Philadelphia chromosome (Ph)-positive or molecularly-rearranged BCR::ABL1 chronic myeloid leukemia (CML) is a myeloproliferative hematopoietic stem cell neoplasm, that accounts for approximately 15% of adult leukemia cases. The advent of TKIs has revolutionized CML management, improving survival rates and transforming it into a highly prevalent chronic malignancy with an estimated global prevalence of 4-5 million.
Despite their well-deserved recognition as frontline therapy, acquired resistance remains a major challenge, driving the search for alternative and complementary strategies. This review explores the molecular and clinical landscape of CML, the mechanisms underlying TKI resistance, and evolving approaches aimed at overcoming it.
Molecular Drivers and Pathophysiology of CML
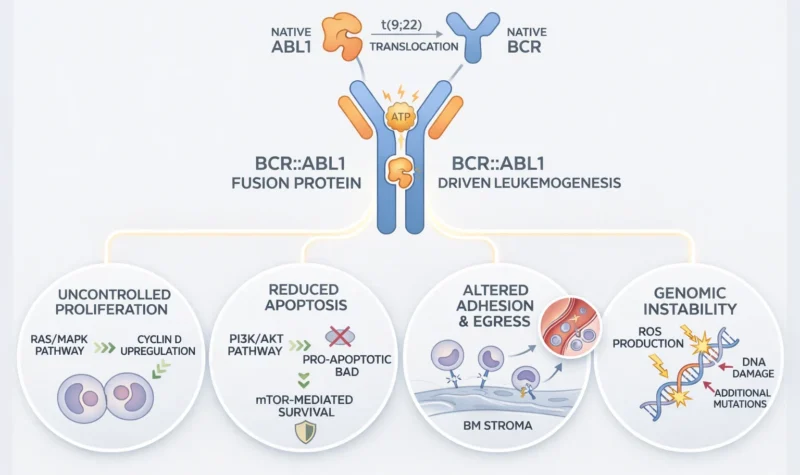
The defining molecular lesion, the BCR::ABL1 fusion gene, arises from the reciprocal translocation t(9;22)(q34;q11), which juxtaposes the breakpoint cluster region (BCR) gene on chromosome 22 with the ABL1 proto-oncogene on chromosome 9. This acquired rearrangement produces a chimeric protein with constitutive tyrosine kinase activity that drives leukemogenesis.
Unlike native ABL1, whose activity is tightly regulated by intramolecular inhibitory domains, the fusion protein adopts an open conformation that promotes continuous ATP binding and substrate phosphorylation. This leads to persistent activation of multiple downstream signaling cascades that collectively drive uncontrolled proliferation, reduced apoptosis, altered adhesion, and genomic instability.
The principal signaling pathways include:
- RAS/MAPK – promotes cell cycle progression through upregulation of cyclin D and other proliferative mediators.
- PI3K/AKT – enhances cell survival by inhibiting pro-apoptotic factors such as BAD and promoting mTOR-mediated protein synthesis.
- JAK/STAT (particularly STAT5) – promotes cytokine-independent growth, survival, and self-renewal of leukemic progenitors.
In addition, BCR::ABL profoundly alters interactions between hematopoietic cells and the bone marrow microenvironment. Leukemic cells exhibit reduced dependence on stromal support and decreased adhesion to ECM components, partly due to altered integrin signaling. This contributes to the egress of myeloid cells into the peripheral blood – a hallmark feature of CML. It also increases ROS production, promoting DNA damage and genomic instability, which over time leads to additional cytogenetic abnormalities and mutations driving disease progression (Sicuranza A et al., 2025).
Clinical Profile and Genetic Diversity
Historically, CML was classified into three phases – chronic (CML-CP), accelerated (CML-AP), and blastic (CML-BP). The recent WHO classification has eliminated CML-AP, incorporating its high-risk features into risk stratification models within CML-CP, reflecting improved overall survival.
More than 90% of patients are diagnosed in the CML-CP. 50-60% of patients are diagnosed accidentally, without manifesting any symptoms during a routine physical examination or blood test. When symptoms or signs are present, they are often related to anemia or splenomegaly (fatigue, malaise, weight loss, left upper quadrant fullness or pain).
