Van Morris, Section Chief of Colorectal Cancer and Associate Professor at MD Anderson Cancer Center, shared a post on X about a paper he co-authored with colleagues published in Cancer Cell:
“Very proud of our MD Anderson Cancer Center team’s work now online Cancer Cell for BRAF+EGFR+PD1 blockade in MSS, BRAF V600E mCRC.
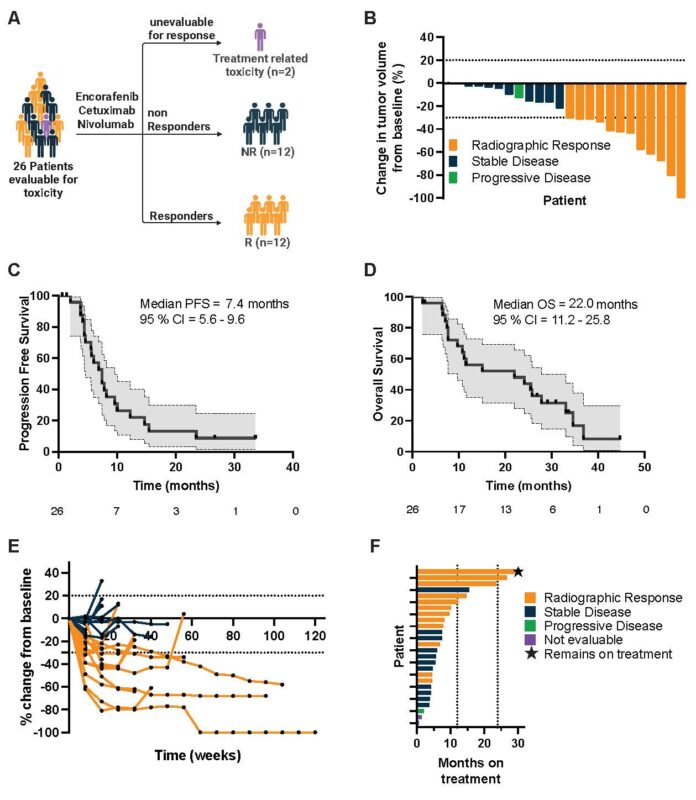
ORR 50%, mPFS >7 mos with encorafenib plus cetuximab plus nivolumab
Thread to expand IO benefit+liquid biopsy utility in high-risk MSS CRC patients
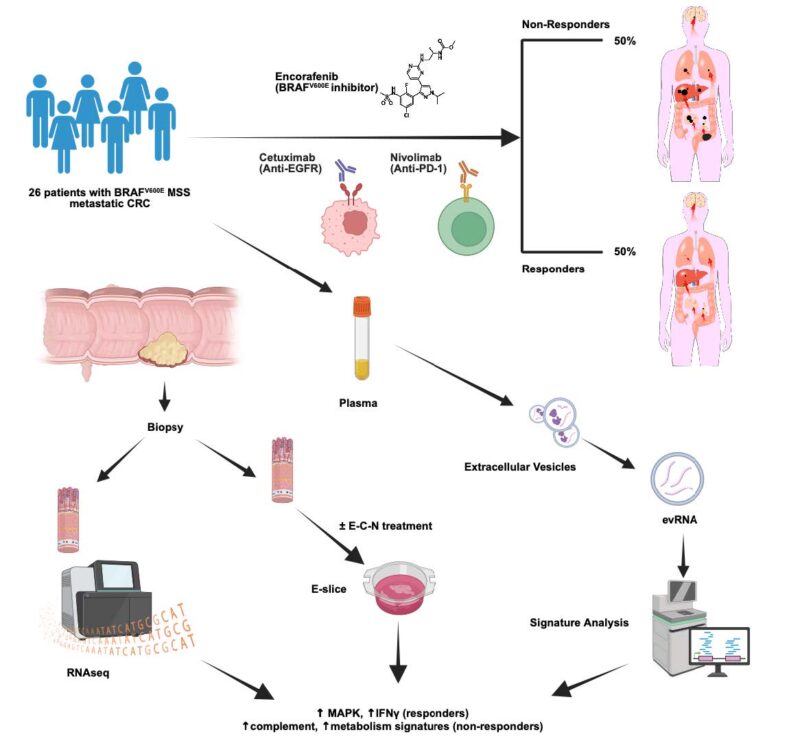
Durable response > 2 years in MULTIPLE patients treated with E+C+N, and triple combo was well tolerated for this 2nd/3rd line MSS BRAF V600E population.
