Myelodysplastic neoplasms (MDS) are a heterogeneous group of clonal bone marrow disorders marked by ineffective hematopoiesis and shaped by a complex interplay between genetic alterations and a dysregulated marrow microenvironment. This review explores the molecular and clinical complexity of MDS as a hematologic entity and sheds light on a particularly intriguing subtype that offers striking morphologic features alongside a unique therapeutic window – MDS with ring sideroblasts.
Concept and Classification of MDS
MDS are characterized by clonal proliferation of hematopoietic stem cells, recurrent genetic abnormalities, myelodysplasia, ineffective hematopoiesis, peripheral blood cytopenias, and an increased risk of progression to acute myeloid leukemia (AML). The incidence of MDS is approximately 4-5 cases per 100,000 people per year, but this likely underestimates the true rate. More comprehensive methods suggest the overall incidence may be 10-13, and in adults aged ≥65 years it can reach 75-162 per 100.000 per year.
In 1982, the French-American-British (FAB) group introduced the term “myelodysplasia” to describe morphologic abnormalities of myeloid cell lines in preleukemic conditions, later termed MDS. In 2001, under the aegis of the WHO, the FAB classification was revised to combine morphologic and genetic data into a clinically practical system, with the most recent update published in 2022.
The classification introduces the term myelodysplastic neoplasms (abbreviated MDS) to replace myelodysplastic syndromes, emphasizing their neoplastic nature and harmonizing terminology with MPN. A threshold of ≥10% dysplasia is recommended for all lineages. MDS entities are now grouped into those with defining genetic abnormalities and those defined morphologically. This reorganization improves classification by prioritizing genetically defined disease types and shifting away from traditional risk-based grouping (based on blast percentage, ring sideroblasts, and the number of dysplastic lineages) toward more comprehensive risk stratification systems.

Hypoplastic MDS (MDS-h) is listed as a distinct MDS type in this edition, characterized by a hypocellular marrow and immune-mediated pathogenesis involving cytotoxic CD8⁺ T-cell expansion with overproduction of IFN-γ and TNF-α. It shares clinical and biological overlap with aplastic anemia and PNH. Many patients respond to immunosuppressive therapies such as anti-thymocyte globulin, underscoring the importance of accurate diagnosis.
Evaluation requires careful morphologic assessment alongside cytogenetic and molecular studies to distinguish MDS-h from other marrow failure states. Importantly, individuals with germline pathogenic variants in GATA2, DDX41, Fanconi anemia, or telomerase complex genes may have hypoplastic bone marrow and can evolve to MDS and/or AML but typically do not respond to immunosuppression.
The boundary between MDS and AML is softened, with a balanced approach that eliminates blast cutoffs for most AML types defined by genetic alterations while retaining the 20% threshold to distinguish MDS from AML. However, there is broad agreement that MDS-IB2 may be considered AML-equivalent for therapeutic and clinical trial purposes when appropriate (Khoury JD et al., 2022).
There are precursor states that may precede MDS, including:
● clonal hematopoiesis of indeterminate potential (CHIP)
● clonal cytopenia of undetermined significance (CCUS)
CHIP is defined by the presence of a somatic mutation in a gene recurrently mutated in myeloid neoplasms with a variant allele frequency (VAF) of at least 2%, occurring in individuals with normal peripheral-blood cell counts and no evidence of hematologic malignancy.
In contrast, CCUS is characterized by persistent, otherwise unexplained cytopenia in one or more lineages, together with a somatic mutation in myeloid neoplasm-associated genes (VAF of ≥20%), but without fulfilling diagnostic criteria for MDS. While CHIP represents a lower-risk state, CCUS carries a higher risk of progression due to the presence of cytopenia and greater clonal burden.
What Drives the Disease Genetically?
The paradox of myelodysplastic hematopoiesis is that the founding mutation provides an advantage at the level of stem cells and progenitor cells, combined with a disadvantage at the level of hematopoietic precursors. Recurrently mutated genes include those involving RNA splicing, DNA methylation, histone modification, transcription regulation, DNA repair control, signaling, and the cohesin complex.
