For decades, the treatment of hormone receptor–positive (HR+), HER2-negative early breast cancer followed a predictable algorithm: surgery, radiotherapy when indicated, chemotherapy for high-risk patients, and long-term endocrine therapy.
And for many patients, that was enough.
But not for all.
Despite optimal endocrine therapy, up to 20–30% of patients with node-positive, biologically aggressive HR+ disease experience distant relapse, often within the first few years (Pan et al., 2017, New England Journal of Medicine). And in HR+ breast cancer, distant recurrence is not just progression, it is transformation into incurable metastatic disease. By 2026, the question is no longer whether endocrine therapy alone is sufficient for high-risk patients.
The question is: who needs more?
The monarchE trial answered that question.

The Biological Bet: Target the Cell Cycle Early
In metastatic HR+ breast cancer, CDK4/6 inhibitors transformed survival expectations. MONALEESA demonstrated overall survival benefit with ribociclib (Slamon et al., 2020, NEJM). PALOMA and MONARCH confirmed progression-free survival gains across multiple settings (Finn et al., 2016; Hortobagyi et al., 2016, NEJM).
The biological rationale was straightforward. Estrogen signaling activates cyclin D, which drives CDK4/6 activation. Inhibiting CDK4/6 arrests tumor cells in G1 phase. In metastatic disease, this translated into durable disease control.
But the leap into early disease was not guaranteed.
Minimal residual disease biology is different. Tumor burden is microscopic. Resistance dynamics differ. Previous adjuvant trials with palbociclib (PALLAS, PENELOPE-B) failed to demonstrate invasive disease-free survival (IDFS) improvement (Loibl et al., 2021, NEJM; Mayer et al., 2021, JCO).
Many questioned whether CDK4/6 inhibition was simply too late for early disease, or too toxic for a curative setting.
Then monarchE trial reported.
monarchE: A Trial Designed for Risk
monarchE trial did not enroll all HR+ early breast cancer. It selected the patients we worry about.
This global phase III trial enrolled 5,637 patients with HR+, HER2-negative, node-positive early breast cancer and clear high-risk features (Johnston et al., 2020, Journal of Clinical Oncology).
Eligibility required:
- Four or more positive lymph nodes, or
- One to three positive nodes plus tumor ≥ 5 cm, grade 3 histology, or Ki-67 ≥ 20%.
These were not borderline cases. These were patients with substantial early relapse risk.
Participants were randomized to:
- Standard endocrine therapy alone, or
- Endocrine therapy plus abemaciclib 150 mg twice daily for 2 years.
The primary endpoint: invasive disease-free survival. And this time, the strategy worked.
The Data That Shifted the Standard
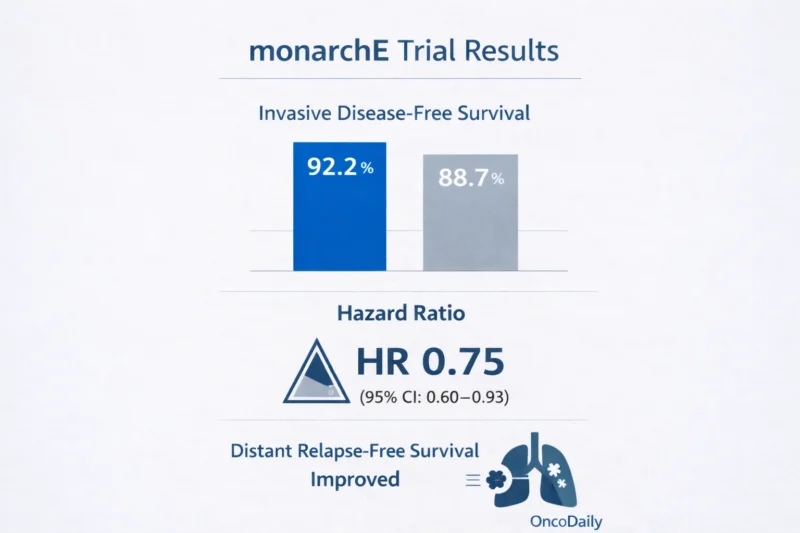
At interim analysis, abemaciclib reduced the risk of invasive recurrence by 25% (HR 0.75; 95% CI 0.60–0.93; P = .01) (Johnston et al., 2020).
Two-year IDFS rates were:
- 92.2% with abemaciclib
- 88.7% with endocrine therapy alone
In absolute terms, that difference matters, especially in a curative-intent setting. But what made monarchE trial compelling was not just IDFS. It was distant relapse-free survival.
Distant recurrence, the event we most fear, was significantly reduced (Harbeck et al., 2021, Annals of Oncology). The separation of curves persisted with longer follow-up.
By 2026, updated analyses continue to show maintained benefit. This was the first time a CDK4/6 inhibitor demonstrated meaningful benefit in the adjuvant setting.
Why Did monarchE Succeed Where Others Failed?
This remains one of the most discussed questions in early breast cancer. PALLAS and PENELOPE-B failed with palbociclib. monarchE trial succeeded with abemaciclib.
