The first-line treatment landscape for EGFR-mutated advanced non-small cell lung cancer has changed from a simple question of which tyrosine kinase inhibitor to use into a more complex discussion about how first-line therapy shapes the biology of resistance. The MARIPOSA trial has already shown that amivantamab plus lazertinib improves progression-free survival and overall survival compared with osimertinib in previously untreated EGFR-mutated advanced NSCLC. A new long-term resistance analysis now adds an important mechanistic layer: the combination appears to reduce the emergence of common EGFR- and MET-driven resistance mechanisms and may improve outcomes after patients move to subsequent therapy.
This brief report evaluates acquired resistance using paired baseline and end-of-treatment circulating tumor DNA and links those molecular findings with second-line progression-free survival. The central message is clinically important: first-line amivantamab plus lazertinib may not only delay first progression, but also change the resistance landscape that patients carry into the second-line setting.
Why Resistance Biology Matters After First-Line EGFR Therapy
Osimertinib has been a major standard in first-line EGFR-mutated advanced NSCLC, but acquired resistance remains nearly inevitable. Common mechanisms include MET amplification, secondary EGFR mutations such as C797S, and a broad range of EGFR/MET-independent pathways involving PI3K, RAS/RAF, cell-cycle alterations, and TP53/RB1 loss-of-function.
These resistance mechanisms matter because they influence what happens next. A patient progressing with complex, tractable resistance may have fewer effective downstream options and shorter disease control with second-line treatment. In contrast, patients without detectable known resistance mechanisms at progression may retain a less complex disease state and may respond better to subsequent therapy.
This analysis asks whether a broader first-line strategy, combining extracellular EGFR/MET targeting with intracellular EGFR inhibition, can reduce the emergence of these resistance pathways.
MARIPOSA Study Context
MARIPOSA enrolled patients with previously untreated locally advanced or metastatic NSCLC harboring common EGFR mutations, including exon 19 deletion and L858R. Patients were randomized to receive amivantamab plus lazertinib, osimertinib, or lazertinib alone, with this post-hoc exploratory analysis focusing on the amivantamab-lazertinib and osimertinib arms.
Amivantamab is an EGFR-MET bispecific antibody with immune cell-directing activity. Lazertinib is a central nervous system-penetrant, third-generation EGFR TKI. Together, the combination is designed to inhibit EGFR through complementary mechanisms while also suppressing MET-mediated escape.
In MARIPOSA, amivantamab plus lazertinib previously demonstrated superior first-line efficacy compared with osimertinib, including improved median progression-free survival and overall survival. This new analysis focuses on what happens at progression and beyond.

Read About MARIPOSA Trial on OncoDaily
Acquired Resistance Was Assessed by ctDNA
Acquired resistance mechanisms were evaluated using Guardant360 next-generation sequencing of circulating tumor DNA from paired baseline and end-of-treatment plasma samples.
Among patients who discontinued study treatment, matched baseline and end-of-treatment ctDNA data were available for 148 patients in the amivantamab-lazertinib arm and 198 patients in the osimertinib arm.
No clear resistance mechanism was detected in 68% of patients treated with amivantamab plus lazertinib compared with 59% of patients treated with osimertinib. While “unknown resistance” does not mean absence of resistance, it suggests that no established molecular resistance pathway was detectable by the assay used.
Amivantamab Plus Lazertinib Reduced MET and EGFR Resistance
The most important finding was a clear reduction in common EGFR/MET-dependent resistance mechanisms with amivantamab plus lazertinib.