Only six genes (SF3B1, TET2, SRSF2, ASXL1, DNMT3A, and RUNX1) are mutated in at least 10% of patients who have MDS, with a long tail of additional genes that are mutated less frequently. Most mutations are C-to-T transitions at CpG dinucleotides, suggesting that they are due to age-related deamination of methylated cytosines. At the onset of clinical disease, the median number of driver mutations is two or three per patient. Spliceosome mutations are generally early genetic events that drive clonal dominance. They are heterozygous and mutually exclusive, most likely because of synthetic lethal interactions.
Most patients with CHIP carry a somatic mutation in an epigenetic regulator gene (DNMT3A, TET2, or ASXL1) and only a minority have a mutation in a spliceosome gene (SF3B1, SRSF2, or U2AF1). This is consistent with the hypothesis that persons with a spliceosome mutation have more rapid progression to MDS or CCUS, whereas those with a mutation in an epigenetic regulator can remain stable in the CHIP phase for years.
Although MDS are mainly sporadic diseases of older adults, increasing evidence shows that a subset of patients – often, but not only, those under 50 years – have a germline predisposition to myeloid neoplasms. In these individuals, somatic mutations develop on a background of inherited genetic alterations that promote higher mutation rates or selective clonal expansion. While individually rare, they may collectively account for up to 15% of MDS cases. Identifying germline mutations is important not only for genetic counseling but also for clinical decision-making, particularly in the context of transplantation (Cazzola M, 2020).
Underlying Pathophysiology in MDS
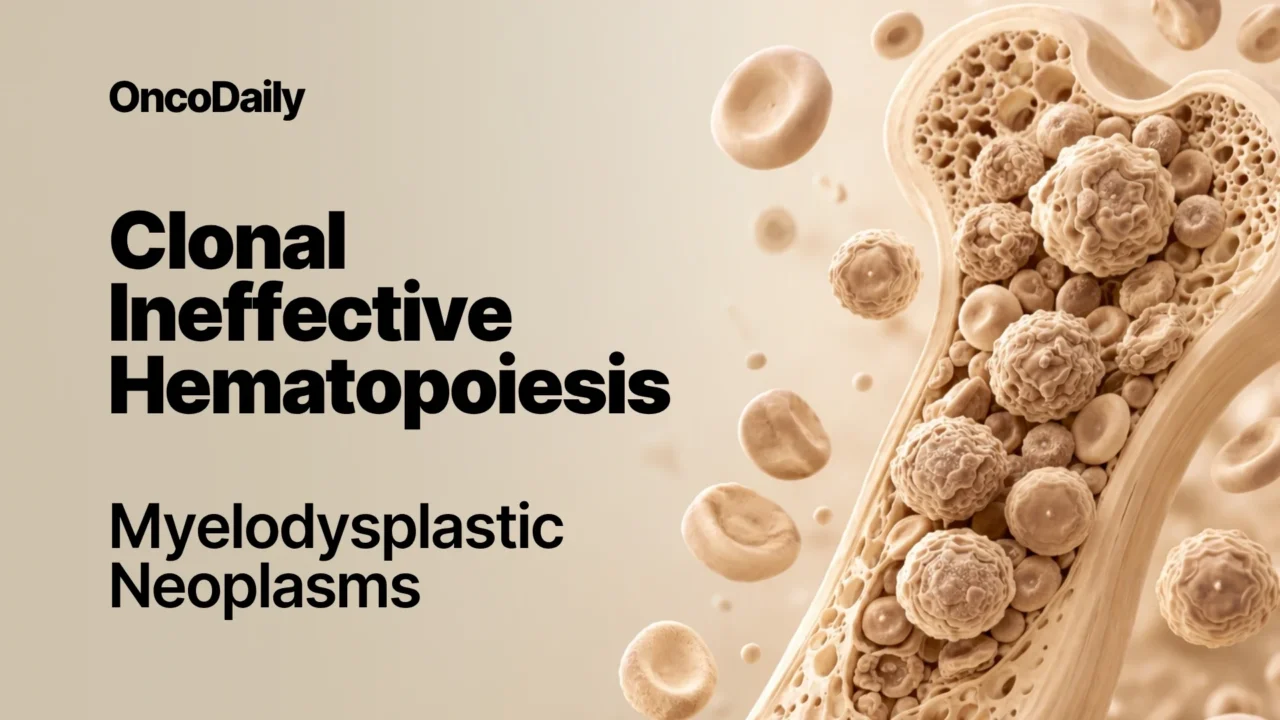
Distinct clinical phases can be identified in the process leading to MDS. During the first phase, an initiating driver mutation occurs in a hematopoietic stem cell, generating a local clone composed of mutant stem cells and abnormal hematopoietic progenitor and precursor cells.
