Follicular lymphoma (FL) is the most common indolent non-Hodgkin lymphoma, although its biology is far from static. Over time, a subset of patients experience progression, transformation, or relapse into more aggressive disease. This duality – prolonged indolence coexisting with the potential for progression – defines the central challenge in FL management.
Against this backdrop, the treatment landscape is rapidly shifting. Long-standing approaches are being refined, while new therapies are redefining what is possible. This article explores FL and how these changes are shaping the future of its care.
Inside FL Pathobiology
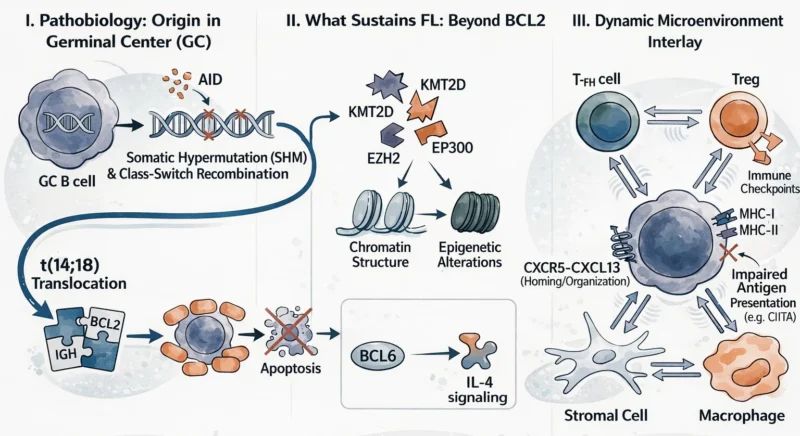
Follicular lymphoma (FL) originates from germinal center (GC) B cells and is shaped by a dynamic interplay between genetic alterations and the tumor microenvironment. Its defining molecular event is the t(14;18)(q32;q21) translocation, which places the BCL2 gene under the control of the immunoglobulin heavy chain (IGH) locus, resulting in constitutive overexpression of BCL2 and consequent inhibition of apoptosis.
The cell of origin has undergone somatic hypermutation and class-switch recombination -physiologic GC processes that carry an inherent risk of genomic instability. Activation-induced cytidine deaminase (AID), central to these mechanisms, introduces off-target mutations and facilitates chromosomal aberrations, seeding early oncogenic events. This ongoing mutagenic activity promotes subclonal diversification that drives disease evolution, progression, and resistance to therapy.
What sustains FL beyond BCL2
BCL2 overexpression is insufficient alone to drive lymphomagenesis. Additional genetic and epigenetic alterations accumulate over time. Among the most characteristic mutations are those involving epigenetic regulators such as CREBBP, KMT2D, EP300, and EZH2, the latter enhances histone methylation, reinforcing the germinal center phenotype, reducing MHC-I levels and promoting immune evasion. Approximately 15% of FL cases harbor BCL6-deregulating mutations, as a transcriptional repressor, BCL6 modulates the IL-4 response in B cells.
FL cells rely heavily on interactions with surrounding non-malignant cells, including T-FH cells, T regs, stromal cells, and macrophages. These interactions are mediated through cytokines, chemokines, and cell surface receptors, such as CD40-CD40L and CXCR5-CXCL13 signaling axes. T-FH cells secrete IL-4 and IL-21, which enhance B-cell survival and proliferation. Concurrently, mutations in MHC class II-related genes such as CIITA impair antigen presentation, while immune checkpoint upregulation drives T-cell exhaustion. Together, these features position FL as a disease of survival advantage rather than proliferative dominance.
Translating Biology into Therapy
Although biomarker-driven epigenetic targeting still requires clinical validation, preclinical data support preferential sensitivity of CREBBP- and/or EZH2-mutant cells to targeted approaches, including:
1. small-molecule inhibitors of the KAT3 acetyltransferase family, exploiting p300 paralogue lethality and currently in clinical trials
2. HDAC3 inhibitors targeting the BCL6 co-repressor complex that counteracts CREBBP activity
3. combination strategies in which HDAC3 or EZH2 inhibitors act as sensitizers to immunotherapy or immunomodulatory agents, potentially using fixed or intermittent dosing to limit toxicity (Merryman et al., 2026).
Clinical Course and Trajectory
FL typically presents with generalized painless lymphadenopathy, which is waxing and waning in nature. FL commonly involves axillary, cervical, femoral, and inguinal lymph nodes, rarely, it may present with an asymptomatic large mediastinal mass. Only 20% of patients with FL experience B symptoms, and increased serum LDH is also seen in 20% of patients.