Several differences stand out:
Patient selection. monarchE focused exclusively on high-risk node-positive disease.
Continuous dosing. Abemaciclib is administered continuously, unlike palbociclib’s 3-weeks-on, 1-week-off schedule. Continuous inhibition may matter in minimal residual disease.
Pharmacologic differences. Abemaciclib has greater CDK4 selectivity and more sustained intracellular activity.
These distinctions highlight that CDK4/6 inhibitors are not interchangeable in early disease.
The Ki-67 Debate
Initial regulatory approvals emphasized Ki-67 ≥ 20% as a selection criterion.
However, subsequent analyses clarified that Ki-67 is prognostic, not predictive (Johnston et al., 2023, JCO). Benefit from abemaciclib was observed regardless of Ki-67 status within the high-risk population.
In 2026, most clinicians select patients based on clinical-pathologic high-risk features rather than Ki-67 alone. The trial taught us something important: risk matters more than proliferation markers alone.
Safety in a Curative Setting
Escalation in early breast cancer requires a higher tolerance threshold than metastatic therapy. The safety profile of abemaciclib in monarchE was consistent with prior experience.
Diarrhea was the most common adverse event, usually occurring early and manageable with loperamide and dose modification. Neutropenia was less severe compared with other CDK4/6 inhibitors. Venous thromboembolism was infrequent but clinically relevant.
In practice, proactive management strategies have made 2-year therapy feasible for most patients.
The key is education, early intervention, and dose flexibility.
monarchE in the Context of 2026
By 2026, abemaciclib plus endocrine therapy is incorporated into major international guidelines for high-risk HR+, HER2-negative early breast cancer. But the conversation has evolved.
Clinicians now consider:
- Absolute risk reduction versus toxicity
- Genomic assay integration
- Patient age and comorbidities
- Competing risk of late recurrence
monarchE shifted early HR+ disease from a “de-escalation era” into a selective intensification model.
Not everyone needs more therapy. But for the right patient, intensification is justified.

What About Overall Survival?
Overall survival data are maturing. While IDFS is an accepted endpoint in adjuvant trials, OS confirmation will further solidify practice.
Given the reduction in distant recurrence, OS benefit remains biologically plausible, though definitive results require longer follow-up.
The Bigger Picture: A Pattern in Breast Oncology
monarchE trial mirrors a broader trend in breast oncology. MONALEESA moved CDK4/6 inhibition from progression-free survival benefit to overall survival impact in metastatic disease.
monarchE trial moved CDK4/6 inhibition into early high-risk disease. And now, NATALEE explores ribociclib in a broader early-stage population.
The cell-cycle pathway has become central to both metastatic and early HR+ breast cancer management.

Final Perspective in 2026
monarchE did not change treatment for all HR+ early breast cancer. It changed it for the patients most likely to relapse early.
In a disease historically characterized by long natural history and late recurrence, monarchE focused on the early danger window, and demonstrated that targeted intensification can alter that trajectory.
In 2026, the shift is clear:
Endocrine therapy alone is no longer the ceiling for high-risk node-positive HR+ early breast cancer. For selected patients, the standard now includes 2 years of CDK4/6 inhibition. And that is a meaningful evolution in curative-intent therapy.
Written by Armen Gevorgyan, MD