The second phase is characterized by the spread of mutant stem cells through the peripheral blood, leading to the propagation of clonal hematopoiesis and the formation of new clones in different bone marrow regions. When hematopoietic cells carrying the somatic mutation account for at least 4% of all bone marrow cells (corresponding to a VAF of at least 2% for the mutation), the condition is defined as CHIP.
The third phase is marked by clonal dominance. Clonal hematopoiesis progressively expands and eventually becomes dominant in the bone marrow. This process is commonly associated with the occurrence of additional somatic mutations. Depending on the extent of clonal dominance, the degree of morphologic dysplasia, and the absence or presence and type of cytogenetic aberrations, this condition can meet the diagnostic criteria for MDS or CCUS.
The fourth phase (secondary AML) is characterized by clonal selection and leukemic transformation. The acquisition of additional driver mutations or the emergence of preexisting ones leads to selection of subclones of hematopoietic cells with increasingly impaired differentiation capacity. When the proportion of blast cells increases to 20% or more, a diagnosis of secondary AML can be made.
Immune dysregulation is a key factor in MDS pathogenesis. Early-stage MDS is marked by a proinflammatory bone marrow environment, with expanded cytotoxic CD8+ T lymphocytes (CTLs) and reduced Tregs, defined by Foxp3 transcription factor expression. In contrast, Tregs and myeloid-derived suppressor cells (MDSCs) expand in advanced disease. Recently, the immunosuppressive function of Tregs has been linked to expression of the exon-2-containing Foxp3 isoform (Foxp3-Ex2), and Treg frequency was proposed as a prognostic factor in MDS. The cytokine milieu shapes Treg and MDSC differentiation and function. TGF-β involvement in such processes and in modulating CTL function has been recognized.
Risk Assessment and Stratification
Traditionally guided by the IPSS-R, MDS risk stratification has evolved into a molecular era with the development of the updated IPSS-M model.
The IPSS-R relies on five hematologic and cytogenetic features:
● hemoglobin
● absolute neutrophil and platelet count
● bone marrow blasts
● cytogenetic abnormalities
Median overall survival decreases progressively from 8.8 years in very low-risk patients to 5.3 years in low-risk, 3.0 years in intermediate-risk, 1.6 years in high-risk, and 0.8 years in very high-risk groups. Clinically, these categories are often consolidated into lower-risk disease (IPSS-R ≤3.5), with a median overall survival of approximately 5.9 years, and higher-risk disease (IPSS-R >3.5), with a median survival of about 1.5 years (Greenberg PL et al.,2012).
IPSS-M represents an evolution of MDS risk stratification by integrating somatic gene mutations. Developed from a cohort of 2,957 patients and validated in an external cohort of 754 patients, it incorporates mutations in 31 genes to generate individualized risk scores across six prognostic categories. At least one oncogenic genomic alteration is identified in approximately 94% of patients. Adverse outcomes are strongly associated with alterations such as multi-hit TP53, FLT3 mutations, and MLL-PTD, while SF3B1 mutations are generally linked to favorable prognosis, influenced by co-mutation patterns. Compared with IPSS-R, IPSS-M improves prognostic discrimination and reclassifies about 46% of patients (Bernard E et al., 2022).
Although IPSS-M provides the most accurate prediction of median OS, the frequent lack of molecular data remains an obstacle in daily clinical practice. To address this, alternative tools such as AIPSS-MDS (Artificial Intelligence Prognostic Scoring System for MDS) have been developed by the Spanish MDS Group, using only clinical and cytogenetic data through machine learning. While this approach has proven useful when molecular testing is not accessible, it is less precise than IPSS-M. In practice, IPSS-R remains the most widely used system (Schulz F et al., 2025).
