing age is the most important risk factor, likely reflecting the cumulative acquisition of genetic alterations and age-related clonal hematopoiesis. Other recognized risk factors include smoking, occupational or environmental benzene, dioxin exposure and prior ionizing radiation.
The relationship between primary immunodeficiency, bone marrow failure syndromes and AML is increasingly recognized. Certain germline disorders (such as Shwachman-Diamond syndrome, Fanconi anemia) impair both hematopoietic precursor growth and immune cell development, creating a combined predisposition to cytopenias, immune dysfunction and leukemic transformation.
One major conceptual change has been the introduction of the category AML myelodysplasia-related (AML-MR), which includes cases with prior MDS or MDS/MPN history. Therapy-related AML is another important subtype that develops after prior exposure to cytotoxic chemotherapy and/or radiation. This form of AML is biologically distinct and is more frequently associated with adverse-risk features and TP53 mutations (Nelson A, et al. 2024. Flores Y, et al. 2025).
Clinical Features and Early Warning Signs
As blast burden rises, normal blood cell production declines. This results in the classic consequences of bone marrow failure: fatigue, weakness, pallor, and shortness of breath from anemia, recurrent infections and fever from neutropenia, and easy bruising, petechiae, epistaxis, or gingival bleeding from thrombocytopenia. Symptoms of AML often evolve rapidly over days to weeks rather than months.
Many patients also develop systemic symptoms such as weight loss, night sweats, malaise, and bone pain related to marrow expansion. In some cases, leukemic cells infiltrate tissues outside the marrow, producing gum hypertrophy (particularly in monocytic AML), skin lesions, lymphadenopathy and hepatosplenomegaly (much rarer compared with ALL). Less common but serious presentations include central nervous system involvement, leukostasis and disseminated intravascular coagulation, especially in acute promyelocytic leukemia (APL).
Because AML symptoms frequently overlap with common benign conditions such as viral illness, iron deficiency, or transient fatigue states, persistent or unexplained cytopenias, recurrent infections, abnormal bleeding, or rapidly worsening constitutional symptoms warrant urgent hematologic evaluation (Fareed S, et al. 2025).
How Is AML Diagnosed?
Initial evaluation typically includes a CBC, peripheral blood smear, coagulation profile, comprehensive chemistry panel, and markers of tumor turnover such as lactate LDH and uric acid. These studies help identify cytopenias, circulating blasts, coagulopathy, and metabolic complications that may require urgent management.
Definitive diagnosis usually requires bone marrow aspiration and biopsy, together with multiparameter flow cytometry, conventional karyotyping, and targeted FISH in selected cases. Molecular analysis by PCR and/or NGS is now central to modern workup. Traditionally, the diagnosis of AML requires ≥20% myeloid blasts in either the bone marrow or peripheral blood. However, specific AML-defining genetic alterations may establish the diagnosis even below this threshold.
The 2022 WHO and ICC classifications have shifted the diagnostic focus toward genetic alterations, allowing for more precise subtyping and personalized treatment decisions.
AML Genetics and Their Clinical Significance
AML is a clonal process that develops from a transformed HSC. This evolution may arise in otherwise healthy individuals and can precede the overt development of leukemia. With advancing age, recurrent AML-related mutations may accumulate in hematopoietic stem and progenitor cells, leading to clonal expansion without development of leukemia, a phenomenon known as clonal hematopoiesis of indeterminate potential (CHIP). These cells have a proliferative advantage, allowing them to expand while maintaining normal hematopoiesis. Progression from clonal hematopoiesis to AML is estimated at 0.5-1% per year.
Epigenetic Regulator Mutations
These are early events. DNMT3A mutation occurs in ~20% of adult AML, leading to reduced methyltransferase activity, dominant-negative effects, and genome-wide hypomethylation. These mutations frequently co-occur with other lesions and often persist after treatment as preleukemic clones, limiting their utility for MRD monitoring.
TET2 mutation is present in ~15% of cases and leads to reduced DNA hydroxymethylation, with generally adverse prognosis. IDH1/2 mutations generate the oncometabolite 2-hydroxyglutarate, disrupting α-ketoglutarate – dependent pathways and cellular metabolism. ASXL1 mutation causes loss of normal chromatin repression, and are generally associated with unfavorable prognosis. KMT2A rearrangements occur in pediatric and adult therapy-related AML.