Less often, symptoms are associated with:
- hyperleukocytosis/thrombocytosis (blood clots, priapism, cardiovascular or vaso-occlusive events, visual disturbances due to retinal bleeding)
- severe basophilia (skin itching, diarrhea, peptic ulcers)
- high tumor burden of neglected CML or disease transformation (massive splenomegaly, persistent fevers, gout, infections, thrombocytopenia and bleeding other complications)
CML diagnosis typically relies on detecting the Philadelphia chromosome by cytogenetics or identifying BCR::ABL1 using FISH or PCR. Though peripheral blood and cytogenetic/molecular tests usually establish CML, bone marrow evaluation provides more accurate blast and basophil assessment for staging. The CML risk profile is determined by commonly used risk scores like Sokal Score and ELTS score (uses age, spleen size, platelet count, and peripheral blood blast percentage), and more recently through consideration of concurrent genomic aberrations (Kantarjian H et al., 2025).
BCR::ABL isoforms are the result of variant exon fusions during the translocation process. Most rearrangements produce the p210 oncoprotein (e13a2 or e14a2 transcripts), while less common variants including p190 and p230, may be associated with distinct clinical behavior. Rare transcript variants can be missed by standard PCR assays, leading to false-negative molecular results. Therefore, baseline RT-PCR is essential to detect atypical BCR::ABL1 transcripts and ensure accurate monitoring during TKI therapy.
Certain additional chromosomal abnormalities (ACAs), including i(17q), -7/del(7q), and 3q26.2 (MECOM) rearrangements, are associated with poor prognosis. Concurrent myeloid mutations are present in a subset of newly diagnosed CML cases, most commonly involving ASXL1, and less frequently DNMT3A, IDH1/2, EZH2, and TET2. Among these, ASXL1 mutations are linked to recurrent cytopenias during TKI therapy, inferior treatment responses, and an increased likelihood of requiring HSCT (Jayavel S et al., 2025).
Contemporary Approaches to Therapy
Most therapeutic strategies in CML rely on TKIs targeting BCR::ABL1, with several available options:
- imatinib (1st generation)
- dasatinib, bosutinib, nilotinib (2nd)
- ponatinib, asciminib (3rd)
The term “third-generation” also includes investigational agents such as olverembatinib, ELVN-001, TGRX-678, and TERN-701.
Based on binding mechanism, they are classified as:
- DFG-in (binding the active kinase conformation)
- DFG-out (binding the inactive conformation, showing greater activity against certain resistant variants)
BCR::ABL kinase domain consists of an N-lobe and C-lobe, with the activation loop regulating transitions between active and inactive conformations. Key residues include the gatekeeper Thr315 in the ATP-binding site, which affects inhibitor binding, as well as catalytic and regulatory residues that control substrate phosphorylation and kinase activity.
Most TKIs, including imatinib, second-generation agents, ponatinib, olverembatinib, and ELVN-001, inhibit the ABL1 kinase domain. In contrast, asciminib, TGRX-678, and TERN-701 act via the ABL myristoyl pocket, inducing an inactive conformation independent of the ATP-binding site (STAMP mechanism) and are therefore called STAMP inhibitors.
The four main goals of CML therapy are:
- improve survival – 10-year OS 82-87% and relative survival 90-95% achieved
- achieve a durable DMR, which may lead to a treatment-free remission status – BCR::ABL1 (IS) <0.01%
- reduce short- and long-term side-effects – relatively novel concept of developing anti-cancer targeted therapies at an OBD rather than the traditional below the MTD.
- provide a good treatment value – affordable generics, new TKIs must remain cost-effective to be viable frontline options
Intermittent TKI administration and treatment de-escalation are alternatives to dose reduction or discontinuation and can improve quality of life. In older CML patients, these strategies are safe, with reversible loss of response and no progression. In the OPTkIMA trial, TKI de-escalation versus intermittent dosing was associated with higher MMR loss (46% vs 27%) but greater TFR eligibility (58% vs 36%), with similar QoL.
Several studies have shown that longer TKI treatment and deeper, sustained responses are associated with higher TFR rates. A durable DMR of at least 2 years leads to TFR rates of 40–50%, with rates increasing to ≥80% when DMR is maintained for 5 years or more. (Sun L et al., 2024, Kantarjian H et al., 2025).
Role of Transplantation
When imatinib became available in 2000, a CML expert joked that this does not mean “Stop Transplant Immediately”, but rather “Select Transplant Intelligently”. In the German CMLIV trial, only 9% of patients with CML-CP on imatinib-based regimens underwent allo-SCT. It should be considered in patients with T315I mutation, MECOM rearrangement, and in patients with recurrent cytopenias on TKI therapy requiring multiple treatment interruptions (often associated with ASXL1 mutations).
Patients with de novo CML-AP are treated with second-generation TKIs and have an estimated 8-year survival of 60–80%. In contrast, patients progressing from CML-CP to CML-AP have a poorer prognosis (median OS 3 years), they are best managed with combination therapy (preferably a 3rd generation TKI plus hypomethylating agents) followed by allo-SCT upon achieving a good response (Akiyama H et al., 2024).
