For years, KRAS G12D has stood among the most consequential and frustrating targets in solid tumor oncology. It is biologically important, clinically common, and therapeutically elusive. In non-small-cell lung cancer (NSCLC), the KRAS p.G12D variant occurs in about 5% of patients, while in pancreatic ductal adenocarcinoma (PDAC) it is the dominant KRAS substitution, occurring in roughly 40% of cases. Despite this prevalence, no KRAS G12D-directed therapy has yet entered routine clinical use. That gap has defined a major unmet need in precision medicine, especially as other KRAS subtypes, most notably KRAS G12C, have already produced approved targeted agents (Park et al., 2026).
Now, first-in-human data published in the New England Journal of Medicine suggest that this landscape may be beginning to change. In a phase 1 study, setidegrasib (ASP3082), a first-in-class KRAS G12D-targeted protein degrader, demonstrated encouraging clinical activity in patients with previously treated advanced NSCLC and pancreatic cancer, together with pharmacodynamic evidence of target degradation and a generally manageable safety profile (Park et al., 2026).
These results are early, and they do not establish a new standard of care. But they do something highly important: they provide one of the clearest demonstrations so far that KRAS G12D can be therapeutically attacked in patients, not merely in models or hypotheses. That makes this study more than another phase 1 signal. It marks a possible shift in what is considered druggable in solid tumors.
Why KRAS G12D Has Been So Difficult to Target
The difficulty of KRAS G12D is partly structural. The success of KRAS G12C inhibitors relied on a specific chemical opportunity: the mutant cysteine residue enabled covalent binding. KRAS G12D lacks that same reactivity and offers only a shallow switch II pocket, making conventional small-molecule inhibition far more difficult. As a result, while KRAS became one of the flagship success stories of modern targeted therapy in one mutation subtype, KRAS G12D remained outside the reach of approved drugs (Park et al., 2026).
That is why setidegrasib is especially interesting. It does not work as a classic inhibitor. Instead, it is a protein degrader, designed to selectively eliminate mutant KRAS G12D. Mechanistically, setidegrasib forms a ternary complex among KRAS G12D, a proteolysis-targeting chimera scaffold, and the von Hippel–Lindau E3 ligase, leading to selective degradation of the mutant protein and suppression of downstream signaling (Park et al., 2026). In practical terms, this means it is not merely trying to block KRAS G12D activity transiently. It is trying to remove the target altogether.
That conceptual shift matters. Protein degradation may create different pharmacologic behavior, different pathway suppression dynamics, and potentially different resistance patterns from standard enzymatic inhibition. In KRAS G12D-driven tumors, where therapeutic options have remained sparse, that distinction could prove clinically meaningful.
Copyright © 2026 Massachusetts Medical Society.
Study Design: A First-in-Human Look at a New Therapeutic Class
The study reported by Park and colleagues was a phase 1, open-label, international, multicenter trial evaluating setidegrasib in patients with previously treated advanced solid tumors harboring KRAS p.G12D variants. The trial included both dose-escalation and dose-expansion phases. Setidegrasib was administered intravenously once weekly at doses ranging from 10 mg to 800 mg (Park et al., 2026).
Eligible patients had locally advanced unresectable or metastatic solid tumors with documented KRAS G12D alterations, measurable disease according to RECIST 1.1, and prior exposure to at least one systemic anticancer therapy. Patients with NSCLC and pancreatic cancer made up the majority of the study population, reflecting the biologic importance of KRAS G12D in those diseases.
The trial’s primary objectives were to define the safety profile, assess dose-limiting toxicities, and determine the recommended phase 2 dose. Secondary and exploratory endpoints included objective response, duration of response, progression-free survival, overall survival, pharmacokinetics, and pharmacodynamic assessment of KRAS G12D degradation in tumor tissue and circulating tumor DNA.