In children, FL majorly affects tonsils and lymph nodes of the head and neck, consistent with what is now recognized as pediatric-type follicular lymphoma in the current WHO 2022 and ICC classifications. Although it may demonstrate high proliferative indices histologically, it behaves in a clinically benign manner, with excellent outcomes.
Marrow involvement occurs in most cases (>50%), with the paratrabecular pattern of infiltration being the most common. In some cases, lymphocytosis with atypical forms showing deeply cleft nuclei may be observed. Spleen infiltration presents as lymphocyte aggregates in the white pulp.
There are subtypes of FL, such as double-hit follicular lymphoma, now better conceptualized within the spectrum of high-grade B-cell lymphomas with MYC and BCL2 rearrangements – and low-grade FL with a high proliferation index, that present with low-grade morphology but a relatively aggressive clinical course.
Several extranodal variants exhibit distinct features. Primary cutaneous follicle center lymphoma typically presents with localized skin lesions – papules, plaques, or nodules – most often involving the scalp or trunk, and is notable for its excellent prognosis. Similarly, duodenal-type (gastrointestinal) follicular lymphoma is usually detected incidentally during endoscopy as small mucosal nodules and follows a highly indolent course, often managed conservatively.
These variants differ from nodal FL not only in their localization but also in their underlying biology, including reduced dependence on classic anti-apoptotic pathways (Freedman, 2024, Kaseb et al., 2024).
When Indolence Turns Aggressive: What Drives Transformation?
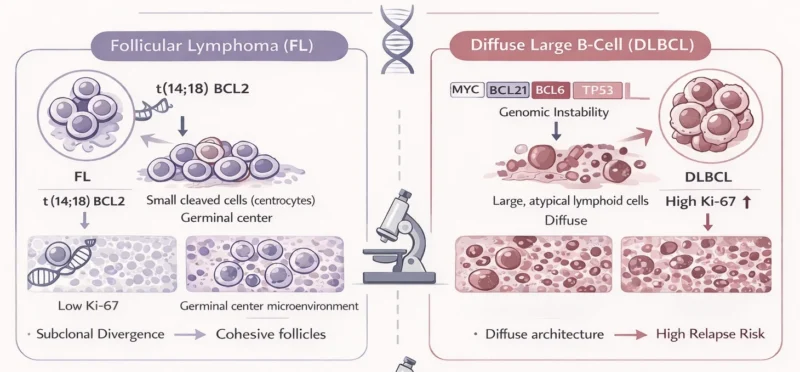
FL carries a lifelong risk of histologic transformation into a more aggressive lymphoma, most commonly diffuse large B-cell lymphoma (DLBCL). The cumulative risk of transformation is approximately 2-3% per year.
At the molecular level, transformation reflects clonal evolution through the acquisition of secondary genetic events, such as alterations in immune evasion pathways (B2M, CD58), MYC activation, biallelic TP53 inactivation, CDKN2A/2B loss. and disruptions in TME interactions (TNFRSF14). Notably, changes in macrophage polarization and T-cell exhaustion phenotypes have been implicated in promoting aggressive disease behavior.
Clinically, transformation often presents with rapidly enlarging lymphadenopathy, new extranodal involvement, rising LDH, and systemic “B” symptoms. However, transformation may be occult and can only be confirmed through tissue biopsy. Functional imaging with PET-CT can aid in identifying sites of suspected transformation, typically characterized by areas of high metabolic activity.
Early transformation, particularly within 24 months of initial diagnosis (POD24), is a strong predictor of poor outcome and reflects underlying high-risk biology. Therapeutically, management strategies often align with those used for aggressive lymphomas, including immunochemotherapy regimens and, in selected patients, ASCT or CAR-T therapy (Alizadeh et al., 2023).
Diagnostic Workup and Assessment
Morphological assessment of a lymph node excisional biopsy is crucial for diagnosing FL. Lymph nodes show variable-sized, closely packed follicles containing small cleaved cells without nucleoli (centrocytes) and larger noncleaved cells with moderate cytoplasm, open chromatin, and multiple nucleoli (centroblasts). The morphology typically demonstrates minimal apoptotic cells or tingible body macrophages. The mantle zones are typically absent, and necrosis is rare. Usually, there is interfollicular involvement or capsular infiltration.