Resistance Pathways in Focus
Resistance remains the Achilles’ heel of TKI therapy and is broadly classified into two main categories:
- BCR::ABL1-dependent
- BCR::ABL1-independent
In addition to mutations, increased BCR::ABL1 expression, often driven by gene amplification, can contribute to resistance. Higher protein levels can overwhelm the inhibitory capacity of TKIs and should be considered in patients with rising transcript levels despite therapy.
BCR::ABL1-dependent resistance primarily arises from mutations within its kinase domain, which are detected in 55% of imatinib-resistant patients and occur in 40-60% of cases with secondary resistance. These mutations (over 90) directly impact the TKI binding site, mostly by steric hindrance, conformational changes, or alterations in the electrostatic interactions.
The T315I mutation is particularly notorious for conferring near-complete resistance to most TKIs, except ponatinib and asciminib. In the PACE trial, ponatinib achieved an MMR rate of 58%, with 5-year PFS of 53% and OS of 73%. Those with mutations sensitive to second-generation TKIs, such as Y253H or E255K, may benefit from dasatinib or nilotinib. Patients with compound mutations often require more aggressive treatment strategies, including allo-SCT or experimental therapies (Pierro F et al., 2025).
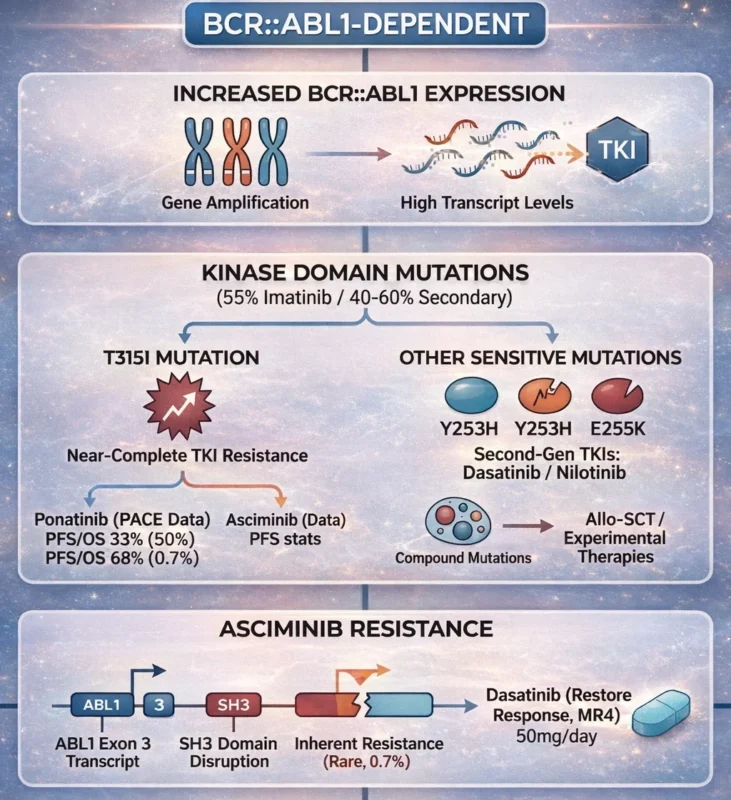
Asciminib, a first-in-class STAMP inhibitor, requires intact SH2/SH3 domains for activity. Accordingly, rare BCR::ABL1 variants involving ABL1 exon 3 disrupt the SH3 domain and can confer primary resistance to asciminib, supported by in vitro and structural data (Jung B et al., 2025).
Although uncommon (0.7% of newly diagnosed cases in their laboratory, 2021-2024), clinical evidence aligns with this finding, as a patient with an exon 3 transcript showed inherent resistance. These variants also complicate molecular monitoring, with BCR::ABL1/ABL1 ratios exceeding 100% due to lack of exon 2 amplification. After 3 months of asciminib treatment, dasatinib was reintroduced at a reduced dose. At 50 mg/day, response was successfully restored, achieving a deep molecular response (MR4).
Non-BCR::ABL1-Mediated Resistance
This type of resistance arises from activation or mutation-driven upregulation of alternative survival pathways (PI3K/AKT/mTOR, RAS/MAPK, JAK/STAT). The clinical implications of these pathways are significant, as combining TKIs with pathway-specific inhibitors may overcome resistance.