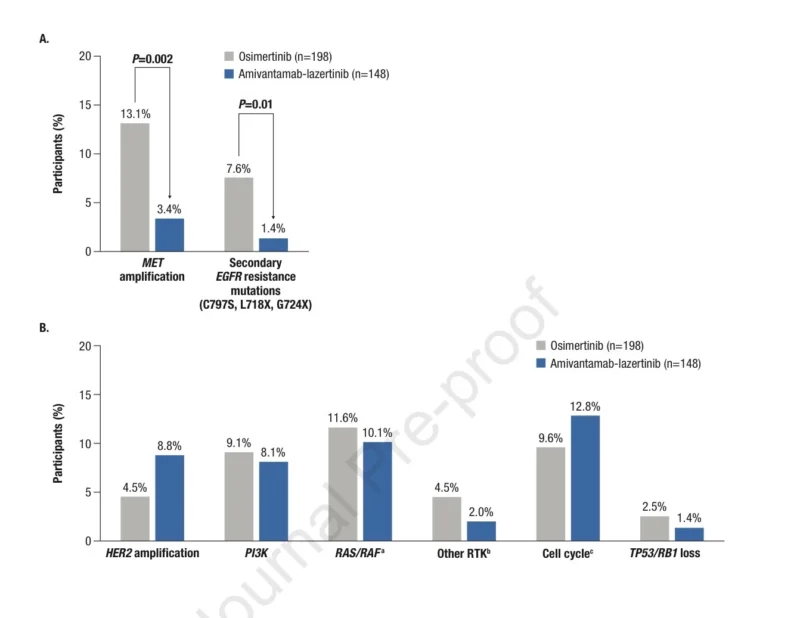
Compared with osimertinib, amivantamab plus lazertinib significantly reduced acquired MET amplification, observed in 3.4% versus 13.1% of patients. This represents an approximately fourfold reduction and was statistically significant.
Secondary EGFR resistance mutations were also reduced, occurring in 1.4% of patients receiving amivantamab plus lazertinib compared with 7.6% receiving osimertinib. These mutations included alterations such as EGFR C797S, L718X, and G724X.
Importantly, the reduction in EGFR/MET-dependent resistance did not appear to be offset by a significant increase in EGFR/MET-independent pathways, such as PI3K, RAS/RAF, cell-cycle alterations, or TP53/RB1 loss-of-function.
This suggests that the combination may suppress key resistance routes without simply shifting the tumor toward another dominant escape pathway.
Longer Amivantamab Exposure Was Associated With Fewer Resistance Events
The analysis also found that longer treatment with amivantamab was associated with fewer acquired MET and EGFR mutations.
Among patients who received at least six months of amivantamab, only 2% acquired MET amplification, and no patients acquired secondary EGFR mutations. This supports the hypothesis that sustained EGFR/MET-directed antibody pressure may help prevent the emergence of common resistance mechanisms.
The authors also reported that acquired MET amplifications were a common early resistance mechanism among patients who discontinued osimertinib within 12 months, while acquired EGFR mutations occurred only after at least 12 months of treatment. In contrast, acquired EGFR mutation rates remained low with amivantamab plus lazertinib across time points.
Resistance Complexity Was Lower With Amivantamab Plus Lazertinib
Beyond individual alterations, amivantamab plus lazertinib reduced resistance complexity and overall mutational heterogeneity at end of treatment compared with osimertinib.
This point is clinically relevant because resistance in EGFR-mutated NSCLC is often polyclonal. More complex resistance can limit the effectiveness of subsequent targeted or chemotherapy-based approaches. A first-line regimen that narrows the resistance landscape may preserve downstream treatment sensitivity.
This is one of the most interesting implications of the analysis: the benefit of first-line therapy may extend beyond the first progression event by shaping the molecular state of the disease that remains.
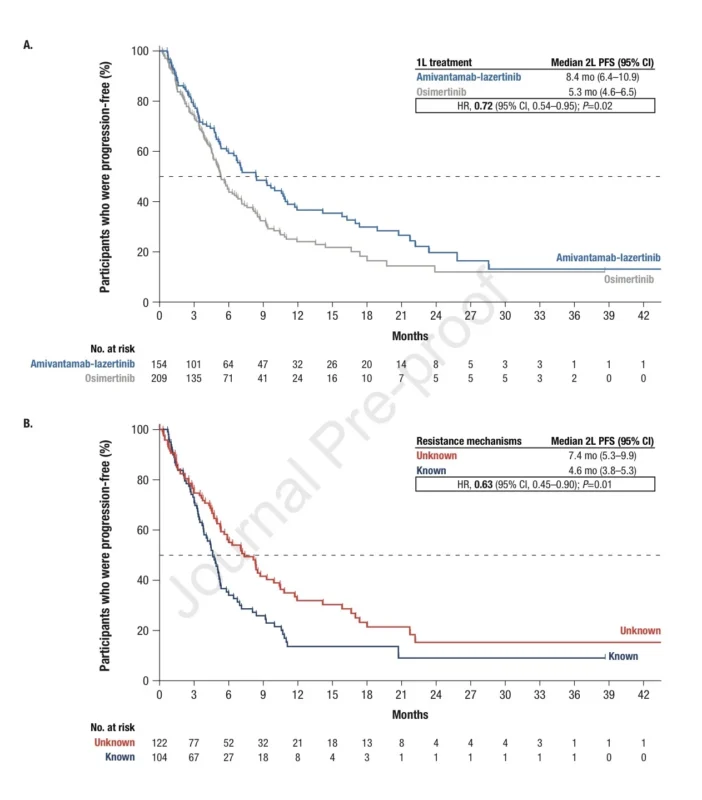
Second-Line PFS Was Longer After Amivantamab Plus Lazertinib
The resistance findings were supported by second-line outcome data.