Prior precedent with E+C (no nivo) in this setting showed ORR 20% and mPFS ~4 mos. Congratulations to Christine Parseghian for writing this protocol.
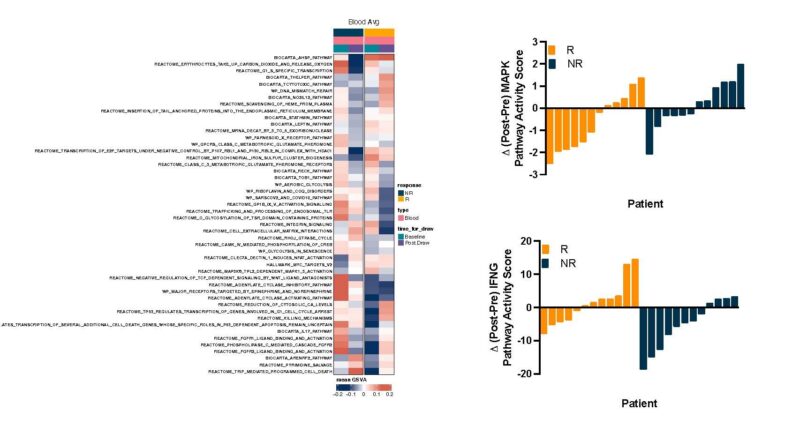
Clinical utility of liquid biopsy beyond ctDNA was shown! Thanks to Anirban Maitra and team, we saw concordance in baseline RNA signatures from tumor tissue AND plasma evRNA alike in association with treatment response:
Resp – high immune activation signatures
Non-resp – high metabolism signatures
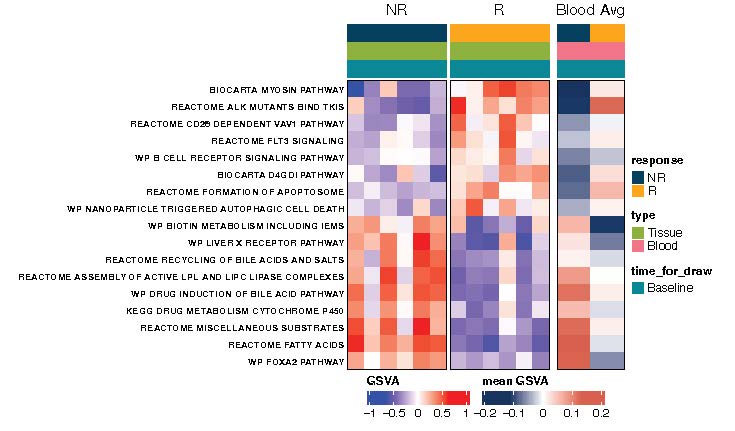
Interestingly, high complement activity uniquely characterized non-response to E+C+N – the sole immune signature in RNA a/w no treatment benefit!
Our talented MD Anderson Cancer Center heme/onc fellow Mir Lim is further studying the role of complement activation in MSS CRC.