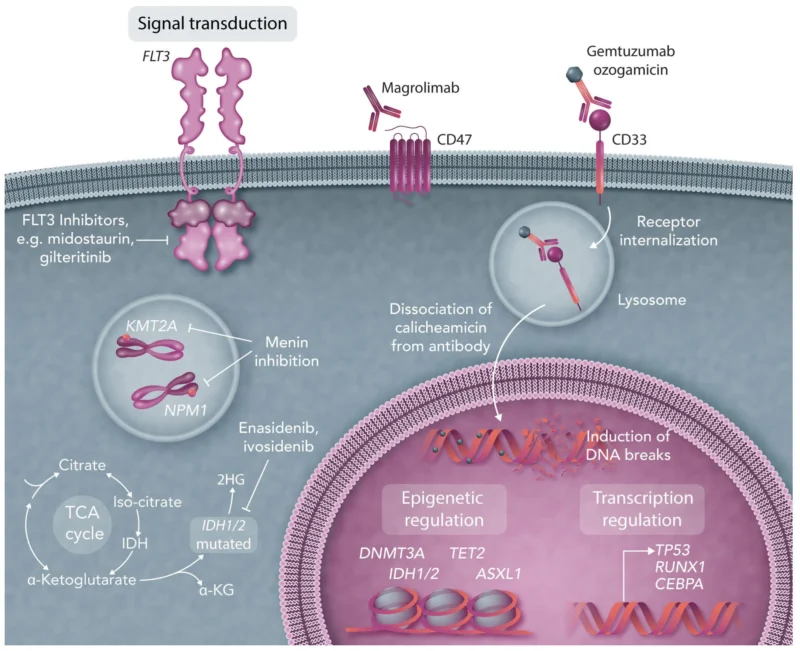
Signaling Pathway Mutations
Among the most clinically relevant are FLT3 mutations, particularly internal tandem duplications (FLT3-ITD), as well as tyrosine kinase domain point mutations (FLT3-TKD). These lesions activate growth and survival pathways such as RAS/MAPK, PI3K/AKT, and JAK/STAT5. Notably, KIT/RAS pathway mutations may also emerge as a mechanism of resistance to FLT3 inhibitors.
Transcription Factor and Differentiation Gene Mutations
Mutations affecting RUNX1 and CEBPA are central. In addition, the fusion rearrangements CBFB::MYH11 and RUNX1::RUNX1T1 define core-binding factor AML, a biologic subtype generally associated with more favorable prognosis. APL is associated with t(15;17) and is highly responsive to differentiation therapy.
NPM1 encodes a nucleophosmin (chaperone protein predominantly located in the nucleus). Mutation causes abnormal cytoplasmic relocalization (NPM1c), while a small residual nuclear fraction alters chromatin interactions and promotes leukemogenesis. NPM1c drives a characteristic transcriptional program and promotes self-renewal, partly through interactions with KMT2A-associated chromatin, which may explain its sensitivity to menin inhibitors. Because NPM1 mutations are highly specific to AML, common at diagnosis, and usually stable at relapse, they are excellent markers for MRD monitoring.
Genomic Stability Alterations
TP53 is a key tumor suppressor that preserves genomic integrity. Its mutations are implicated in MDS, de novo and therapy-related AML. The prevalence rises to 20-40% in older patients and therapy-related myeloid neoplasms. Clinically, TP53-mutated AML is strongly associated with complex karyotype, genomic instability, chemoresistance, rapid progression, and poor OS (Padmakumar D. et al., 2021, Kayser S. et al., 2023, Wachter F. et al., 2024).
Risk Stratification in AML
By NCCN and ELN criteria, patients are broadly categorized into favorable, intermediate, and adverse-risk groups based on cytogenetic abnormalities and recurrent molecular mutations. Prognostic guidelines for AML have largely been derived from cohorts of intensively treated patients younger than 60 years, which may limit their applicability in older adults.
To address this gap, a recent study developed a prognostic model for patients aged 60 years and older using the NCRI-AML18 and HOVON-SAKK cohorts. The model identified four risk categories – favorable, intermediate, poor, and very poor – based on clinical variables (male sex, white blood cell count ≥20 × 10⁹/L, age >65 years) and genetic features, including monosomal karyotype and mutations in TP53, RUNX1, FLT3-ITD, ASXL1, and DNMT3A.
Compared with ELN 2022, this classification showed improved prognostic calibration in derivation and internal validation cohorts. In lower-risk groups, reductions in relapse risk appeared to be offset by higher non-relapse mortality related to transplantation. Although promising, these findings require external validation before broader clinical adoption (Shimony S, et al. 2025).
Multi-omics risk models combine genomics, transcriptomics, epigenomics, proteomics, metabolomics, and MRD dynamics to improve prognostic accuracy beyond conventional cytogenetics or mutation panels. AI and machine-learning platforms are expected to accelerate this field by identifying complex interactions across large datasets that are not apparent through standard analysis, and better predict relapse risk, treatment resistance, and survival (Dasdemir E et al., 2026).
AML Treatment in 2026
The past 5 decades in AML therapy were dominated by one modestly effective and poorly tolerated standard of care: the combination of cytarabine and anthracyclines, often referred to as the 7 + 3 regimen. However, outcomes were suboptimal in many subgroups, particularly older or medically unfit patients. Over the last decade, treatment paradigms have evolved rapidly.