Overall, 203 patients were enrolled:
- 59 with NSCLC
- 124 with pancreatic ductal adenocarcinoma
- 20 with other solid tumors
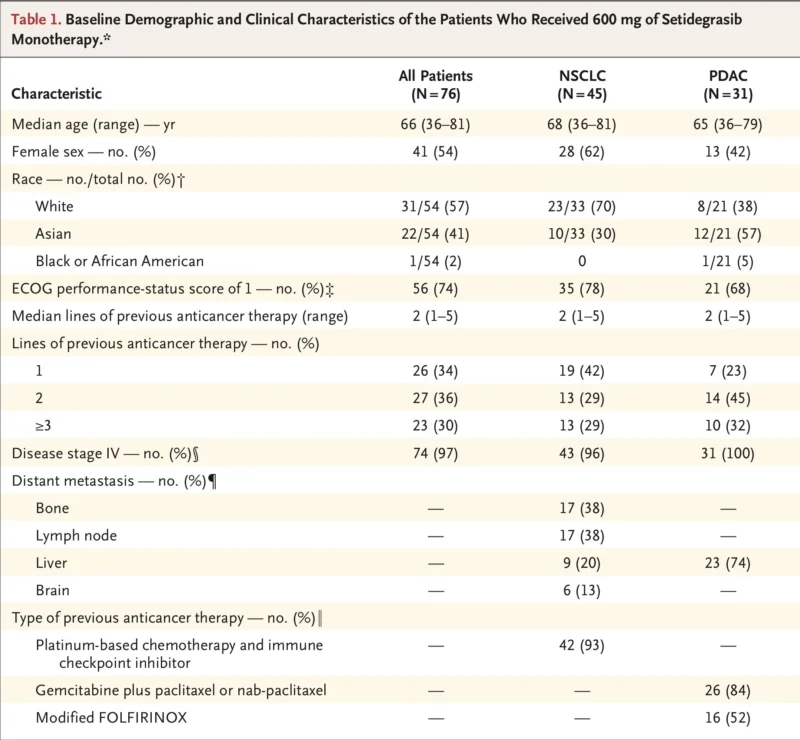
Of these, 76 patients ultimately received the dose selected as the recommended phase 2 dose: 600 mg intravenously once weekly. This included 45 patients with NSCLC and 31 with pancreatic ductal adenocarcinoma.
Why 600 mg Was Chosen
The choice of the 600-mg dose was based on a combination of safety, pharmacokinetics, pharmacodynamics, and early efficacy. The maximum tolerated dose was not reached. However, the 800-mg dose was not considered clinically practical because of low solubility, which required a larger volume and longer infusion time. The 600-mg dose provided a favorable balance of drug exposure, target degradation, and antitumor activity while remaining feasible for clinical development (Park et al., 2026).
This is an important point because phase 1 studies often focus too narrowly on toxicity. In this case, the decision-making also incorporated biological evidence that the drug was doing what it was designed to do, which strengthens confidence in the selected dose.
Safety: Frequent Infusion Reactions, But a Manageable Profile Overall
Among the 76 patients treated at 600 mg, all patients experienced treatment-emergent adverse events, and 42% had adverse events of grade 3 or higher. However, the more clinically relevant figure is that only 9% had treatment-related adverse events of grade 3 or higher, and adverse events led to discontinuation in only 2 patients overall (Park et al., 2026).
The most common treatment-related toxicities were:
- Infusion-related reactions: 80%
- Nausea: 30% (Park et al., 2026)
The infusion-related reactions deserve special attention because they were common, but their pattern was reassuring. They were all grade 1 or 2, occurred predominantly during the first infusion, and became less frequent after subsequent doses. The most commonly reported manifestations included pruritus, rash, and urticaria. These events often led to temporary treatment interruption and supportive measures such as antihistamines, but none led to permanent treatment discontinuation (Park et al., 2026).
This distinction is important. A headline number of 80% infusion-related reactions sounds alarming at first glance. But the actual clinical implications were far more manageable than the number alone suggests. They were mostly early, transient, low-grade, and operationally manageable.
Serious treatment-related adverse events occurred in 4 patients (5%), and treatment-related adverse events leading to dose reduction also occurred in 4 patients (5%). No treatment-related adverse events led to death, and none led to discontinuation at the recommended phase 2 dose.
From a development standpoint, this safety profile is notable because it may support future combination strategies. The authors specifically point out that the absence of severe gastrointestinal or dermatologic toxicity at the 600-mg dose, together with low discontinuation rates, may facilitate use in rational combinations.