Treatment Decision-Making in MDS
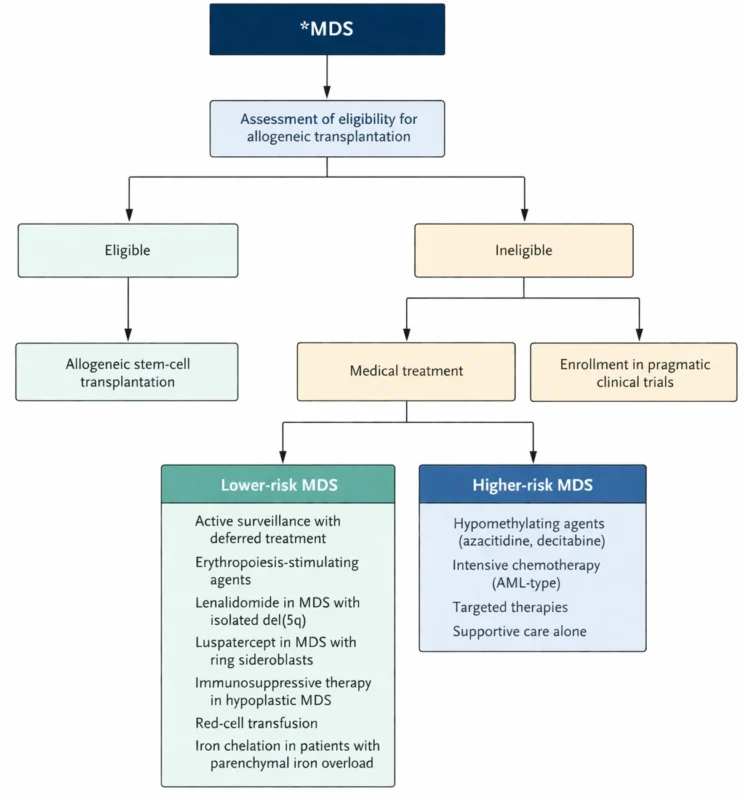
A patient-centered approach is essential in MDS management and is mainly based on risk stratification. Not all patients require immediate treatment. When therapy is indicated, the first step is to assess eligibility for allogeneic stem-cell transplantation – the only potentially curative option, though associated with significant morbidity and mortality. This decision should be individualized through shared decision-making, incorporating patient preferences, performance status, comorbidities, and biologic rather than chronological age.
While all patients with higher-risk MDS should be evaluated for transplantation at diagnosis, only a subset of lower-risk patients are candidates early in the disease course. Fit patients, even into their 70s, may be considered, with 5-year survival rates of approximately 40-50%. In contrast, patients with poor performance status or significant comorbidities are better suited for non-transplant therapies. Importantly, adverse genetic features (particularly biallelic TP53 mutations) are strongly associated with relapse and inferior survival outcomes (Niscola P et al., 2024).
A Closer Look at MDS-SF3B1
MDS-SF3B1 is a distinct subtype characterized by ineffective erythropoiesis, macrocytic anemia, and the presence of ring sideroblasts. Diagnosis is established via bone marrow aspiration in the presence of either ≥15% RS or 5–14% RS and an SF3B1 mutation (Mortuza S et al., 2024).