To date, 11 novel agents have been approved for different AML indications by the FDA: Venetoclax (BCL2 inhibitor), three FLT3 inhibitors – Gilteritinib, Quizartinib, and Midostaurin, two IDH1 inhibitors – Ivosidenib and Olutasidenib, one IDH2 inhibitor – Enasidenib, Gemtuzumab ozogamicin, a CD33 antibody – drug conjugate, Oral azacitidine, CPX-351 (a liposomal formulation with a 5:1 cytarabine:daunorubicin), and Glasdegib (Hedgehog pathway inhibitor)
Oral Decitabine-cedazuridine, a highly absorbable bioequivalent hypomethylating agent, was approved as an alternative to parenteral HMAs in MDS and CMML, and is also used in AML, with approval from the EMA but not yet the FDA. Revumenib was approved by the FDA in 2024 for refractory or relapsed acute leukemia with KMT2A translocation (Kantarjian HM et al., 2025).
haematologica.org
Personalizing the Treatment Path
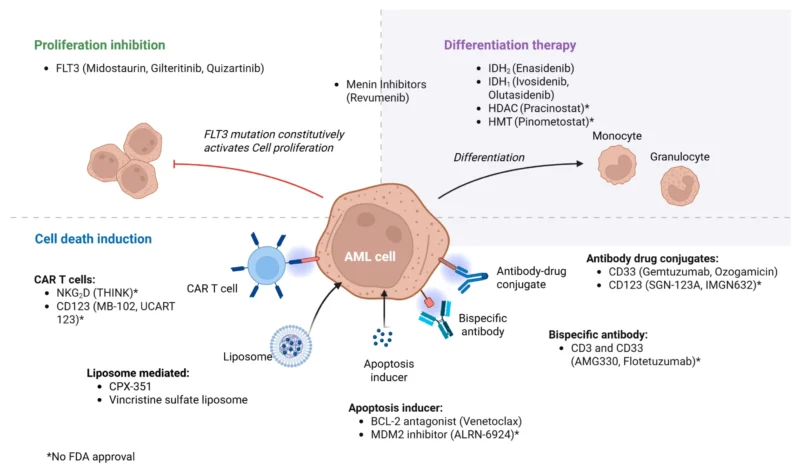
Current therapeutic paradigms in AML, emphasize the growing role of personalized medicine across age and risk groups. For younger, fit patients, intensive induction therapy with the classic 7 + 3 regimen remains the standard, with remission rates reaching 60-80%, particularly when combined with targeted agents. In FLT3-mutated AML, the RATIFY trial showed that adding Midostaurin to chemotherapy significantly improved overall survival, establishing FLT3-directed therapy as frontline standard care. Similarly, IDH1-mutated patients may benefit from Ivosidenib plus azacitidine, as demonstrated in the AGILE trial.
Older or medically unfit patients now commonly receive low-intensity therapy with hypomethylating agents plus Venetoclax. In the VIALE-A trial, azacitidine plus venetoclax achieved a CR rate of 66% vs 28% with azacitidine alone and improved median OS to 14.7 months vs 9.6 months. This combination is now considered a frontline standard for many older AML patients. CPX-351 improved survival compared with conventional 7 + 3 in older high-risk patients, supporting its use in therapy-related or secondary AML.
Post-remission strategies are risk-adapted: patients with favorable-risk disease typically receive 3-4 cycles of high-dose cytarabine, whereas those with intermediate- or adverse-risk features are generally recommended for allo-SCT in first remission. In CBF-AML and intermediate-risk AML with NPM1 mutations, MRD-positive patients derive greater benefit from transplantation. In QUAZAR AML-001, oral azacitidine significantly prolonged median OS to 24.7 months vs 14.8 months with placebo in older patients achieving remission after induction who were not candidates for transplantation (Kantarjian HM et al., 2025, Shimony et al., 2025).
Ongoing Expansion of the Therapeutic Landscape
CD47 is a macrophage immune-checkpoint “do not eat me” signal frequently overexpressed in AML and MDS. Magrolimab, an anti-CD47 monoclonal antibody, is designed to restore macrophage-mediated clearance of leukemic cells. In combination with azacitidine, magrolimab showed promising activity in TP53-mutated AML, achieving complete remission with incomplete count recovery rates of 75% in an initial cohort and, in a larger unfit population, an OR rate of 48.6% with a median OS of 10.8 months.
Menin is essential for the proliferation and survival of KMT2A- and NPM1-mutated AML. Four different menin-MLL1 inhibitors are currently being evaluated in early-phase clinical trials and have shown promising activity in R/R AML. In particular, single-agent SNDX-5613 achieved an OR rate of 55% in heavily pretreated patients, with a composite complete remission MRD-negative rate of 31%. Responses were also durable, lasting more than 6 months in six of the 12 patients who achieved composite complete remission.
