Breast cancer screening is gradually moving beyond detection alone. A growing body of research suggests that a routine mammogram may contain important signals not only about cancers that are visible today, but also about cancers that may develop in the future. The Clairity model reflects this emerging direction in breast imaging, combining artificial intelligence with screening mammography to estimate a woman’s future breast cancer risk.
The technology recently reached an important clinical milestone. Clairity Model received FDA authorization through the De Novo pathway, becoming the first AI platform designed to estimate a woman’s five-year breast cancer risk directly from a screening mammogram. Shortly afterward, the first patient received a Clairity Breast risk score in February 2026, marking the beginning of real-world clinical implementation of AI-based mammography risk prediction.
This development builds on more than a decade of scientific work demonstrating that deep learning models can extract complex predictive patterns from mammographic images. These patterns often cannot be recognized by the human eye but may still contain important information about future cancer risk.

Why Breast Cancer Risk Prediction Needs Improvement
Traditional breast cancer risk models rely on variables such as age, reproductive history, family history, breast density, and known genetic mutations. While these tools remain valuable, they have clear limitations. Many women who develop breast cancer do not have strong family histories or identifiable genetic risk factors.
According to data reported by Clairity, approximately 85% of women diagnosed with breast cancer have no family history of the disease. This means that a large proportion of patients who ultimately develop breast cancer may initially be classified as average risk using traditional tools.
That gap has motivated researchers to explore whether mammographic images themselves could reveal risk signals that extend beyond density or visible abnormalities.
AI Models Can Learn Risk Patterns Directly From Mammograms
One of the most influential breakthroughs in this field came with the development of deep learning models that analyze mammograms to predict future breast cancer risk. A key example is the mammography-based deep learning model introduced in Radiology, which demonstrated that AI systems trained on screening mammograms could predict future cancer risk more accurately than several conventional risk calculators (Yala et al., 2019).
This work showed that mammograms contain complex structural information about breast tissue architecture. Deep learning algorithms are capable of identifying patterns across millions of pixels that may correlate with later cancer development, even when no visible lesion is present at the time of screening.
The idea that a mammogram could serve as a predictive biomarker represented a major shift in how imaging could be used in breast cancer prevention.
The Mirai Model Demonstrated Strong Predictive Performance
The field advanced further with the development of Mirai, a deep learning model designed to estimate breast cancer risk across multiple future timepoints. In the study Toward Robust Mammography-based Models for Breast Cancer Risk, Mirai was evaluated using screening mammography datasets from multiple international populations.
Across held-out datasets from the United States, Sweden, and Taiwan, Mirai achieved C-indices of 0.76, 0.81, and 0.79, demonstrating strong predictive discrimination across diverse populations (Yala et al., 2021).
Even more striking were the comparisons with established clinical models. For five-year breast cancer risk prediction, Mirai identified 41.5% of women who developed breast cancer as high risk, compared with 22.9% identified using the Tyrer-Cuzick model (Yala et al., 2021).
These results suggested that image-based AI models may detect risk signals that traditional clinical models cannot capture.
Validation Across International Screening Populations
Strong predictive performance in a single dataset is not enough to justify clinical use. AI models must demonstrate reliability across different healthcare systems, imaging equipment, and patient populations.
A multi-institutional validation study therefore evaluated mammography-based AI risk prediction across 128,793 mammograms from hospitals in several countries, confirming the model’s ability to estimate breast cancer risk across diverse populations (Yala et al., 2022).
Large-scale validation is particularly important in breast imaging because screening programs differ widely across regions. Models that maintain accuracy across multiple settings are more likely to translate successfully into routine clinical workflows.
This type of validation helped establish the broader scientific foundation for AI-driven mammography risk tools, including the Clairity platform.
Real-World Evidence From Large Screening Datasets
By 2025, research in this field had expanded into large real-world screening populations. Analyses presented at RSNA evaluated AI-based mammography risk prediction in more than 245,000 screening mammograms collected from five U.S. sites and one European site.
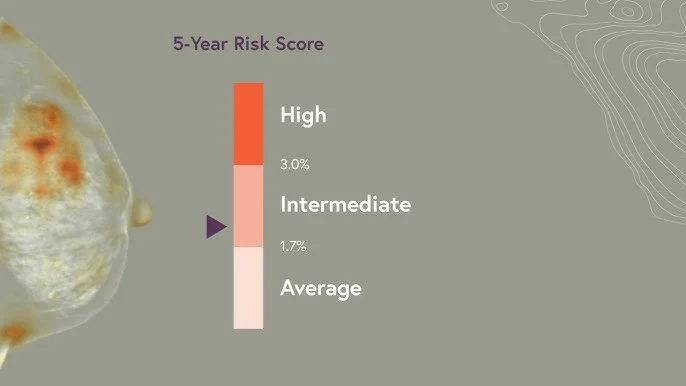
In these datasets, women categorized as high risk by the AI model had a breast cancer incidence of 5.9%, compared with 1.3% among women classified as average risk. For comparison, breast density alone separated risk much less effectively, with cancer incidence of 3.2% in high-density breasts versus 2.7% in lower-density breasts.
