Breast cancer remains the most commonly diagnosed malignancy in women worldwide and continues to represent a major public health challenge. Among its molecular subtypes, hormone receptor–positive, HER2-negative (HR+/HER2−) disease accounts for the majority of cases. Over the past two decades, treatment for this subtype has evolved from a relatively uniform approach based largely on clinicopathologic features to a refined, biologically driven strategy guided by genomic profiling and targeted therapy. What was once a debate between “chemotherapy versus endocrine therapy” has transformed into a sophisticated framework of molecular risk stratification, biomarker-directed escalation, and selective de-escalation.
The modern era of HR+/HER2− breast cancer management reflects one of oncology’s most important achievements: the ability to tailor treatment intensity without compromising outcomes. Large prospective trials have not only improved survival but have also prevented overtreatment for thousands of patients.
In this article, we highlight the pivotal trials that reshaped clinical practice in early-stage and metastatic HR+/HER2− breast cancer, beginning with the study that fundamentally changed how we decide who truly needs chemotherapy.
Early-Stage HR+/HER2− Breast Cancer
TAILORx Study – what it changed
The TAILORx trial represents one of the most practice-changing studies in early breast cancer. Prior to its publication, the use of adjuvant chemotherapy in hormone receptor–positive, node-negative disease was often recommended broadly, driven by historical data demonstrating proportional reductions in recurrence risk. However, clinicians increasingly recognized that many patients were likely being overtreated. The clinical question was clear: could genomic risk assessment safely identify women who derive little or no benefit from chemotherapy?
TAILORx was a large, prospective trial enrolling 10,273 women with hormone receptor–positive, HER2-negative, axillary node–negative breast cancer. All tumors underwent central testing using the 21-gene recurrence score assay. Women with low scores (0–10) received endocrine therapy alone. Those with high scores (≥26) received chemoendocrine therapy. The critical cohort consisted of 6,711 women (69% of the study population) with midrange recurrence scores of 11–25, who were randomly assigned to endocrine therapy alone or chemoendocrine therapy. The primary endpoint was invasive disease–free survival.
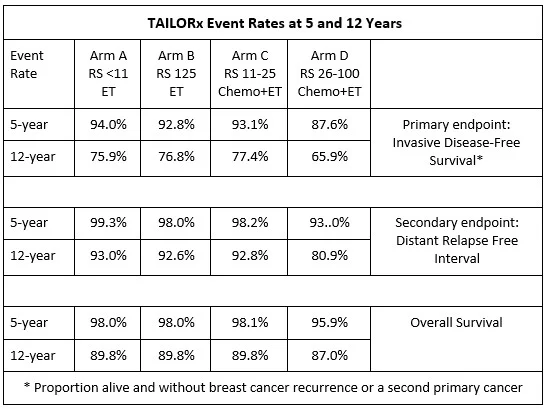
After a median follow-up of approximately 9 years, endocrine therapy alone was found to be noninferior to chemoendocrine therapy. The hazard ratio for invasive disease recurrence, second primary cancer, or death was 1.08 (95% CI, 0.94–1.24; P=0.26). At 9 years, invasive disease–free survival was 83.3% in the endocrine-only group and 84.3% in the chemoendocrine group. Freedom from distant recurrence was similarly high (94.5% vs. 95.0%), and overall survival was virtually identical (93.9% vs. 93.8%). These findings demonstrated that chemotherapy provided no meaningful benefit for the majority of women with midrange recurrence scores.
Importantly, exploratory analyses revealed an interaction with age. Among women 50 years of age or younger with recurrence scores between 16 and 25, chemotherapy was associated with a modest reduction in distant recurrence. However, in women over 50 with recurrence scores up to 25, and in younger women with scores ≤15, chemotherapy did not confer benefit. These nuanced findings refined clinical decision-making rather than producing a simplistic binary rule.
The implications of TAILORx were profound. The study prospectively validated the clinical utility of the 21-gene assay and demonstrated that approximately 70% of women with node-negative HR+/HER2− breast cancer could safely avoid adjuvant chemotherapy. More broadly, it established genomic-guided de-escalation as a standard principle in oncology. Rather than asking whether chemotherapy works in general, clinicians could now ask whether it works for a specific molecular risk group — and spare treatment when it does not.
MonarchE Trial- From Recurrence Reduction to Survival Benefit
If TAILORx defined who could safely receive less treatment, monarchE defined who requires more intensive therapy. Hormone receptor–positive, HER2-negative early breast cancer is characterized by a prolonged natural history, but patients with high-risk clinicopathologic features—particularly those with nodal involvement—remain vulnerable to distant relapse despite optimal endocrine therapy.
 The monarchE trial addressed a critical clinical question: can adjuvant CDK4/6 inhibition not only reduce recurrence but also improve survival?