Different architectural patterns include diffuse, floral, and incipient forms. FL cells express pan-B-cell markers. Occasionally, high-grade FL can lose CD10 expression while retaining BCL6 expression. The absence of BCL2 protein expression may be explained by an advanced histological grade or by mutations in BCL2.
Following the WHO HAEM5 (2022) guidelines, the traditional grading of FL (Grades 1, 2, and 3A) is no longer recommended. These grades are unified under the designation Classic Follicular Lymphoma (cFL). By contrast, grade 3B has been reclassified as a distinct high-grade entity: Follicular Large B-cell Lymphoma (FLBL), which is biologically similar to DLBCL. ICC 2022 maintains a similar lineage framework but retains grading (1-2, 3A, 3B) based on centroblast thresholds (>15 centroblasts/HPF for Grade 3), arguing for its continued therapeutic relevance in certain protocols.
MRD monitoring in FL has emerged as a powerful tool for refining response assessment and prognostication, although it is not yet universally integrated into routine clinical practice. The most established approach targets patient-specific immunoglobulin gene rearrangements or the BCL2/IGH fusion, using qPCR.
More recently, NGS-based assays have enabled more standardized detection of clonal sequences. Analysis of ctDNA in plasma is gaining attention for dynamic monitoring. Clinically, MRD negativity correlates with improved PFS and may help identify patients for treatment de-escalation or at risk of POD24 (Li et al., 2024, Peñalver et al., 2025).
Getting First-Line Therapy Right in FL
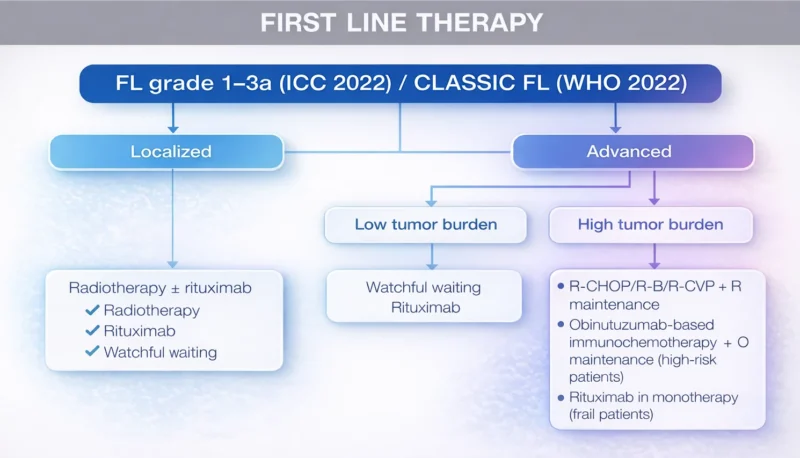
Management of FL depends on tumor burden and symptoms, with a nuanced approach for low tumor burden (LTB) disease. For asymptomatic patients, a watch-and-wait (W&W) strategy remains appropriate, with a median time to next treatment of 2.7 years. Early intervention with rituximab induction (RI), with or without maintenance (RM), prolongs this interval (RI: 9.9 years, RI + RM: not reached) without improving OS.
The RESORT trial confirmed that 63%-73% of patients remained cytotoxic-free at 7 years with either maintenance or retreatment at progression. While RI + RM improves health-related Qol, this must be balanced against risks of B-cell depletion, including infections and hypogammaglobulinemia, notably, nearly one-third of W&W patients avoid treatment even after 10 years.
In advanced-stage symptomatic or high tumor burden (HTB) FL, anti-CD20 antibody (rituximab or obinutuzumab) plus chemo is standard. Long-term outcomes are favorable, with 10-year PFS of 35%-51% and OS around 80%, suggesting a “functional cure” in some patients. The GALLIUM trial demonstrated improved PFS with obinutuzumab-based regimens compared to rituximab (7-year PFS: 63% vs. 56%), though without OS benefit.
Lower MRD negativity rates with R-CVP and R-CHOP compared to R-bendamustine are largely mitigated with obinutuzumab combinations. However, increased infectious toxicity (particularly with obinutuzumab and bendamustine) limits its use in older or comorbid patients.
Chemotherapy-free approaches, such as lenalidomide plus rituximab (R²), provide comparable 6-year PFS and OS to immunochemotherapy, as shown in the RELEVANCE trial, with distinct toxicities including rash and gastrointestinal effects. Although rituximab maintenance improves PFS, its lack of OS benefit and prolonged immunosuppression have led to variable adoption, with increasing interest in response-adapted and MRD-guided strategies (Kumar et al., 2025).