These findings suggested that image-based AI risk prediction may provide more precise risk stratification than density alone, which has historically been one of the most widely discussed imaging risk markers.
Another real-world validation study analyzed more than 120,000 mammograms across ten U.S. health systems, reporting an area under the curve (AUC) of 0.72 with good calibration between predicted and observed cancer outcomes.
Calibration is a critical feature for risk models used in clinical care. Accurate calibration ensures that predicted risk estimates correspond closely to real-world cancer incidence.
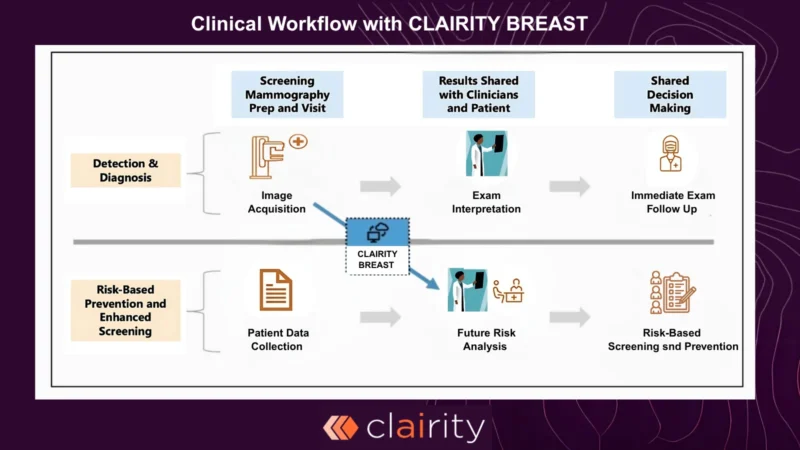
The Role Of The Clairity Model
The Clairity model represents one of the first attempts to translate these research advances into a clinical platform. Unlike traditional risk calculators, Clairity Model generates a five-year breast cancer risk estimate using only a screening mammogram, without requiring family history or other external clinical variables.
This design may allow the model to integrate seamlessly into routine screening workflows. When a patient undergoes mammography, the same image used for cancer detection can also be analyzed to estimate future risk.
Importantly, Clairity Model is not intended to detect cancer directly or replace radiologist interpretation. Instead, it provides a complementary risk assessment that can help guide conversations about screening strategy and preventive care.
What This Means For Breast Cancer Screening
The introduction of AI-based mammography risk prediction could have significant implications for breast cancer screening strategies. Current screening guidelines are largely based on age thresholds and general population risk estimates. While effective at a population level, this approach does not account for the substantial variation in risk between individuals.
AI-derived risk scores could support more personalized screening pathways. Women identified as higher risk might benefit from earlier follow-up imaging, supplemental screening techniques, or preventive interventions. At the same time, more precise risk stratification could potentially reduce unnecessary imaging in lower-risk individuals.
The growing evidence base supporting image-based risk prediction suggests that mammography may eventually serve a dual role: detecting cancers that are already present and identifying individuals who may develop cancer in the future.
A New Direction In Breast Imaging
The clinical introduction of the Clairity platform illustrates how rapidly the field of breast imaging is evolving. Research studies demonstrating predictive AI models, international validation datasets, and large real-world screening analyses have collectively shown that mammograms contain far more information than previously recognized.
Statistics from recent research illustrate the scale of this transformation. Deep learning models have achieved C-indices approaching 0.8 across international datasets, identified nearly twice as many future breast cancers as high risk compared with traditional models, and demonstrated risk separation of 5.9% versus 1.3% in large screening populations.
With FDA authorization now granted and the first patient already receiving a Clairity Breast risk score, AI-based mammography risk prediction has moved from research concept to early clinical practice.
For breast oncology and breast imaging communities, the key question now is how these tools will be integrated into real-world screening programs. If ongoing studies continue to confirm accuracy, calibration, and equitable performance across populations, the Clairity model and similar technologies could play an important role in the next generation of personalized breast cancer screening.