The monarchE trial addressed a critical clinical question: can adjuvant CDK4/6 inhibition not only reduce recurrence but also improve survival?
monarchE was a global, randomized, open-label phase III trial enrolling 5,637 patients with HR-positive, HER2-negative, node-positive, high-risk early breast cancer. Patients were randomized to receive standard endocrine therapy for at least five years with or without abemaciclib for two years. High-risk disease was defined by ≥4 positive lymph nodes or 1–3 nodes with additional high-risk features such as tumor size ≥5 cm, grade 3 disease, or elevated Ki-67.
With longer follow-up, the trial has now delivered one of the most important advances in early breast cancer in decades: a statistically significant improvement in overall survival.
At a median follow-up of 76.2 months (approximately 6.3 years), abemaciclib plus endocrine therapy reduced the risk of death by 15.8% compared with endocrine therapy alone (HR 0.842, 95% CI 0.722–0.981; P=0.027). The estimated 7-year overall survival was 86.8% vs 85.0%, representing an absolute improvement of 1.8%.
Beyond survival, the durability of recurrence prevention remains a defining strength of monarchE. The benefit in invasive disease–free survival (IDFS) and distant relapse–free survival (DRFS) persisted well beyond treatment completion:
- IDFS: HR 0.734 (95% CI 0.657–0.820)
- DRFS: HR 0.746 (95% CI 0.662–0.840)
At 7 years:
- IDFS: 77.4% vs 70.9% (absolute benefit 6.5%)
- DRFS: 80.0% vs 74.9% (absolute benefit 5.1%)
Importantly, this translated into fewer patients progressing to metastatic disease, with 6.4% in the abemaciclib arm versus 9.4% in the control arm.

These findings are particularly significant in HR+/HER2− disease, where demonstrating an overall survival benefit in the adjuvant setting has historically been challenging due to long disease trajectories and effective subsequent therapies. monarchE therefore represents a rare and meaningful shift—from reducing recurrence risk to clearly improving survival outcomes.

“The results of monarchE represent a milestone in the adjuvant therapy of operable hormone receptor-positive/HER2-negative breast cancer. They are the first real advance in this field since the introduction of aromatase Inhibitors more than 20 years ago. In addition to its relevant efficacy in the reduction of relapses, adjuvant abemaciclib is probably the first drug to show a statistically significant increase in overall survival in a single trial”.
saied Miguel Martin, MD, PhD, Emeritus Professor of Medicine, Hospital General Universitario Gregorio Marañón, Universidad Complutense, Madrid
NATALEE: broadening CDK4/6 benefit beyond the “highest-risk only” group
NATALEE extended the CDK4/6 adjuvant concept into a broader stage II–III population, using ribociclib at a lower starting dose (400 mg) for a longer duration (3 years) alongside a nonsteroidal aromatase inhibitor. In the prespecified interim analysis (5101 patients; data cutoff January 11, 2023), ribociclib plus endocrine therapy significantly improved invasive disease–free survival compared with endocrine therapy alone (3-year iDFS 90.4% vs 87.1%; HR 0.75; 95% CI 0.62–0.91; P=0.003). Secondary endpoints also aligned in favor of ribociclib, including distant disease–free survival and recurrence-free survival, while overall survival remained immature at the time of reporting.
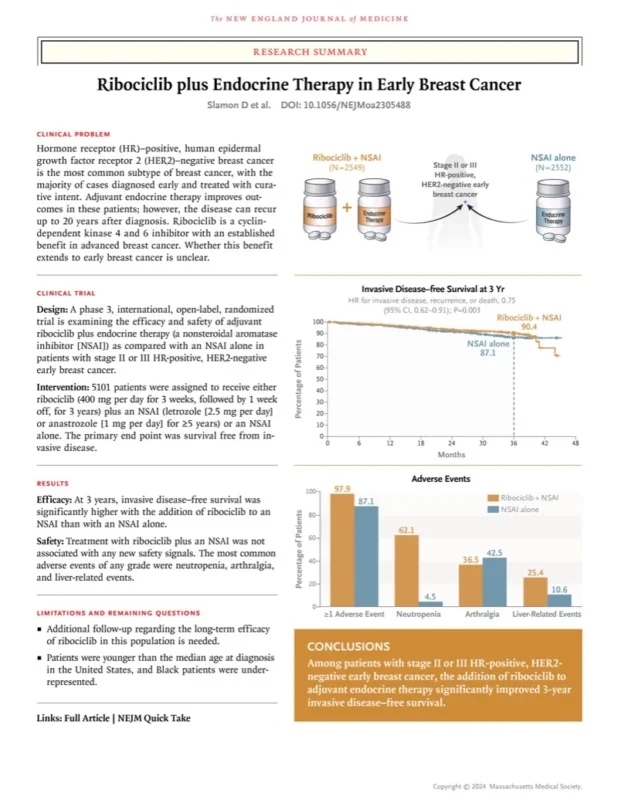
Clinically, NATALEE matters because it tests whether meaningful risk reduction can be delivered to a wider “at-risk” early-stage population—not only the most enriched, node-positive, highest-risk groups—while balancing tolerability through dose strategy and monitoring.
To Be Continued…