Lintuzumab-Ac225 (Actimab-A) is an anti-CD33 radioimmunoconjugate that delivers the alpha-emitting radionuclide directly to leukemic cells. IMGN632 (Pivekimab sunirine), targets CD123 and is linked to a DNA-alkylating payload. It has demonstrated encouraging early clinical activity in R/R AML, with minimal toxicity to normal hematopoietic stem cells owing to its more selective target profile.
BiTEs and related bispecific antibodies are emerging immunotherapeutic strategies in AML. One of the best-studied examples is AMG330, which targets CD33 on blasts and CD3 on T cells. In a first-in-human dose-escalation study involving heavily pretreated patients, 8 of 60 evaluable patients achieved complete remission or morphologic leukemia-free state, while 37% of nonresponders still demonstrated at least a 50% reduction in bone marrow blasts, supporting meaningful biologic activity.
mdpi.com
The first phase I human trial of CD123 CAR-T cells by Budde et al. evaluated six heavily pretreated R/R AML patients after prior allogeneic transplantation. Two patients treated at the higher dose achieved complete remission and subsequently underwent further transplantation, remaining MRD-negative at 161 days post-transplant. Additional patients demonstrated meaningful blast reduction, including one case with marrow blasts decreasing from 77.9% to 0.9%. Toxicities, including CRS, adenoviral pneumonia, and recurrent cutaneous GVHD, were reversible. These findings support CD123 as one of the most promising CAR-T targets in AML.
Therapeutic vaccines and dendritic cell (DC)-based immunotherapies represent another investigational strategy. Vaccine targets such as WT1, proteinase 3, and RHAMM have shown favorable safety profiles, particularly in older patients. WT1-directed approaches are especially promising. A systematic review of 11 DC-vaccine trials reported complete or partial remissions, disease stabilization, and enhanced immune activation, including increased Th1 cytokines, CD4+ and CD8+ T cells, WT1-specific T cells, and activated NK cells. (Yadav et al., 2025, Ebinama et al., 2025, Jeurkar C et al., 2026).
Immunotherapy Challenges in AML
A biologically important subset of leukemic cells in AML consists of leukemic stem cells (LSCs), which possess the capacity for self-renewal, sustained leukemic repopulation, and marked resistance. Similar to normal HSCs, many LSCs exhibit cell-cycle quiescence, allowing them to evade cytotoxic therapies and persist after treatment, thereby contributing to relapse. Ongoing therapeutic efforts aim to overcome this through targeted immunotherapies. However, the heterogeneity of AML cells creates hurdles in identifying an optimal antigen that is preferentially expressed on LSCs rather than HSCs, while also ensuring efficient internalization of ADCs.
Spatial marrow profiling is emerging as a transformative tool in AML research because it preserves the architectural context of the bone marrow niche. These platforms allow analysis of how leukemic blasts interact with immune cells, stromal elements, and vasculature in their native locations, while identifying localized immune suppression, LSC niches, and protective microenvironments. In practical terms, spatial marrow profiling may help identify patients most likely to benefit from checkpoint inhibitors, bispecific antibodies, or macrophage-directed therapies (Cooper et al., 2025).
How Well Do Published Trial Outcomes Translate Into Real-World Practice?
An analysis of U.S. SEER data, representative of AML outcomes in community oncology practice, demonstrated results inferior to those reported in single-institution and cooperative group trials. Multiple factors likely explain this disparity, including selection of fitter patients for trials, exclusion of older patients or those with secondary AML, poor performance status, or organ dysfunction, differences in regimens offered, supportive care resources, and cumulative leukemia experience.
The SEER data nevertheless indicate significant survival improvements since 2000, particularly in distinct AML subtypes. In acute promyelocytic leukemia, 5-year survival reached ≥60% with incorporation of all-trans retinoic acid and arsenic trioxide, while in CBF-AML, 5-year OS reached ≥50% with gemtuzumab ozogamicin and high-dose cytarabine. Outcomes also improved in younger AML patients overall.
The high cost of AML treatment remains a major barrier, leaving many patients untreated or undertreated. Cost-effective strategies are urgently needed to ensure that these life-saving medications become accessible to the broader population. Future research efforts should coalesce around optimizing frontline therapy, integrating novel targeted agents earlier in treatment, advancing MRD assessment through more precise technologies, and refining the need, timing, and optimal use of HSCT. Strengthening psychosocial support remains an equally important component of comprehensive care.

You can also read: BCR::ABL1-Rearranged Chronic Myeloid Leukemia: Molecular Basis, TKI Resistance, and Emerging Therapies
Written by Susanna Mikayelyan, MD