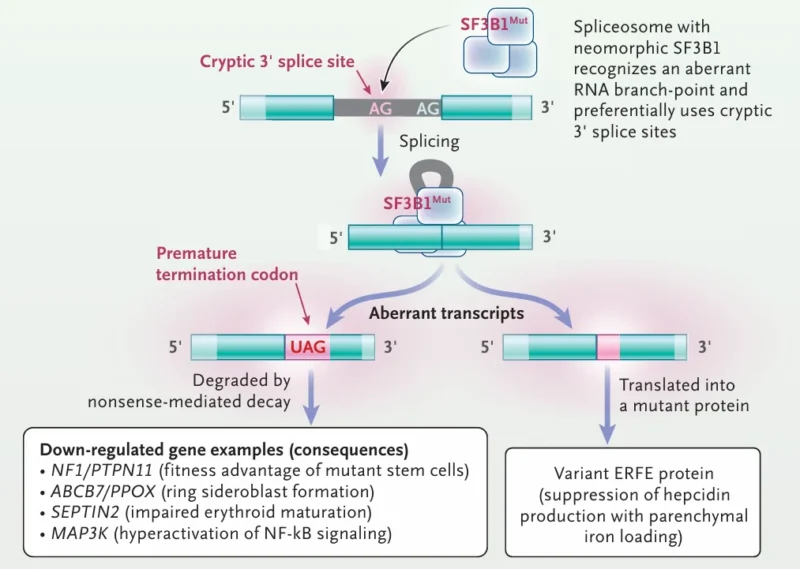
Hematopoietic cells are heterozygous for the mutation, so in each cell, roughly half of the splicing events are carried out by normal spliceosomes, while the remaining half involve spliceosomes containing the mutant SF3B1 splicing factor. This mutation alters RNA branch-point recognition, leading to preferential use of cryptic 3′ splice sites, resulting in the insertion of nucleotides at the authentic exon–exon junction.
Only trace amounts of aberrant transcripts of several genes are detectable in SF3B1-mutated myelodysplastic cells. Most aberrant transcripts are rapidly degraded by nonsense-mediated decay, primarily because the inserted nucleotide sequence introduces a premature termination codon. The overall effect is reduced production of canonical transcripts, and consequently, lower protein levels. This reduction impacts multiple genes, with diverse functional consequences.
www.nejm.org
Importantly, ABCB7, a transporter in the mitochondrial membrane, is affected by missplicing. ABCB7 is described as a transporter that exports an as-yet unidentified product of iron–sulfur (Fe–S) cluster assembly from the mitochondrial matrix to the cytosol. When ABCB7 is defective, cytosolic aconitase is deprived of its Fe–S cluster and converted into iron regulatory protein 1 (IRP1), a shift that is sensed as cellular iron deficiency and consequently drives increased iron uptake. This represents only part of the story.
In erythroid cells, ABCB7 also participates in heme biosynthesis by stabilizing ferrochelatase, the final enzyme in the pathway that inserts Fe²⁺ into protoporphyrin IX. When ABCB7 is dysfunctional, ferrochelatase loses stability and can’t efficiently incorporate Fe²⁺ into heme. As a result, iron accumulates in mitochondria, even though ferrochelatase itself is neither mutated nor underexpressed, ultimately contributing to the sideroblastic phenotype.
Systemic iron overload arises from ERFE mutations, in which erythroferrone, an erythroid regulator of hepcidin, produces a variant protein. If the signal is active over a long time, it precipitates systemic iron overload even before the patient becomes transfusion dependent(Cazzola M, 2020, Gattermann N, 2024).
Mainstay of Treatment for Patients With MDS-SF3B1
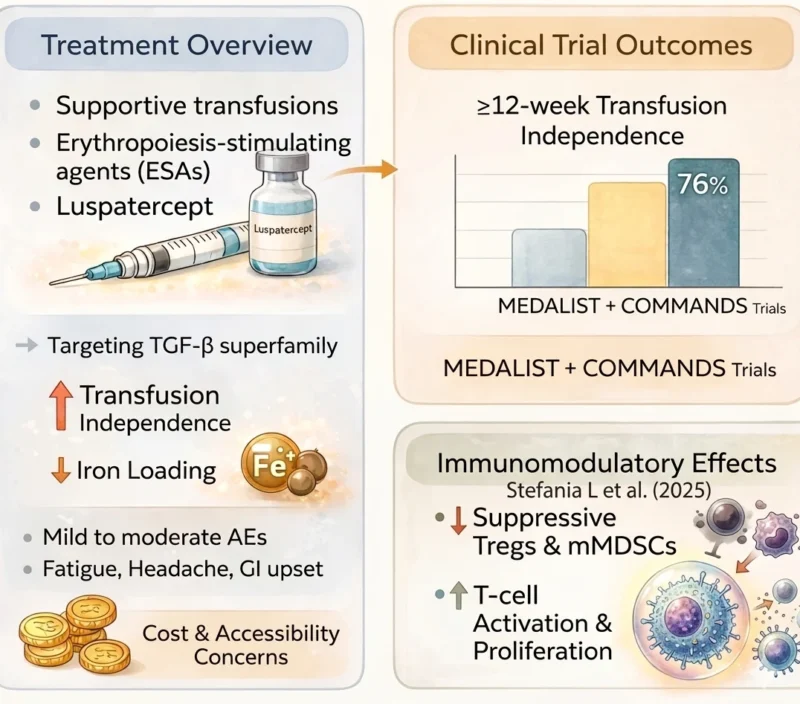
Currently, treatment relies on supportive transfusions, erythropoiesis-stimulating agents (ESAs), and erythroid maturation agents such as luspatercept. This first-in-class agent targets TGF-β superfamily ligands to inhibit Smad2/3 signaling and enhance late-stage erythropoiesis. Luspatercept reduces transfusion needs and limits further iron loading but does not significantly decrease existing iron overload.