Is Frontline Treatment for FL About to Change?
Multiple randomized phase III trials are evaluating BsAbs in untreated FL, with the potential to significantly reshape treatment paradigms. Two CD3×CD20 BsAbs – mosunetuzumab and epcoritamab – are approved in multiply relapsed FL, while odronextamab and the CD3×CD19 agent surovatamig remain in late-stage development. In phase II trials of ≥2 prior lines, BsAbs achieved high ORR (78%-96%) and durable remissions (PFS 15-24 mo), with early-phase data also demonstrating promising frontline activity as monotherapy or combined with lenalidomide.
Epcoritamab showed CRR 63% and 18-mo PFS 49% in third-line FL and is being evaluated in the EPCORE FL-2 phase III trial (epcoritamab-R² vs CIT/R²), with dual primary endpoints of PFS and CR30 and secondary endpoints including QoL and MRD negativity. The inclusion of rituximab reflects the feasibility of combining BsAbs with anti-CD20 mAbs due to non-overlapping epitopes (epcoritamab 7D8), although CR30 – validated in CIT – requires further confirmation in non-CIT, PET-based settings.
Odronextamab has demonstrated robust efficacy in R/R FL in the ELM-2 trial (CRR 73%, median PFS 20.7 mo) and is being evaluated in frontline settings, including OLYMPIA-1 (monotherapy vs CIT) and OLYMPIA-2 (odronextamab-CHOP/CVP ± maintenance vs R-CHOP+RM), which will clarify the roles of monotherapy, chemo-combinations, and maintenance strategies.
In parallel, CD19-directed approaches are emerging, AZD0486 has shown activity in R/R FL and is being studied in the SOUNDTRACK-F1 (AZD0486+rituximab vs CIT, primary endpoint PFS). Collectively, at least six phase III trials enrolling nearly 5000 patients are evaluating BsAb monotherapy and combinations.
BsAbs exhibit a distinct toxicity profile compared to CIT, including predominantly low-grade CRS, infections, and immunosuppression, with long-term safety still evolving. Emerging data suggest that intermittent dosing may preserve T-cell fitness, highlighting dosing and sequencing as key determinants of efficacy and integration (Ng et al., 2025, Merryman et al., 2026).
Relapsed/Refractory FL: Where Complexity Begins
R/R FL represents a biologically and clinically heterogeneous entity, in which outcomes are largely dictated by disease kinetics and prior treatment exposure. Consequently, management is increasingly individualized, although optimal sequencing remains an area of active investigation.
POD24: Why Early Progression Matters
POD24, affects 10%-20% of patients with FL. Events are enriched for histological transformation (POD24 HT), underscoring the importance of repeat biopsy at progression in both clinical trials and real-world settings. Data from the LEO consortium (CReWE) demonstrated particularly poor outcomes for POD24 HT, with 5-year OS < 31%, whereas patients with biopsy-proven indolent disease (POD24 FL) had more favorable survival (71%). Notably, in some series, POD24 FL patients with low-intermediate FLIPI scores showed outcomes comparable to non-POD24 patients.
Therapeutic strategies remain an area of active investigation, and ASCT may offer a survival benefit in selected POD24 patients, although randomized evidence is lacking. The SWOG1608 trial (randomized, phase II) compares obinutuzumab-bendamustine, obinutuzumab-lenalidomide, and obinutuzumab-umbralisib to evaluate targeted and chemotherapy-sparing approaches in this high-risk population. Umbralisib (oral PI3Kδ inhibitor)-containing regimens were discontinued due to safety concerns, while the remaining arms demonstrated clinically meaningful activity.
Loncastuximab tesirine (CD19-directed ADC) demonstrated a CR rate of 67% with manageable toxicity in phase II data. The inMIND trial demonstrated that adding tafasitamab to R² improved PFS by 57% at a median follow-up of 14 mo. While BTK inhibitor monotherapy has shown limited efficacy in FL, the ROSEWOOD trial established the superiority of obinutuzumab-zanubrutinib (ZO) over obinutuzumab alone (ORR/CR: 69%/39% vs 46%/19%, median PFS 28 vs 10.4 mo), supporting its regulatory approval.
The MAHOGANY trial is comparing ZO with R², with PFS as the primary endpoint and QoL as secondary. Zanubrutinib is given continuously, whereas lenalidomide is limited to 12 cycles, which should be considered when interpreting results. ZO may be particularly suitable for frail patients due to its oral administration after obinutuzumab induction.