Copyright © 2026 Massachusetts Medical Society.
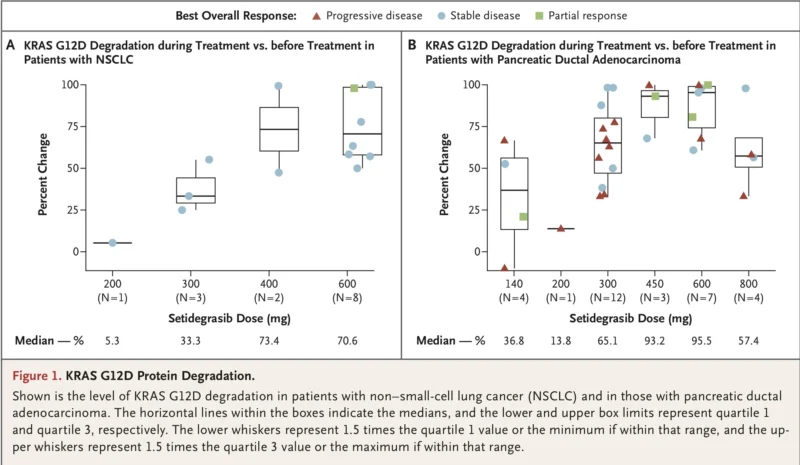
Pharmacodynamics: Strong Evidence of On-Target KRAS G12D Degradation
One of the strongest aspects of the paper is that it does not rely solely on radiographic outcomes. It also demonstrates that setidegrasib is biologically active in patients.
In paired tumor biopsies, the median reduction in KRAS G12D protein levels at the 600-mg dose was:
- 70.6% in NSCLC
- 95.5% in pancreatic ductal adenocarcinoma
That degree of degradation is striking, especially in pancreatic cancer. It confirms that the drug is hitting the intended target in human tumors, not just in vitro systems.
Circulating tumor DNA findings were also informative. In patients with stable disease or partial response, the median greatest reduction in KRAS p.G12D variant allele frequency exceeded 90% in both NSCLC and pancreatic cancer. Clearance of detectable mutant allele occurred in multiple responders, and reductions greater than 50% were common in patients who derived clinical benefit.
These molecular findings add real weight to the clinical efficacy data. They suggest that the tumor responses were not incidental. They were accompanied by measurable suppression of the driver alteration itself.
Clinical Activity in NSCLC: One of the Most Encouraging Signals in the Study
The clearest efficacy signal in the trial emerged in advanced KRAS G12D-mutant NSCLC.
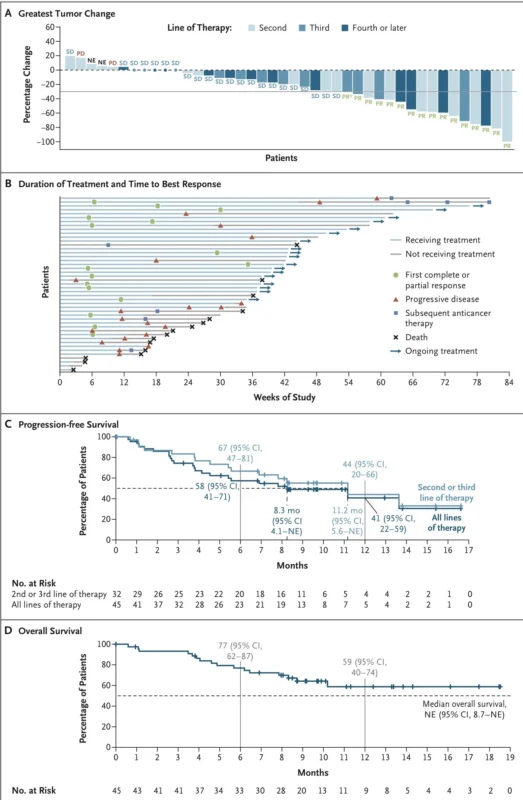
Among the 45 patients with NSCLC treated with 600 mg as second-line therapy or later:
- Objective response rate (ORR): 36% (95% CI, 22 to 51)
- Median progression-free survival (PFS): 8.3 months
- Estimated 12-month overall survival (OS): 59%
Of the 16 patients counted in the response rate, 15 had confirmed partial responses and 1 had an unconfirmed partial response pending confirmation. The median time to response among confirmed responders was 1.4 months, suggesting that clinical activity can emerge relatively early in treatment.
