Rarely in hematology does such profound clinical severity arise from such an empty bone marrow. Patients with Aplastic Anemia (AA) may present with life-threatening infections, bleeding and profound pancytopenia, while the marrow is markedly hypocellular rather than infiltrated by malignant cells.
Once viewed as a purely nonmalignant disorder, AA now occupies a more complex position in hematology, where marrow failure, clonal hematopoiesis and leukemic evolution appear far closer than thought. As molecular profiling continues to reveal the overlapping nature of bone marrow failure disorders (BMFs), understanding the main concepts has become increasingly important in contemporary clinical practice.
What Are the Origins of AA?
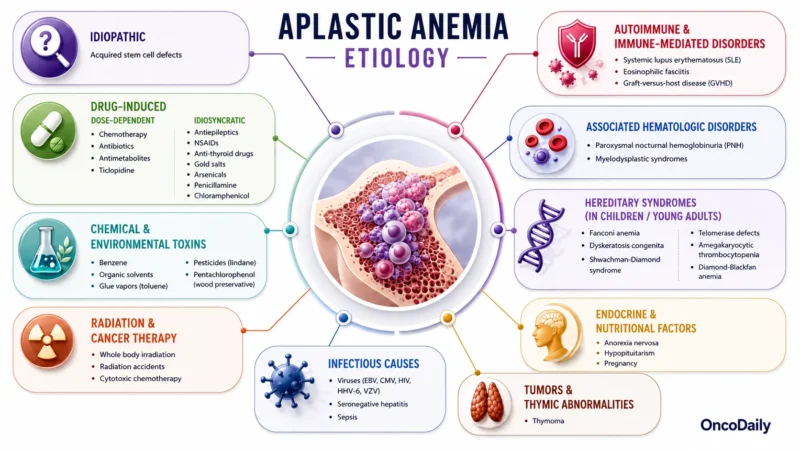
The underlying etiologies of aplastic anemia can be broadly classified into acquired or inherited categories. The most common etiology, idiopathic, accounts for 65%. Fanconi anemia is the most common hereditary cause. Seronegative hepatitis is responsible for 10% of total cases. Telomerase defects are found in 5% -10% of adult-onset. AA is a rare disorder with a bimodal age distribution. The incidence ranges from 0.6-6.1 cases for every million individuals. The ratio of males to females is roughly 1:1.
What Do We Know About Pathogenesis?
Immune suppression is considered a central mechanism in acquired AA. Autoreactive T cells produce cytokines which suppress hematopoiesis and promote apoptosis of HSCs.
Several observations support an immune-mediated failure:
- The clinical efficacy of immunosuppressive therapy
- Associations with specific HLA class I alleles suggest CD8+ T-cell-driven marrow damage.
- Hepatitis-associated AA often develops within months after autoimmune hepatitis and demonstrates a distinct immune profile with T-cell oligoclonality.
- Increased levels of myeloid dendritic cells (mDC1) have been linked to severe disease and may represent future therapeutic targets.
Intrinsic stem cell defects, critically shortened telomeres, inherited and acquired genetic variants may impair stem cell longevity and regenerative capacity.
A variety of environmental and infectious exposures may trigger AA in susceptible individuals. These triggers may directly impair hematopoietic stem cells or trigger secondary marrow suppression. More recently, COVID-19 has also been associated with cases of AA through mechanisms of immune dysregulation.
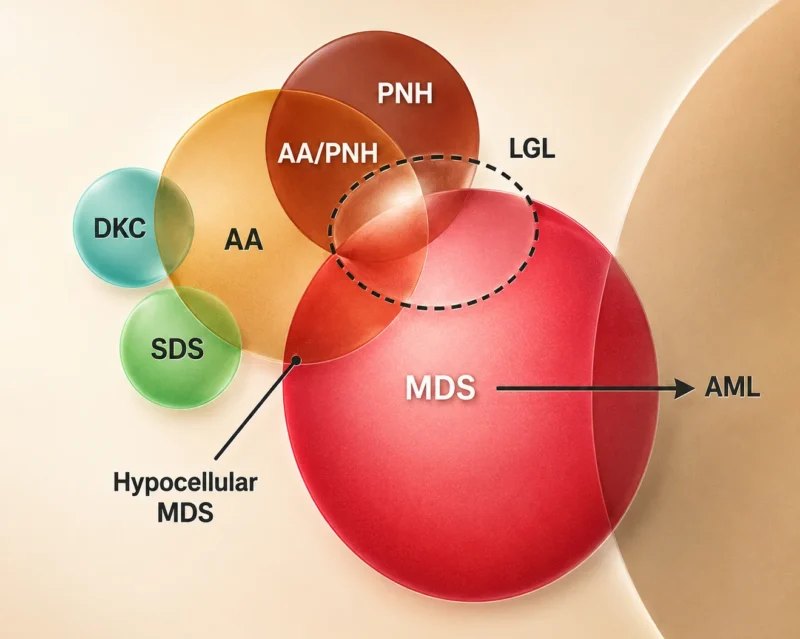
Overlapping Acquired BMFs
AA exists within a broader biologic spectrum that intersects with:
1. hypoplastic myelodysplastic syndrome (hMDS)
2. paroxysmal nocturnal hemoglobinuria (PNH)
3. clonal hematopoiesis of indeterminate potential (CHIP)
4. idiopathic cytopenias of undetermined significance (ICUS)
5. T-cell large granular lymphocytic leukemia (T-LGL leukemia)
6. undiagnosed congenital marrow failure syndromes,
Somatic Mutations and Clonal Architecture
CH is highly prevalent in AA (∼50%) and appears to involve 2 major categories of genetic alterations:
- Mutations and CNAs commonly associated with MDS/AML, including −7, DNMT3A and ASXL1.