Clinical trials, including MEDALIST and final analyses of the COMMANDS trial, have shown that luspatercept achieves higher and more durable transfusion independence than epoetin alfa in lower-risk MDS, with up to 76% of patients achieving ≥12-week transfusion independence and prolonged response durations (Garcia-Manero G et al., 2025).
Given that immune dysregulation in MDS represents both a pathogenetic feature and an independent prognostic marker, Stefania L et al. (2025) investigated the immunomodulatory effects of luspatercept in 13 patients with MDS-RS treated between January 2021 and April 2023, with response assessed at 24 weeks (T1), alongside an additional cohort of 7 long-term responders (median follow-up 36 months, T2). All patients received luspatercept at 1 mg/kg. The study showed that treatment reduced suppressive Tregs and mMDSCs, while increasing T-cell activation and proliferation.
Favorable responses were associated with baseline immune profiles characterized by low mMDSCs and increased cytotoxic T-lymphocyte activity, consistent with prior findings linking higher lymphocyte counts to better outcomes. These results suggest that luspatercept modulates the immune-regulatory network, likely through TGF-β-dependent pathways, promoting sustained immune activation and reduced immunosuppression in long-term responders. Additionally, effective residual erythropoiesis was inversely associated with immunosuppressive profiles, supporting the prognostic value of immune parameters, although validation in larger MDS-RS cohorts remains necessary.
Luspatercept is generally well tolerated, with mostly mild to moderate adverse effects, including fatigue, headache, dizziness, diarrhea, nausea, and musculoskeletal pain, less commonly, hyperuricemia and thromboembolic events may occur. Monitoring is recommended to avoid excessive increases in hemoglobin, particularly in patients with underlying cardiovascular risk. However, its use may be limited by cost and accessibility, potentially restricting its availability in routine clinical practice.
What is the Rationale for Iron Chelation Therapy?
Given the tendency of this subtype to lead to iron overload, together with transfusion-related iron accumulation, another important question arises in practice: what is the rationale for ICT?
Even if iron-related complications add up and act as a strong risk factor, their effect may hide behind the common causes of death in older patients. It is therefore difficult to determine the extent to which iron overload contributes to morbidity and mortality in older patients with MDS.
However, an SF >1000 ng/mL showed a clear dose-dependent impact on overall survival in patients with low-risk MDS, and remained an independent prognostic factor even after adjusting for transfusion burden. In addition, TSAT levels >70-80% can serve as a surrogate marker for toxic labile plasma iron, which is not routinely measurable in clinical practice, such levels are also a prerequisite for parenchymal iron loading, and TSAT >80% is associated with inferior clinical outcomes in MDS.
Data from the prospective European LeukemiaNet MDS (EUMDS) Registry, adjusted for relevant prognostic factors, demonstrated improved survival with ICT. In a large US Medicare-based cohort, deferasirox (DFX) was associated with a reduced risk of death, particularly with longer treatment duration among patients receiving ≥20 transfusions. A meta-analysis of 9 observational studies also showed a significantly lower mortality risk with ICT (adjusted hazard ratio 0.42, 95% CI 0.28-0.62, P < 0.01), despite some heterogeneity across studies.
