The management of metastatic EGFR-mutant non-small cell lung cancer (NSCLC) has been transformed by the introduction of third-generation EGFR tyrosine kinase inhibitors, particularly osimertinib. While systemic therapy achieves meaningful disease control, most patients eventually experience progression, often driven by residual disease sites. This has led to growing interest in integrating local treatment strategies to improve long-term outcomes.
NorthStar trial local consolidative therapy in EGFR-mutant NSCLC represents one such approach, exploring whether targeting residual disease with radiotherapy or surgery can enhance the efficacy of systemic treatment.
At the European Lung Cancer Congress 2026, a secondary analysis of the phase II randomized NorthStar trial provided deeper insights into which patients derive the greatest benefit from local consolidative therapy (LCT) and how patterns of failure evolve after treatment.

Study Design and Treatment Approach
The NorthStar trial previously demonstrated that adding LCT to osimertinib significantly improved progression-free survival compared with osimertinib alone. This secondary analysis focused on refining patient selection by evaluating predictors of benefit and patterns of recurrence.
A total of 119 patients who had not progressed after 6–12 weeks of induction osimertinib were included in this analysis. Patients were randomized to continue osimertinib alone or to receive osimertinib plus LCT.
Local consolidative therapy included a range of approaches, with the majority of patients receiving radiotherapy alone, while others underwent surgery or a combination of both. Importantly, most patients continued osimertinib during radiotherapy, reflecting a modern multimodality treatment strategy.