- Alterations overrepresented in AA, such as PIGA, BCOR/BCORL1 mutations.
The first group of mutations demonstrates clear age dependency and is dominated by C-to-T transitions, similar to ARCH, suggesting that many are acquired during aging. In some patients, these mutated populations gradually expand over time and may eventually contribute to progression toward MDS/AML, although not all patients ultimately evolve into overt malignancy. In contrast, several mutations commonly observed in ARCH, including TET2, JAK2 and spliceosome mutations, are less frequent in AA.
PNH clones are frequently detected in AA and are thought to escape immune-mediated marrow destruction. Their presence is often associated with better response to immunosuppressive therapy, although large clones may increase the risk of thrombosis or hemolysis.
Following IST, repopulation from a limited pool of residual stem cells may favor populations with proliferative advantages (“bottleneck effect”). In addition, the autoimmune marrow environment may preferentially select mutations such as DNMT3A and ASXL1.
springer.com
Distinguishing hMDS from AA
Typical AA is usually associated with a normal karyotype, whereas cytogenetic abnormalities favor MDS. However, some isolated changes may also occur in benign clonal hematopoiesis and a normal karyotype does not exclude MDS, since only about half of cases demonstrate detectable chromosomal alterations.
Morphologic findings such as dysmegakaryopoiesis, severe dysgranulopoiesis, ring sideroblasts, marrow fibrosis, increased CD34+ cell clusters or elevated blasts strongly support MDS over AA. Both hypocellular may respond to immunosuppressive therapy, supporting partially shared immune-mediated mechanisms. The presence of small PNH clones are relatively common in idiopathic AA.
SNP-array karyotyping and NGS have improved diagnostic discrimination. Secondary MDS evolving from AA, which develops in 10%-20% of patients and is frequently associated with monosomy 7/del(7q), appears to possess a distinct molecular profile, further supporting biological differences between primary hMDS and post-AA clonal evolution.
Inherited marrow failure syndromes may also demonstrate baseline dysplastic changes that raise concern for MDS.
Germline BMF
Recent genomic screening studies have identified germline BMFs in approximately 5%-10% of patients with idiopathic BMF or MDS. Reliance solely on physical stigmata or family history may therefore fail to recognize a subset of patients. MDS or AML may be the feature that first brings the patients to medical attention.
Fanconi Anemia presents with pancytopenia, organ hypoplasia, bone defects and genomic instability. Diagnosis is supported by increased chromosomal breakage after exposure to DNA cross-linking agents.
Dyskeratosis Congenita and related telomere disorders result from defective telomere maintenance, commonly present with marrow failure, nail dystrophy, reticular skin pigmentation and oral leukoplakia.
GATA2 Deficiency combines immunodeficiency, marrow failure, and high risk of MDS/AML. Recurrent infections, warts, lymphedema and monocytopenia are characteristic findings. Germline GATA2 mutations are particularly common (37%) in pediatric MDS.
Shwachman-Diamond Syndrome is associated with pancreatic insufficiency, skeletal abnormalities, cytopenias and increased risk of myeloid neoplasia. Low pancreatic enzyme levels may support the diagnosis.
Diamond-Blackfan Anemia presents with erythroid failure, usually during infancy and is often accompanied by craniofacial or cardiac abnormalities. Elevated erythrocyte adenosine deaminase activity is a typical laboratory finding.
Congenital Amegakaryocytic Thrombocytopenia presents with severe early thrombocytopenia and may later evolve into AA. Although occasional cases of MDS/AML have been reported, its leukemic predisposition remains less clear.
Diagnosing these disorders is important not only for surveillance and treatment decisions, but also for transplantation planning, particularly donor selection and avoidance of affected family donors.
What Else Can Mimic AA?
The differential diagnosis of pancytopenia in AA includes both acquired and inherited causes:
- Myelophthisic syndromes caused by metastatic tumors, fibrosis and hematologic malignancies which typically demonstrate marrow infiltration rather than hypocellularity
- Isolated lineage failures such as pure red cell aplasia or agranulocytosis
- Transient marrow suppression secondary to chemotherapy, radiation, sepsis or viral infections
- Megaloblastic anemia, often associated with macroovalocytes and hypersegmented neutrophils
- Hypersplenism, commonly accompanied by splenomegaly
Diagnostic Workup
The diagnostic evaluation of AA focuses on confirming marrow failure, assessing the severity of cytopenias and excluding alternative causes.
