The treatment paradigm for advanced non-squamous non-small cell lung cancer (nsqNSCLC) has undergone a profound transformation with the integration of immune checkpoint inhibitors into first-line therapy. Among these, Toripalimab, an anti–PD-1 monoclonal antibody, has demonstrated clinical efficacy in combination with platinum-based chemotherapy and is approved in China for this indication.
Despite these advances, practical challenges remain. Intravenous (IV) administration requires repeated hospital visits, infusion time, and healthcare resource allocation. As immunotherapy is increasingly used in long-term treatment settings, there is a growing need to optimize not only outcomes but also treatment delivery.
At the ELCC 2026, Lin Wu presented results from a randomized phase III trial evaluating whether a subcutaneous (SC) formulation of toripalimab could provide comparable pharmacokinetics, efficacy, and safety to IV administration in patients with advanced nsqNSCLC.

Background
The success of PD-1/PD-L1 inhibitors in NSCLC has been well established, particularly in combination with chemotherapy (Reck et al., 2016; Gandhi et al., 2018). However, as treatment duration extends and patient volumes increase, the logistical burden of IV therapy becomes increasingly relevant.
Subcutaneous administration offers several theoretical advantages. It simplifies drug delivery, reduces chair time in infusion units, and may improve patient comfort and adherence. In other oncology settings, SC formulations of biologics have already demonstrated feasibility and patient preference.
This study was designed to determine whether these practical benefits could be achieved without compromising pharmacologic exposure or clinical outcomes.

Methods
This randomized, open-label, multicenter phase III trial (NCT06505837) enrolled patients with histologically or cytologically confirmed recurrent or metastatic nsqNSCLC. Eligible patients had no EGFR-sensitive mutations or ALK rearrangements, reflecting a population appropriate for immunotherapy-based treatment.
A total of 396 patients from 72 centers across China were randomized in a 1:1 ratio to receive:
- SC toripalimab (360 mg) every 3 weeks
- IV toripalimab (240 mg) every 3 weeks
Both groups received combination chemotherapy consisting of pemetrexed plus carboplatin or cisplatin for up to four cycles. Patients without disease progression transitioned to maintenance therapy with toripalimab plus pemetrexed.
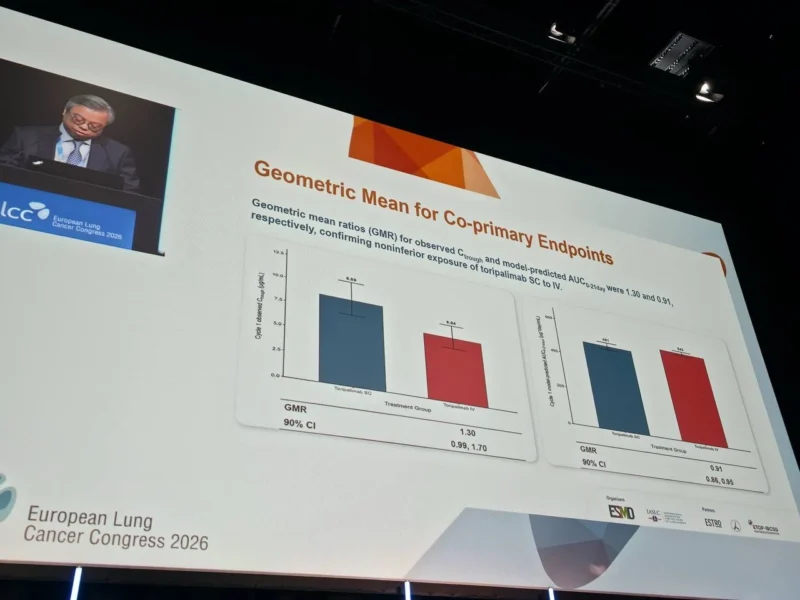
The trial incorporated co-primary pharmacokinetic endpoints, including trough serum concentration (Ctrough) and area under the curve from 0 to 21 days (AUC₀–21), assessed during the first treatment cycle. These endpoints were critical to establishing noninferiority of systemic exposure between SC and IV formulations.
Secondary endpoints included objective response rate (ORR), progression-free survival (PFS), and safety.
Results
The study met its primary pharmacokinetic objectives. The SC formulation demonstrated noninferior exposure compared with IV toripalimab:
- Ctrough geometric mean ratio: 1.31 (90% CI, 1.00–1.72)
- AUC₀–21 ratio: 0.90 (90% CI, 0.86–0.95)
These findings confirm that subcutaneous administration achieves comparable systemic drug levels, a prerequisite for equivalent clinical efficacy.
Efficacy
Clinical outcomes were consistent across both treatment arms:
- Objective Response Rate (ORR):
- 57.6% in the SC arm vs 49.5% in the IV arm
- Median Progression-Free Survival (PFS):
- 8.1 months in both arms
With a median follow-up of 7.1 months, there was no evidence of reduced efficacy with SC administration. The comparable PFS across both groups further supports the therapeutic equivalence of the two formulations.