Among patients who received a first subsequent therapy, median second-line progression-free survival was 8.4 months after first-line amivantamab plus lazertinib compared with 5.3 months after first-line osimertinib. The hazard ratio was 0.72, favoring the amivantamab-lazertinib arm.
This endpoint is distinct from PFS2 measured from randomization. Second-line PFS starts at initiation of subsequent therapy and therefore more directly reflects how prior first-line therapy may influence outcomes after progression.
The finding suggests that patients progressing after amivantamab plus lazertinib may enter the next treatment line with less resistant or less molecularly complex disease.
Unknown Resistance Was Associated With Better Second-Line Outcomes
Across both treatment arms, patients with unknown resistance at end of treatment had better second-line outcomes than those with known resistance mechanisms.
Median second-line PFS was 7.4 months in patients with unknown resistance compared with 4.6 months in those with known resistance. The hazard ratio was 0.63, suggesting that detectable resistance complexity was associated with shorter benefit from subsequent therapy.
Within the known resistance group, second-line PFS was similar for EGFR/MET-dependent and EGFR/MET-independent resistance mechanisms, at approximately 4.6 to 4.8 months.
This finding reinforces a practical concept: the molecular profile at progression is not only descriptive, but prognostic.
Why This Matters for First-Line Treatment Selection
The MARIPOSA resistance analysis supports a broader view of first-line therapy in EGFR-mutated advanced NSCLC. The goal is no longer only to maximize initial response or first PFS, but also to reduce the probability of biologically difficult resistance and preserve treatment sensitivity over time.
Amivantamab plus lazertinib appears to reduce the emergence of two major resistance pathways seen after osimertinib: MET amplification and secondary EGFR mutations. If confirmed with longer follow-up and larger resistance datasets, this could help explain the combination’s durable first-line and post-progression benefit.
This is especially important because a meaningful proportion of patients with EGFR-mutated advanced NSCLC never receive second-line treatment after progression. A first-line regimen that extends disease control and reduces resistance complexity may therefore have a larger impact than a sequence-dependent strategy that assumes all patients will remain fit enough for later therapy.
Limitations
Several limitations should be considered. This was a post-hoc exploratory analysis, and the acquired resistance dataset included only patients with paired baseline and end-of-treatment ctDNA samples. This introduces potential selection bias.
The analysis also relies on liquid biopsy, which may miss some resistance mechanisms, particularly histologic transformation or alterations with low ctDNA shedding. Some patients categorized as having unknown resistance may have had mechanisms not detectable by plasma sequencing.
Additionally, resistance data are still maturing because a substantial proportion of patients remained on treatment at the data cutoff, especially in the amivantamab-lazertinib arm.
The p values reported are nominal, so the findings should be viewed as exploratory rather than definitive.
Clinical Takeaway
This MARIPOSA analysis suggests that first-line amivantamab plus lazertinib changes the acquired resistance landscape in EGFR-mutated advanced NSCLC. Compared with osimertinib, the combination reduced MET amplification and secondary EGFR resistance mutations, lowered resistance complexity, and was associated with longer second-line progression-free survival among patients who received subsequent therapy.
The results support a key concept in modern thoracic oncology: first-line treatment can shape not only how long patients remain controlled initially, but also what kind of disease biology emerges at progression.
For EGFR-mutated advanced NSCLC, this may become one of the most important arguments for dual EGFR/MET targeting in the first-line setting.
Read full article here