CBC with differential typically demonstrates pancytopenia, while the reticulocyte count is markedly reduced, reflecting impaired erythropoiesis. Peripheral blood smear findings are usually nonspecific but help exclude dysplastic or malignant processes.
Telomere length analysis and chromosome breakage testing may be performed for inherited marrow failure syndromes, particularly in younger patients. Flow cytometry is important for identifying PNH clones and distinguishing AA from hematologic malignancies.
Bone marrow biopsy classically reveals severe hypocellularity with fatty replacement and absence of fibrosis or malignant infiltration. Aspirates may result in a “dry tap”.
FISH or SNP-array analysis can be helpful when cellular material is limited. NGS panels may detect somatic mutations which have diagnostic and prognostic implications.
Diagnostic Criteria for AA generally require:
- Bone marrow hypoplasia/aplasia without evidence of infiltrative disease
- Presence of at least two of the following: Reticulocytes <40,000/µL (or <1%), ANC <500/µL, Platelets <20,000/µL
Management of AA in Adults
Management is guided by disease severity, patient age, performance status, donor availability and the presence of reversible causes.
Severe and Very Severe AA
In medically fit younger patients with a matched related donor, allogeneic HSCT remains the preferred first-line approach. In the absence of a suitable donor, immunosuppressive therapy (IST) with horse antithymocyte globulin, cyclosporine, and eltrombopag remains the standard treatment.
Patients receiving IST require close monitoring and surveillance for evolution toward MDS/AML In older or medically frail patients, treatment emphasizes tolerability and supportive care. Febrile neutropenia constitutes a medical emergency.
Moderate AA
Some patients remain stable with observation alone, whereas others require lower-intensity IST. HSCT is rarely used as first-line therapy in moderate disease but may be considered in younger patients with progressive transfusion dependence or recurrent infections.
A complete response includes transfusion independence, hemoglobin levels above 10 g/dL, and platelet counts exceeding 50,000/µL.
Refractory and Relapsed Settings
Relapse occurs in up to one-third of patients with SAA. A second course of IST can be effective in 55% to 60% of patients, but salvage HSCT may be a more preferable option for relapses or refractory cases.
In a retrospective cohort study conducted by Beatrice Drexler et al., 302 patients were analyzed to assess long-term outcomes after first-line IST or HSCT. IST was used in 75% of patients, while 25% underwent upfront HCT. IST was associated with a cumulative relapse incidence of 24% at 25 years from diagnosis.
IST was associated with higher rates of iron overload (18.1% vs. 3.9%) and cardiovascular events (10.6% vs. 1.3%), while HCT carried risks of graft failure and GVHD. Secondary malignancies occurred at similar rates. Overall, the study highlights the trade-off between the early toxicity but curative potential of transplantation and the chronic relapse- and complication-prone course following IST.
You can also read: Clones and Clonality: Darwinian Natural Selection at the Cellular Level
Written by Susanna Mikayelyan, MD
FAQ
Can aplastic anemia turn into leukemia?
Yes. Although AA is initially a nonmalignant marrow failure disorder, a subset of patients may later develop myelodysplastic neoplasms (MDS) or acute myeloid leukemia (AML), particularly after long-standing disease or clonal evolution involving abnormalities such as monosomy 7.
Is aplastic anemia considered an autoimmune disease?
In many acquired cases, yes. Autoreactive T cells suppress hematopoietic stem cells and contribute to marrow failure, which explains why immunosuppressive therapies can be highly effective in selected patients.
What makes hypocellular MDS difficult to distinguish from aplastic anemia?
Both disorders may present with pancytopenia and hypocellular marrow. Features such as dysplasia, increased blasts, ring sideroblasts, cytogenetic abnormalities, or somatic mutations generally favor MDS, although overlap between the two entities is increasingly recognized.
Can COVID-19 trigger aplastic anemia?
Rare cases of AA have been reported after COVID-19 infection, likely related to immune dysregulation and abnormal immune activation. However, this remains uncommon and the exact mechanisms are still being investigated.
Are somatic mutations always a sign of leukemia in aplastic anemia?
Not necessarily. Clonal hematopoiesis is common in AA and some mutations may remain stable for years without progression to overt malignancy. Their clinical significance depends on the specific mutation, clone size, and evolutionary dynamics over time.
Can aplastic anemia remain stable without treatment?
Yes. Some patients, particularly those with moderate AA, may remain clinically stable for years with careful monitoring alone. Others gradually progress, develop transfusion dependence, or evolve clonally over time, making long-term surveillance essential even in seemingly indolent disease.
Could future therapies for aplastic anemia become more targeted?
Potentially. Increasing understanding of immune signaling, clonal selection, telomere biology, and marrow microenvironment interactions may eventually allow more personalized approaches beyond broad immunosuppression alone.