Leukemia stem cells (LSCs) play a crucial role in both primary and secondary resistance, which are relatively insensitive to tyrosine kinase inhibition. These cells reside in protective bone marrow niches and maintain survival through activation of alternative signaling pathways such as Wnt/β-catenin, Hedgehog. Combining TKIs with β-catenin inhibitors may offer a promising strategy, while clinical trials of Hedgehog pathway inhibitors are ongoing.
TKI efficacy is regulated by drug transporters: reduced influx (OCT1/OCTN2) and increased efflux lower intracellular drug levels, promoting resistance and suboptimal responses, thus, transporter variability and genetics are key determinants of TKI response and targets for personalized therapy.
Dysregulation of DNA damage response (DDR) pathways – driven by BCR::ABL1-induced genomic instability, oxidative stress, and error-prone DNA repair – promotes resistance. Epigenetic dysregulation – driven by altered DNA methylation, histone modifications, microRNA expression, and metabolic reprogramming – contributes to resistance, with mutations in regulatory genes becoming more prevalent during disease progression (Lahmouad M et al., 2025, Shvachko L et al., 2025).
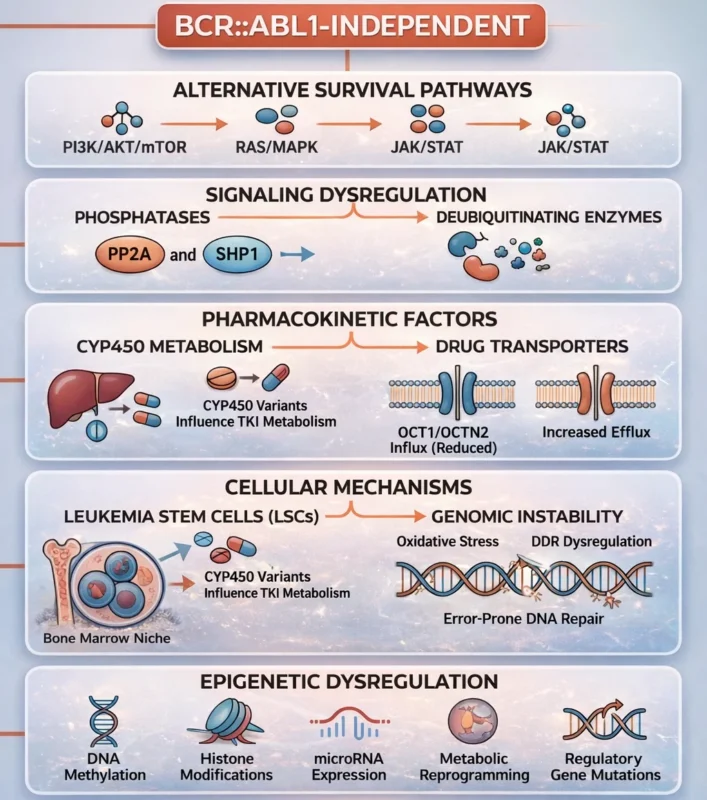
Recent studies show that changes in phosphatases (PP2A, SHP1) can drive TKI resistance by altering signaling, with reduced PP2A activity particularly linked to resistance. Deubiquitinating enzymes contribute to resistance by regulating protein stability and degradation pathways affecting BCR::ABL1 signaling. Additionally, pharmacogenetic variants in cytochrome P450 enzymes influence TKI metabolism linking liver function and genomic profile to variability in drug response and toxicity.(Lahmouad M et al., 2025, Shvachko L et al., 2025).
Emerging Approaches to Overcome Resistance
Strategies to overcome TKI resistance are being actively explored. One such approach is the use of BCL-2 inhibition, which has emerged as a potential game-changer in targeting CML stem cells resistant to TKIs. However, monotherapy approaches have limitations, leading to growing interest in combination with immunotherapies.
Ongoing clinical trials evaluate combinations of TKIs with anti-PD-1 and anti-CTLA-4 agents. In parallel, CAR T-cell therapy, which has demonstrated remarkable success in other hematologic malignancies, is now being explored for TKI-resistant CML. Synthetic lethality is emerging as another promising strategy, in which researchers aim to identify and target genes or pathways that, when inhibited alongside BCR::ABL1, selectively induce leukemic cell death while sparing normal cells.
In recent years, A Chinese group investigated a novel class of selective BCR::ABL1 T315I PROTAC degraders, among which some compounds demonstrated potent degradation activity in vitro and in vivo, achieving 90.8% tumor growth inhibition in a xenograft model. Another study analyzed the interaction between protein-tyrosine phosphatase 1B (PTP1B) and BCR::ABL1 using SBF-1, showing that disruption of interaction induces degradation of BCR::ABL1, including T315I-mutated forms. Quezada Meza et al. demonstrated that inhibition of protein kinase CK2 reduces aberrant signaling and induces apoptosis in T315I-mutated CML cells.