The only prospective randomized trial of ICT in MDS is the Telesto study, which showed a significant advantage for DFX versus placebo, with event-free survival as the primary endpoint, defined as the time from randomization to the first documented nonfatal event (cardiac, hepatic dysfunction or transformation to AML) or death, the delayed clinical benefit was plausible, as iron-related organ damage developed gradually and the effects of ICT required time to become evident (Garcia-Manero G, 2023, Gattermann N, 2024).
Overall, ICT in MDS may provide multiple clinical benefits, including reduced cardiac, hepatic, and metabolic complications, fewer infections, and improved hematopoietic function. It has also been associated with a lower risk of leukemic transformation and better outcomes following allo-HSCT, with these effects collectively suggesting a potential improvement in overall survival.

You can also read Saint Carlo Acutis: Faith, Courage, and Cancer Journey of the First Millennial Saint on OncoDaily.
Written by Susanna Mikayelyan, MD
FAQ
What defines myelodysplastic neoplasms (MDS)?
MDS are clonal myeloid disorders characterized by ineffective hematopoiesis, dysplasia, cytopenias, recurrent genetic abnormalities, and a risk of progression to acute myeloid leukemia (AML).
How are MDS currently classified into main categories?
Modern MDS classification distinguishes between entities driven by defining genetic abnormalities and those categorized based on morphologic features, aligning diagnosis more closely with underlying disease mechanisms.
What is the difference between IPSS-R and IPSS-M?
Risk stratification in MDS has evolved from the IPSS-R, which uses clinical and cytogenetic parameters (hemoglobin, neutrophils, platelets, bone marrow blasts, and cytogenetics), to the IPSS-M, which incorporates somatic gene mutations. IPSS-M provides more precise, individualized prognostic assessment, improves risk prediction, and reclassifies a significant proportion of patients based on molecular features.
What is the role of genetic mutations in MDS, and is there a germline predisposition?
Genetic mutations drive clonal hematopoiesis and disease progression. Commonly affected pathways include RNA splicing, DNA methylation, and transcription regulation. While most cases are acquired, a subset of patients have a germline predisposition, particularly in younger individuals, which influences disease risk and clinical management.
What are the key pathophysiological phases leading to MDS and AML?
MDS develops through sequential phases: initial clonal expansion of a mutated stem cell, propagation of clonal hematopoiesis (CHIP), progression to clonal dominance with additional mutations (CCUS/MDS), and eventual leukemic transformation into AML driven by further genetic evolution and impaired differentiation
How are treatment decisions made in MDS?
Treatment decisions in MDS are guided by risk stratification and a patient-centered approach. Key factors include disease risk category, symptoms, cytopenias, genetic features, performance status, comorbidities, and patient preferences, with consideration of eligibility for allogeneic stem cell transplantation in appropriate candidates.
How does the SF3B1 mutation contribute to ring sideroblast formation?
SF3B1 mutations alter RNA splicing, leading to reduced expression of key mitochondrial proteins such as ABCB7. This disrupts iron–sulfur cluster metabolism and heme synthesis, resulting in mitochondrial iron accumulation and ring sideroblast formation.
Why does iron overload occur in MDS patients, and how does it affect outcomes?
Iron overload in MDS arises from both chronic transfusions and dysregulated iron metabolism, where ineffective erythropoiesis suppresses hepcidin, increasing iron absorption and promoting systemic accumulation. This excess iron can lead to cardiac and hepatic damage, increased infection risk, and may negatively impact survival.
What are the current Management options for MDS with ring sideroblasts?
Management includes supportive transfusions, erythropoiesis-stimulating agents (ESAs), and luspatercept, an erythroid maturation agent that enhances late-stage erythropoiesis and reduces transfusion dependence.
What is the evidence supporting iron chelation therapy in MDS, and when should it be performed?
The Telesto trial demonstrated improved event-free survival with deferasirox (DFX), suggesting that reducing iron burden may delay organ damage and disease progression. Iron chelation therapy is typically considered in lower-risk, transfusion-dependent patients with evidence of iron overload, such as elevated ferritin (>1000 ng/mL).