Unlike traditional IMiDs, CELMods (cereblon E3 ligase modulators) are designed for greater potency and selectivity, translating into enhanced antitumor activity with potentially improved tolerability. Agents such as golcadomide and iberdomide are under investigation across B-cell malignancies.
The GOLSEEK-4 phase III trial is evaluating golcadomide+rituximab vs investigator’s choice (R² or CIT) in R/R FL after ≥1 prior line including CD20 mAb and alkylator, with PFS as the primary endpoint, MRD and QoL as key secondary endpoints. This study directly challenges the role of lenalidomide in the second-line setting (El-Haddad, 2025, Ng et al., 2025)
The Rise of BsAbs in the R/R Setting
In the R/R setting, several phase III trials are directly comparing BsAb-based regimens with established standards. The CELESTIMO trial evaluates mosunetuzumab-lenalidomide versus R² in the second-line setting, with PFS as the primary endpoint. The OLYMPIA-5 trial investigates odronextamab-lenalidomide versus R².
The EPCORE FL-1 trial (phase III) comparing epcoritamab+R² vs R² after ≥1 prior line of CIT, met its dual primary endpoints at interim analysis (median follow-up 14.8 mo), demonstrating superior ORR (95% vs 79%) with estimated 16-mo PFS 85.5% vs 40.2%. Grade ≥3 AEs were higher with epcoritamab+R² (90% vs 68%), though CRS was predominantly low grade (grade 1-2, 26%) and manageable, with no severe events. Collectively, these trials will determine whether CD3×CD20 BsAbs can challenge the current second-line standard, which is largely dominated by R², and potentially redefine treatment sequencing in FL (Falchi et al., 2026).
Glofitamab, a CD3×CD20 IgG1 BsAb with a 2:1 CD20:T-cell binding configuration, is well studied in aggressive lymphomas, although data in FL remain limited. In a small study using step-up dosing (SUD), glofitamab demonstrated high efficacy: in the combination cohort with obinutuzumab, ORR was 100% with CR 74%, while in the monotherapy cohort, ORR was 81% with CR 67%-72% across dosing schemas. CRS was the most common AE (66% mono, 79% combo), predominantly low-grade, with only one grade ≥3 event, no ICANS was reported. Ongoing studies are evaluating glofitamab across multiple lines, combinations (including mAbs, ADCs), and post-CAR-T settings (El-Haddad, 2025).
You Can Watch More on OncoDaily Youtube TV
All Roads to Precision‐Approaches?
Three anti-CD19 CAR-T products – axicabtagene ciloleucel (axi-cel), tisagenlecleucel (tisa-cel), and lisocabtagene maraleucel (liso-cel) – are approved in the third-line setting for R/R FL. Axi-cel (CD28 costimulatory domain) demonstrated high efficacy in the ZUMA-5 trial (CRR 79%, median PFS 57 mo), albeit with higher toxicity (grade ≥3 CRS 6%, ICANS 15%).
In contrast, 4-1BB-based products showed favorable safety profiles with preserved efficacy: in ELARA (median 4 prior lines), tisa-cel achieved CRR 68% and 3-year PFS 53%, with no grade ≥3 CRS and 1% ICANS, in TRANSCEND FL (≥2 prior lines), liso-cel reported CRR 94% and 2-year PFS 73%, with grade ≥3 CRS 1% and ICANS 2%.
Ongoing phase III trials are evaluating earlier use and optimal positioning. ZUMA-22 compares axi-cel vs SOC (BR/R-CHOP or R²) after ≥2 prior lines, includes a POD24 second-line cohort, and uses PFS as the primary endpoint with QoL secondary endpoints, importantly, it incorporates long-term follow-up up to 15 years.
The LEDA trial is assessing tisa-cel vs SOC in a similar population, although without a dedicated POD24 cohort, which may influence its positioning in high-risk disease. For liso-cel, the planned TRANSFORM FL trial will compare liso-cel vs SOC in the second line, with PFS as the primary endpoint and PFS-2 as a key secondary endpoint.
A study indicated that bendamustine exposure <9 months before CAR-T apheresis had a detrimental effect on CAR-T efficacy. Although the impact of previous bendamustine on BsAbs efficacy is less pronounced, this is based on limited number of retrospective cases. As these trials unfold, we will gain valuable insights into the best strategies for positioning CAR-Ts in FL (Neelapu et al., Morschhauser et al., 2024 Ng et al., 2025).