Key Efficacy Findings
The results reinforce the role of NorthStar trial local consolidative therapy in EGFR-mutant NSCLC as a strategy to prolong disease control, particularly in selected patients.
Patients who received LCT in addition to osimertinib experienced a significant improvement in progression-free survival compared with those receiving osimertinib alone. The magnitude of benefit, however, varied substantially depending on baseline disease characteristics after induction therapy.
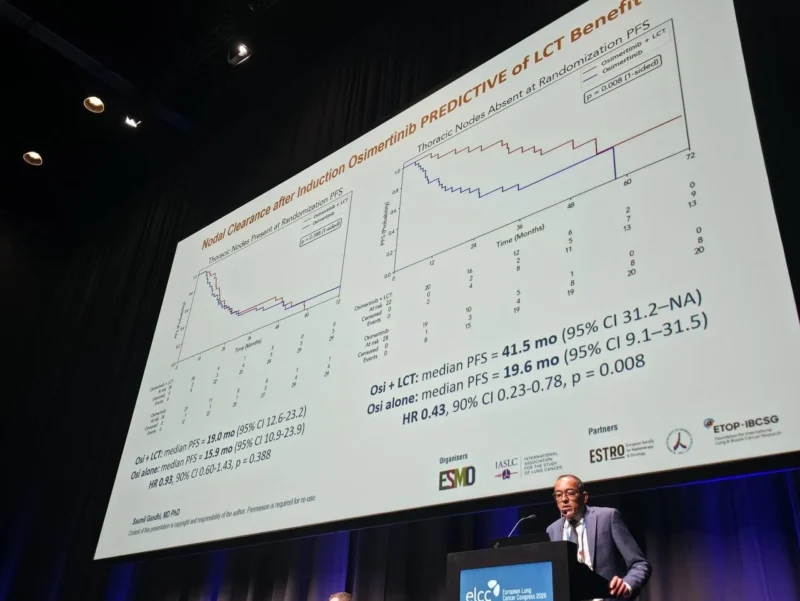
The most striking findings were observed in patients who achieved clearance of thoracic nodal disease following initial osimertinib treatment. In this subgroup, median progression-free survival reached 41.5 months with the addition of LCT, compared with 19.6 months in patients treated with osimertinib alone. This represents a clinically meaningful improvement, highlighting the importance of disease response prior to local intervention.
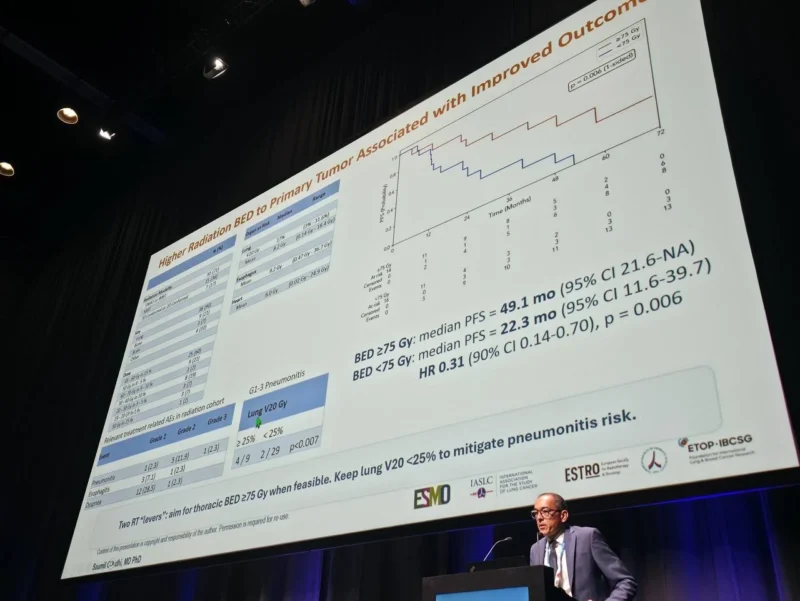
Similarly, patients without persistent pleural effusion after induction therapy also appeared to benefit from LCT, with longer progression-free survival compared with systemic therapy alone. Maintaining lung V20 <25% was associated with reduced toxicity.
In contrast, patients with residual thoracic nodal disease or persistent pleural effusion did not derive a clear benefit from the addition of local therapy. These findings suggest that treatment response to initial systemic therapy may serve as a key biomarker for selecting patients most likely to benefit from LCT. Findings reflect a real-world, unselected patient population.
Importantly, higher radiation dose (BED ≥75 Gy) was associated with significantly improved outcomes, highlighting that not only patient selection, but also radiation quality may influence the benefit of LCT.
Patterns of Disease Progression
Understanding how and where disease recurs after treatment is critical for optimizing future strategies. This analysis provides important insights into patterns of failure following NorthStar trial local consolidative therapy in EGFR-mutant NSCLC.
Among patients who experienced recurrence after receiving LCT, the majority of failures were distant rather than local. Only a small proportion of recurrences were confined to the original treatment field, while most occurred at new metastatic sites outside the radiotherapy field.
Specifically, among patients treated with radiotherapy, only 8% of recurrences were in-field, whereas 92% occurred outside the irradiated areas. This suggests that while LCT effectively controls local disease, systemic progression remains the dominant challenge in metastatic EGFR-mutant NSCLC.
These findings emphasize the need for continued optimization of systemic therapy, even when aggressive local approaches are used.
Clinical Implications
This secondary analysis highlights several key considerations for clinical practice.
First, patient selection is critical. Not all patients benefit equally from local consolidative therapy, and careful assessment of disease response after induction osimertinib is essential.
Second, the findings support a multimodal treatment paradigm, where systemic therapy is complemented by local interventions in appropriately selected patients.
Third, the predominance of distant progression underscores the importance of integrating effective systemic strategies alongside local therapy to achieve durable disease control.

Key Takeaway
The secondary analysis of the NorthStar trial demonstrates that NorthStar trial local consolidative therapy in EGFR-mutant NSCLC significantly improves progression-free survival when added to osimertinib, particularly in patients with favorable response to induction therapy.
Clearance of thoracic nodal disease and absence of pleural effusion emerge as key predictors of benefit, while patterns of failure highlight the ongoing challenge of distant disease progression.
These findings provide an important step toward personalized treatment strategies in metastatic EGFR-mutant NSCLC, refining the role of local therapy in the era of targeted treatment.
You can read full abstract here