The outcomes were even more interesting in the subgroup treated in the second- or third-line setting, which may be the most clinically relevant near-term population. Among 32 patients in that category:
- ORR: 38%
- Median PFS: 11.2 months
Those numbers are difficult to ignore. In previously treated metastatic NSCLC, especially after platinum-based chemotherapy and immunotherapy, the benchmark remains modest. Standard later-line therapy such as docetaxel, with or without ramucirumab, is typically associated with response rates in the teens and median progression-free survival of roughly 3 to 4.5 months. Cross-trial comparisons should always be cautious, but a 36% response rate and an 8.3-month median PFS, rising to 11.2 months in second- and third-line use, compare favorably with what clinicians are accustomed to seeing in this setting (Park et al., 2026).
This does not mean setidegrasib is ready for routine use. But it does mean the efficacy signal in KRAS G12D-mutant NSCLC looks real, clinically relevant, and worthy of rapid follow-up.
Copyright © 2026 Massachusetts Medical Society.
Clinical Activity in Pancreatic Cancer: More Modest, Still Meaningful
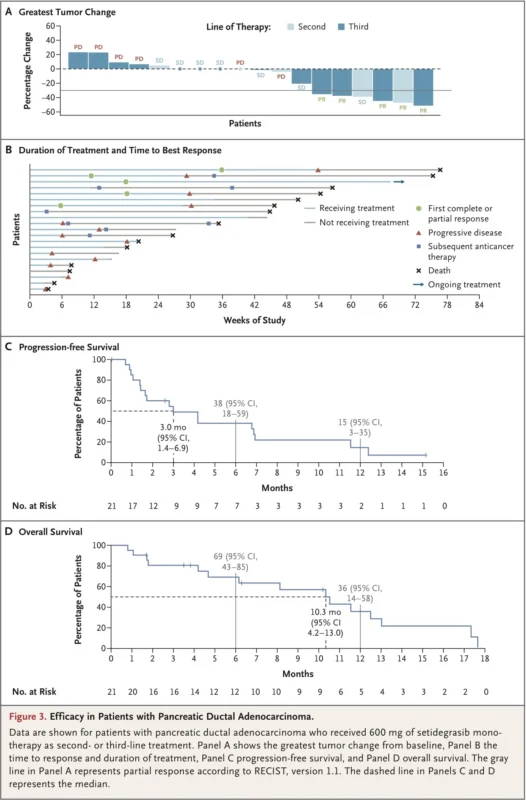
The pancreatic cancer results were less dramatic, but still important. Among 21 patients with metastatic pancreatic ductal adenocarcinoma treated with 600 mg in the second- or third-line setting:
- ORR: 24% (95% CI, 8 to 47)
- Median PFS: 3.0 months
- Median OS: 10.3 months
In the broader group of all 31 pancreatic cancer patients treated at 600 mg, the response rate was 19% (Park et al., 2026).
These data require careful interpretation. Pancreatic cancer remains one of the most treatment-resistant tumor types in oncology, and outcomes in the second- and third-line setting are generally poor. Against that backdrop, a 24% response rate is noteworthy. At the same time, the median progression-free survival of 3.0 months is more modest than what was observed in NSCLC, and the small sample size makes the results especially preliminary.
The paper also notes that patient selection issues may have influenced outcomes, including baseline albumin level and frailty, which became relevant enough during the study to prompt amended eligibility criteria excluding certain pancreatic cancer patients with marked hypoalbuminemia (Park et al., 2026). That context matters because it suggests the pancreatic results may not yet fully define the drug’s potential in that disease.
What Makes the NSCLC Data Stand Out
Among all the efficacy findings in this study, the NSCLC signal is the most compelling. That is partly because the absolute numbers are stronger, but also because the clinical benchmark is better defined. In KRAS G12D-mutant NSCLC, there is no approved matched targeted therapy. Patients generally receive standard chemotherapy and immunotherapy sequences, with limited options once those fail. A targeted protein degrader producing 36% confirmed responses and a median PFS of 8.3 months, with even better outcomes in earlier lines of salvage therapy, represents a meaningful step forward (Park et al., 2026).