CAR-T vs BsAbs as third or later-line therapy
According to a meta-analysis by Lawrence Cheng Kiat et al., (2025) the comparative efficacy of CAR-T and bispecific antibodies (BsAbs) in R/R FL remains primarily informed by single-arm trials, with no phase III randomized comparisons currently available. The analysis included 12 studies (n=795), evenly distributed between CAR-T (n=398) and BsAbs (n=397), and demonstrated superior response rates with CAR-T.
Pooled ORR was 93% for CAR-T versus 82% for BsAbs , while CRR was 82% versus 67%, corresponding to absolute benefits of 11% and 15%, respectively . However, in high-risk POD24 patients, this advantage was attenuated, with CRR 75% for CAR-T versus 69% for BsAbs , highlighting comparable activity in this subgroup.
These differences translated into improved durability with CAR-T therapy. At 6 months, pooled PFS was 85% for CAR-T compared to 74% for BsAbs, with sustained benefit at 1 year (74% vs 62%) and 2 years (62% vs 47%). A similar trend persisted at 3 years, suggesting more durable remissions with CAR-T. Despite these advantages in disease control, OS differences were modest, with comparable long-term outcomes between modalities, reflecting the indolent nature of FL and availability of multiple subsequent therapies .
Toxicity profiles differed between approaches. Rates of grade ≥3 CRS were low and comparable (CAR-T 3% vs BsAbs 4%), whereas ICANS was more frequent with CAR-T (8% vs 0%). Conversely, BsAbs were associated with higher rates of severe infections (17% vs 9%), likely related to prolonged B-cell suppression and extended treatment duration. Importantly, non-relapse mortality remained low and similar (3%).
Overall, CAR-T and BsAbs should be viewed as complementary rather than competing modalities in R/R FL. While CAR-T may confer superior efficacy in later lines, BsAbs offer accessible and effective alternatives, particularly in patients unsuitable for cellular therapy. Ongoing trials and longer follow-up will help determine optimal sequencing in this evolving landscape.
You can also read: Bispecific Antibodies vs CAR-T in Multiple Myeloma: Mechanisms, Clinical Outcomes, and Key Considerations
Written by Susanna Mikayelyan, MD
FAQ
What is follicular lymphoma and why is it considered indolent?
Follicular lymphoma (FL) is a type of non-Hodgkin lymphoma arising from germinal center B cells. It is termed indolent because it typically grows slowly and may not require immediate treatment, although it retains the potential to progress or transform into a more aggressive disease.
Can follicular lymphoma transform into a more aggressive cancer?
Yes, FL can transform into aggressive lymphomas such as diffuse large B-cell lymphoma (DLBCL). This occurs at a rate of approximately 2–3% per year and is associated with poorer outcomes.
What is “watch-and-wait” in follicular lymphoma management?
“Watch-and-wait” is an active surveillance strategy used in asymptomatic patients with low tumor burden. Treatment is delayed until disease progression or symptom development, as early therapy does not improve overall survival in many cases.
When is treatment required for follicular lymphoma?
Treatment is typically initiated in patients with high tumor burden, symptomatic disease, organ compromise, or rapid progression. Clinical criteria such as GELF or BNLI are often used to guide this decision.
What are the standard first-line treatments for follicular lymphoma?
First-line therapy usually includes anti-CD20 antibodies (e.g., rituximab or obinutuzumab) combined with chemotherapy (such as R-CHOP, R-CVP, or bendamustine-based regimens), or chemotherapy-free options like lenalidomide plus rituximab (R²).
What is POD24 and why is it important in follicular lymphoma?
POD24 refers to disease progression within 24 months of initial therapy. It identifies a high-risk subgroup of patients with significantly worse prognosis and often guides more aggressive or alternative treatment strategies.
What is the use of bispecific antibodies in follicular lymphoma?
Bispecific antibodies (BsAbs) are engineered therapies that simultaneously bind CD3 on T cells and CD20 on B cells, redirecting immune responses against lymphoma cells. They are currently approved in relapsed/refractory FL and being studied in earlier lines.
How does CAR-T therapy compare to bispecific antibodies in follicular lymphoma?
CAR-T therapy generally shows higher response rates and more durable remissions, but with increased toxicity. Bispecific antibodies offer a more accessible, off-the-shelf alternative with a different safety profile.



