For years, hormone receptor–positive (HR+) metastatic breast cancer was described as “indolent.”
It wasn’t.
It was slower than triple-negative disease. It was more responsive to endocrine therapy. But resistance was inevitable. Median overall survival rarely crossed four years in metastatic settings. Treatment sequencing revolved around endocrine therapy first, chemotherapy later.
Then came the MONALEESA trial program.
Not loud. Not dramatic. No revolutionary biomarker discovery. Just survival. And in oncology, survival changes everything.
Before the MONALEESA Trial: The Endocrine Ceiling
Before the MONALEESA trial, first-line treatment for HR+/HER2-negative metastatic breast cancer centered on aromatase inhibitors or fulvestrant. Endocrine therapy delayed progression, but resistance developed through cyclin D–CDK4/6 pathway activation.
The biology was clear. HR-positive tumors rely heavily on cell-cycle progression mediated by CDK4 and CDK6. These kinases phosphorylate retinoblastoma protein (Rb), allowing transition from G1 to S phase.
Block the hormone receptor, and the tumor adapts. Block the cell cycle, and you may stop proliferation at its core. That was the rationale behind ribociclib , and the foundation of the MONALEESA trial.
What Is the MONALEESA Trial?
The MONALEESA trial program evaluated ribociclib, a selective CDK4/6 inhibitor, in combination with endocrine therapy in HR+/HER2-negative advanced breast cancer.
The program includes:
- MONALEESA-2 (postmenopausal, first-line letrozole)
- MONALEESA-3 (fulvestrant ± prior therapy)
- MONALEESA-7 (premenopausal women, ovarian suppression + endocrine therapy)
Each MONALEESA trial was randomized, phase III, and designed not just to show progression-free survival, but overall survival. That decision would define its legacy.
MONALEESA-2: Establishing the First-Line Standard
MONALEESA-2 evaluated ribociclib plus letrozole versus letrozole alone in postmenopausal women with previously untreated advanced disease. Initial results demonstrated a significant progression-free survival benefit (Hortobagyi et al., 2016, NEJM).
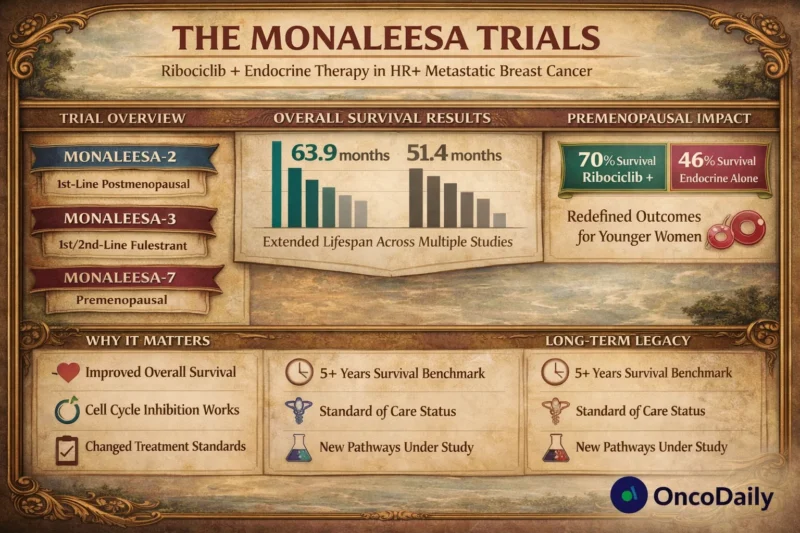
But the true impact came later. With extended follow-up, overall survival reached:
- 63.9 months with ribociclib + letrozole
- 51.4 months with letrozole alone
- Hazard ratio 0.76
This was one of the longest median overall survivals ever reported in this setting (Hortobagyi et al., 2022, J Clin Oncol). The MONALEESA trial had moved survival beyond five years.
Quietly.
MONALEESA-7: The Trial That Changed Premenopausal Treatment
Perhaps the most practice-changing part of the MONALEESA trial program was MONALEESA-7.
Historically, premenopausal women with metastatic HR+ disease were underrepresented in trials. MONALEESA-7 specifically addressed this population using ovarian suppression plus endocrine therapy with or without ribociclib.
The results were striking. Five-year overall survival rates:
- 70% with ribociclib
- 46% with endocrine therapy alone
Median overall survival was not reached at early analyses and later showed a clear advantage (Im et al., 2019, NEJM). This was not incremental benefit. It was survival redefinition in younger women. The MONALEESA trial did not just extend life, it extended life in a population historically overlooked.
MONALEESA-3: Beyond First-Line
MONALEESA-3 tested ribociclib plus fulvestrant in both first- and second-line settings. Again, overall survival benefit emerged.
Median overall survival:
- 53.7 months with ribociclib
- 41.5 months with placebo
- Hazard ratio 0.73 (Tripathy et al., 2018; updated OS 2021).
Three separate MONALEESA trials. Three separate overall survival benefits. Few oncology drug programs achieve this level of consistency.
Why the MONALEESA Trial Was Different
Other CDK4/6 inhibitors demonstrated progression-free survival benefit. The MONALEESA trial demonstrated overall survival across multiple populations. That distinction matters. Progression-free survival delays growth. Overall survival changes prognosis.
By proving OS benefit in postmenopausal, premenopausal, first-line, and later-line settings, the MONALEESA trial positioned ribociclib as one of the strongest CDK4/6 data packages in oncology.

Mechanistic Precision: Why Ribociclib Works
CDK4/6 inhibition halts cell-cycle progression by maintaining Rb in its hypophosphorylated state. HR-positive tumors are particularly dependent on cyclin D–CDK4 signaling.
In endocrine-resistant states, cell-cycle activation becomes a dominant escape mechanism (Finn et al., 2016). The MONALEESA trial validated that dual blockade, hormone receptor + CDK4/6 creates durable control.
This is not cytotoxic chemotherapy. It is biologic interruption of proliferation.
Safety and Long-Term Management
Ribociclib’s primary toxicity is neutropenia, typically manageable and rarely febrile. QT interval prolongation requires monitoring but is predictable and manageable.
Importantly, quality-of-life analyses within the MONALEESA trial program demonstrated maintained or improved patient-reported outcomes compared with endocrine therapy alone. In metastatic disease, survival without preserved quality is incomplete progress.
The MONALEESA Legacy in 2026
By 2026, CDK4/6 inhibitors are standard of care globally in HR+/HER2-negative metastatic breast cancer. But the MONALEESA trial stands apart because of its mature overall survival data.
The treatment algorithm today:
- First-line: CDK4/6 inhibitor + endocrine therapy
- Second-line: PI3K inhibitors, oral SERDs, or antibody-drug conjugates depending on mutation status
This structured sequencing exists because the MONALEESA trial reset expectations for first-line survival.
Is the Story Over? No.
Resistance to CDK4/6 inhibition emerges through:
- RB1 loss
- Cyclin E amplification
- PI3K pathway activation
New combinations targeting these escape pathways are under investigation. But the MONALEESA trial established a durable backbone. And that is its historical contribution.

Final Perspective
The MONALEESA trial did not introduce a flashy new drug class. It introduced survival consistency. It proved that targeting the cell cycle in HR-positive metastatic breast cancer extends life across populations, across lines of therapy, and across age groups. More than a decade ago, HR-positive metastatic disease was described as manageable.
Today, many patients live beyond five years. That shift is not accidental. It is the legacy of the MONALEESA trial.
Written by Armen Gevorgyan, MD