The estimated 12-month overall survival of 59% also deserves notice. In a phase 1 study, survival endpoints are exploratory and immature, but that level of 1-year survival in a pretreated molecularly defined subgroup reinforces the impression that the radiographic activity may translate into genuine clinical benefit.
How Does This Compare With Other KRAS G12D Programs?
Although cross-trial comparisons are inherently limited, the paper places setidegrasib in the context of other emerging KRAS G12D programs. Early-phase KRAS G12D inhibitors have reported objective responses in the range of approximately 30% to 60% in NSCLC and 20% to 40% in pancreatic cancer, with varying survival maturity. Setidegrasib’s activity therefore appears broadly competitive in early development, particularly in NSCLC (Park et al., 2026).
What differentiates setidegrasib is not necessarily that it is already proven superior. It is that it uses a different therapeutic mechanism. Rather than conventional inhibition, it relies on targeted protein degradation. The possibility that degradation may alter both the depth and durability of pathway suppression, and perhaps even resistance biology, makes this more than another me-too agent in development.
Copyright © 2026 Massachusetts Medical Society.
Limitations: Why These Data Are Exciting but Not Definitive
The authors are appropriately careful about the limitations of the study. This was a first-in-human phase 1 trial, so the numbers are still small, the follow-up remains limited, and the study was not designed for formal efficacy comparisons against standard therapy or against other KRAS-targeted approaches (Park et al., 2026).
There are also other limitations:
- Small subgroup sizes, especially in pancreatic cancer
- Exploratory biomarker analyses
- Lack of randomized control
- Limited ability to identify predictors of response or resistance
- Shorter follow-up in NSCLC than in pancreatic cancer
Those points matter because phase 1 success does not always translate into later-phase success. The current results justify enthusiasm, but they do not eliminate uncertainty.
Why This Study Matters Beyond KRAS G12D
Even beyond its immediate implications for KRAS-mutant cancer, this paper may matter for a broader reason: it provides credible clinical support for targeted protein degradation as a therapeutic strategy in solid tumors.
Protein degraders have attracted enormous interest across oncology and beyond, but the transition from elegant biology to reproducible patient benefit remains the critical test. Setidegrasib’s results suggest that degradation can be more than a platform promise. It can produce measurable, clinically relevant activity in an oncogene-driven solid tumor population.
That may prove to be one of the most lasting contributions of this study.
Where the Field Goes Next
The next phase of development will be crucial. Several questions now become central.
First, can the NSCLC signal be confirmed in larger, disease-specific expansion or registration-oriented studies?
Second, can the pancreatic cancer activity be optimized through better patient selection or combinations?
Third, what rational combinations might enhance depth and durability of response? The safety profile described here appears favorable enough to make that question clinically realistic (Park et al., 2026).
Finally, what biomarkers beyond KRAS G12D itself will identify patients most likely to benefit? Co-occurring genomic alterations, ctDNA dynamics, and tumor microenvironment features may all matter.
Conclusion
The phase 1 study of setidegrasib represents one of the most important early signals yet in the search for effective KRAS G12D-targeted therapy. In pretreated advanced NSCLC, setidegrasib produced an objective response rate of 36%, a median progression-free survival of 8.3 months, and an estimated 12-month overall survival of 59%. In pancreatic ductal adenocarcinoma, it also showed antitumor activity, with an objective response rate of 24% and median overall survival of 10.3 months in second- and third-line use (Park et al., 2026).
Just as importantly, the trial demonstrated strong pharmacodynamic evidence of KRAS G12D degradation, confirming that the drug is reaching and eliminating its intended target in human tumors. The safety profile was manageable, with infusion-related reactions common but generally low-grade, and treatment discontinuation due to adverse events remained rare.
Setidegrasib is not yet a new standard. But it is something potentially just as significant at this moment: proof that KRAS G12D degradation can translate into clinical activity. In a field that has spent years trying to turn KRAS G12D from a biologic fact into a therapeutic opportunity, that is a major step forward.