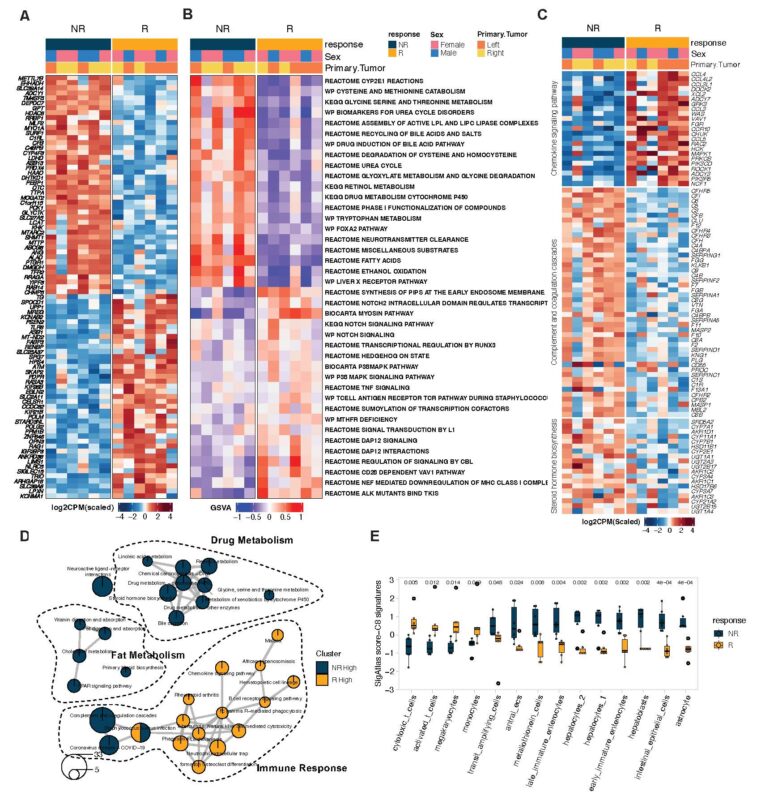
Dynamic evRNA changes linked E+C+N response to high IFNg signatures in paired blood samples (thanks Kyuson Yun and team)
Serial plasma collection on future trials for evRNA analyses is feasible, increases sample size, and improves translational interpretations = better patient care.
Addition of PD1 blockade to BRAF+EGFR inhibition with E+C+N offers clear, extended benefit for patients with MSS, BRAF V600E mCRC.
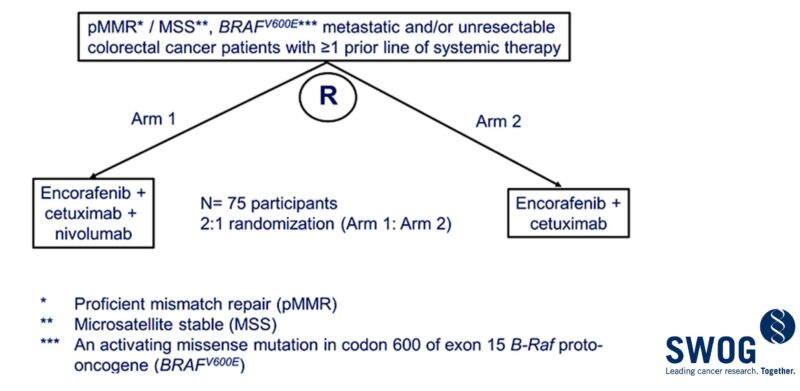
The follow-up SWOG S2107 study comparing E+C with or without N in this same population has completed enrollment – results awaited.
Patients with BRAF V600E CRC need better therapies – targeting the immune activation signature for this CRC subpopulation is critical.
Grateful to be on a highly collaborative + passionate + fun CRC team at MD Anderson Cancer Center alongside the incredible Scott Kopetz to end cancer for CRC patients.”

Title: Phase 1/2 trial of encorafenib, cetuximab, and nivolumab in microsatellite stable BRAFV600E metastatic colorectal cancer
Authors: Van K. Morris, Christine M. Parseghian, Vahid Bahrambeigi, Nourhan Abdelfattah, Lianchun Xiao, Anjali Agrawal, Kangyu Lin, Kanwal P.S. Raghav, Robert A. Wolff, Arvind Dasari, Ryan W. Huey, Bryan K. Kee, Michael J. Overman, Jason A. Willis, Phat H. Le, Michelle Escano, Yunyu C. Baig, Kelsey Pan, David Menter, Alda L. Tam, Wai C. Foo, Li Shen, Hey Min Lee, Thomas D. Gallup, Cori Margain, Dave Gallup, Kimal I. Rajapakshe, Paola A. Guerrero, Jing Wang, Ryan B. Corcoran, Anirban Maitra, Kyuson Yun, Scott Kopetz
You can read the Full Article on Cancer Cell.

More posts featuring Van Morris.