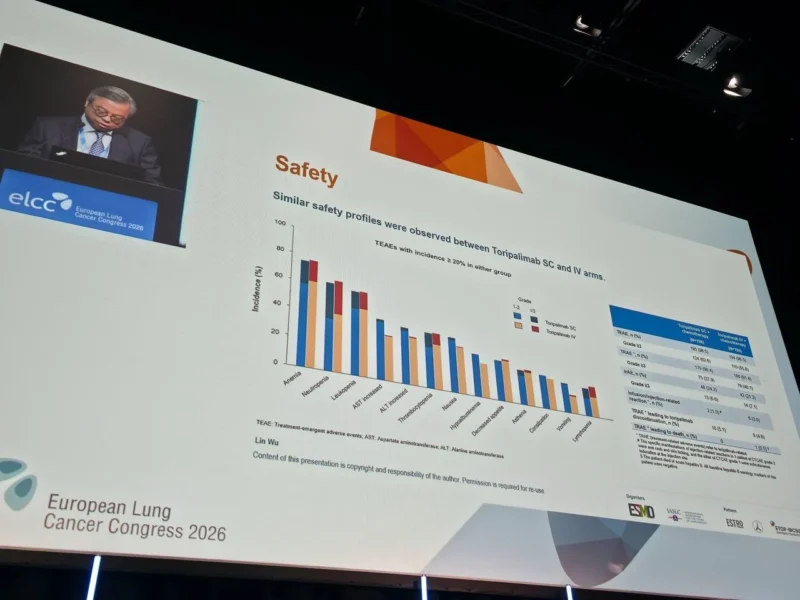
Safety
The safety profiles of SC and IV toripalimab were similar, with no new or unexpected adverse events reported. Toxicities were consistent with known effects of PD-1 inhibitors combined with chemotherapy, including hematologic and immune-related events.
Importantly, no additional safety concerns were identified with subcutaneous delivery, reinforcing its feasibility in clinical practice.
Clinical Implications
The results of this trial extend beyond demonstrating noninferiority. They highlight a broader shift in oncology toward optimizing treatment delivery alongside efficacy.
Subcutaneous toripalimab offers several potential advantages:
It reduces the need for prolonged infusion visits, which can be burdensome for patients undergoing repeated treatment cycles.
It may improve workflow efficiency in oncology centers, particularly in high-volume settings.
It aligns with patient-centered care by offering a more convenient mode of administration.
As healthcare systems increasingly face resource constraints, such innovations could play a critical role in maintaining access to effective therapies.
Positioning Within Current Treatment Landscape
The current standard of care for advanced nsqNSCLC includes PD-1/PD-L1 inhibitors combined with chemotherapy. While efficacy has improved significantly over the past decade, attention is now turning toward optimizing how these therapies are delivered.
This study suggests that SC toripalimab could become a practical alternative to IV administration without compromising clinical outcomes. Similar strategies are being explored with other monoclonal antibodies across oncology, reflecting a broader trend toward more flexible treatment models.
Limitations
Although the findings are promising, several considerations should be noted. The median follow-up of 7.1 months is relatively short, and longer-term outcomes, including overall survival, remain to be determined. Additionally, the study population was limited to China, and broader validation in diverse populations may be warranted.
Conclusion
The phase III trial presented at ELCC 2026 demonstrates that subcutaneous toripalimab provides comparable pharmacokinetics, efficacy, and safety to intravenous administration in first-line treatment of advanced nsqNSCLC.
These findings support the potential integration of SC toripalimab into clinical practice as a more convenient and resource-efficient alternative to IV therapy. As oncology continues to evolve, innovations in treatment delivery will play an increasingly important role in improving both patient experience and healthcare sustainability.
You can read full abstract here