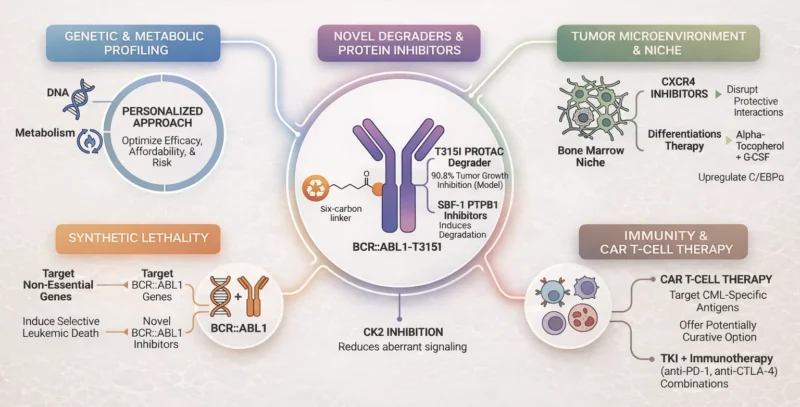
Increasing attention is being given to the tumor microenvironment and its role in CML progression and drug resistance. Leukemic cells interact with the bone marrow niche in ways that can promote survival and limit TKI efficacy. Strategies aimed at disrupting these protective interactions, such as CXCR4 inhibitors, are being explored to weaken the supportive environment.
Differentiation therapy may represent a potential therapeutic approach in CML. Alpha-tocopherol may be one of the agents with differentiation-inducing potential in CML blast cells. In vitro studies have shown that alpha-tocopherol, in combination with G-CSF, upregulates genes associated with differentiation in K562 CML cells in vitro, particularly the myeloid master regulator C/EBPα (Shvachko L et al., 2025, Quezada et al., 2025).
The growing availability of next-generation TKIs and combination therapies offers new opportunities, but also brings challenges related to cost and side effects. Optimizing treatment requires a more personalized approach that considers drug efficacy, affordability, and patient-specific risk factors. At the same time, integrating genetic profiling and understanding how metabolic differences affect drug response might be the key to improving long-term outcomes in CML.
You can also read Acute Myeloid Leukemia: What patients should know about on OncoDaily.

Written by Susanna Mikayelyan, MD
FAQ
What causes chronic myeloid leukemia (CML)?
CML is caused by the formation of the BCR::ABL1 fusion gene, resulting from the t(9;22) translocation (Philadelphia chromosome). This leads to constitutive tyrosine kinase activity that drives uncontrolled cell proliferation.
How does BCR::ABL1 cause leukemia?
The BCR::ABL1 protein has constant tyrosine kinase activity, activating pathways like RAS/MAPK, PI3K/AKT, and JAK/STAT, which promote proliferation, survival, and genomic instability.
What are the common symptoms of CML?
Many patients are asymptomatic at diagnosis, but symptoms can include fatigue, weight loss, night sweats, and splenomegaly-related discomfort.
What are tyrosine kinase inhibitors (TKIs) in CML?
Tyrosine kinase inhibitors (TKIs) are targeted drugs that block the abnormal BCR::ABL1 kinase, preventing leukemic cell growth and survival. They are the cornerstone of CML treatment and can achieve long-term disease control.
Why does resistance to TKIs develop in some patients?
Resistance can occur due to kinase domain mutations, activation of alternative pathways, pharmacokinetic factors, or persistence of leukemia stem cells.
Are there biomarkers that predict response to TKI therapy?
Yes, factors such as BCR::ABL1 transcript levels, kinase mutations, and additional genomic abnormalities can influence treatment response and prognosis.
What is the significance of the T315I mutation in CML?
The T315I mutation is a “gatekeeper” mutation that confers near-complete resistance to most TKIs, except for agents like ponatinib and asciminib.
Can patients with CML stop treatment after achieving remission?
Yes, some patients achieving a deep molecular response (DMR) may attempt treatment-free remission (TFR), especially after sustained response for several years.
What emerging therapies are being explored for resistant CML?
New approaches include BCL-2 inhibitors, immunotherapy, PROTAC degraders, and combination therapies targeting multiple resistance pathways.
What is the role of stem cell transplantation in CML?
Allogeneic stem cell transplantation (allo-SCT) is now reserved for high-risk, TKI-resistant CML, or treatment failure. It remains the only potentially curative option but is used selectively due to its risks.



