In the era of evolving immunotherapy and personalized medicine, T cell-redirecting therapies have emerged as a promising addition to the anti-myeloma armamentarium. By harnessing the immune system against malignant plasma cells, they offer unique therapeutic advantages. This review highlights the key differences in mechanisms, toxicity profiles, therapeutic efficacy, resistance pathways, and emerging approaches aimed at optimizing patient outcomes.

Multiple Myeloma: Symptoms, Causes, Stages, Diagnosis and Treatment
Quick Take: Multiple Myeloma and the Rising Need for T-Cell Therapies
Multiple myeloma (MM) is a plasma cell malignancy characterized by the proliferation of clonal plasma cells in the bone marrow and the production of monoclonal immunoglobulin and/or light chain, with resultant end-organ damage, including bone destruction, hypercalcemia, anemia, renal damage, and increased susceptibility to infections. MM accounts for approximately 15% of hematological cancers. The age of onset culminates in the 6th and 7th decade of life. The global incidence of MM is estimated at 1.8/100,000 people.(Huang J et al., 2022)
The treatment paradigm in myeloma is constantly changing. Upfront use of monoclonal antibodies (MoAbs) along with proteasome inhibitors (PIs), and immunomodulators (IMiDs) has significantly improved survival outcomes. Nonetheless, relapse is inevitable with subsequent remissions being shorter due to acquired drug resistance and development of refractory disease.
Patients that are triple-class refractory (refractory to a PI, IMiD and anti-CD38 MoAb) have a dismal prognosis with a reported overall response rate (ORR) of 31%, a median progression free survival (PFS) of 3.4 months and a median overall survival (OS) of 9.3 months with the subsequent treatment regimens following anti-CD38 MoAb failure. Additionally, patients with penta-refractory myeloma (refractory to lenalidomide, pomalidomide, bortezomib, carfilzomib and an anti-CD38 MoAb) have an even more grim prognosis with a median OS of about 6 months with subsequent therapies.(Parrondo RD et al., 2024)
Ineffective T-cell immunity has been associated with the development of relapsed/refractory multiple myeloma (RRMM) and disease progression due to mechanisms such as T-cell anergy, exhaustion and senescence. However, studies show that endogenous T cells from RRMM patients can mount anti-myeloma cytotoxic activity when stimulated ex vivo, therefore, immune T-cell engagement through chimeric antigen receptor T-cell (CAR T-cell) therapy and bispecific antibodies (BsAbs) has emerged as an important therapeutic strategy in RRMM.
Living Medicine Engineered to Fight: CAR-T in Multiple Myeloma
CAR-Ts are genetically engineered cells equipped with a new receptor to specifically recognize and destroy antigen-positive target cells. This receptor (CAR) is an artificial fusion protein that comprises an extracellular antigen-binding domain, followed by a hinge region, a transmembrane domain, a costimulatory domain, and an intracellular signaling domain. After recognition of its specific antigen on the surface of tumor cells, CARs initiate a cascade of cytotoxic signaling (slow-acting/fast-acting killing mechanisms), leading to tumor lysis. A major advantage of CAR-T is the MHC- independent target antigen recognition, moreover, CAR-T are a living drug, which proliferates and persists within the patient providing long-term tumor surveillance.
B-cell maturation antigen (BCMA, CD269) is a type-III transmembrane protein belonging to the tumor necrosis factor receptor family. BCMA by binding to natural ligands, induces B-cell proliferation, survival as well as maturation and differentiation into plasma cells. It is expressed on terminally differentiated normal B cells, plasma blasts and on MM cells in most of the patients making it a suitable target for CAR-T therapy. At present, two BCMA-targeted CAR T-cell therapies, idecabtagene vicleucel and ciltacabtagene autoleucel, are approved by the FDA and EMA for the treatment of MM.
Idecabtagene vicleucel (Ide-cel, ABECMA) is manufactured from autologous peripheral blood mononuclear cells (PBMCs), stimulated with CD3/CD28 mABs in addition with IL2. Following stimulation, the cells are transduced with a lentiviral vector encoding a BCMA-directed CAR. This CAR consists of a murine anti-BCMA single-chain variable fragment (variable light and variable heavy regions of antibodies which are connected through a linker) fused to a CD8α-derived hinge and transmembrane region, a 4-1BB co-stimulatory domain and CD3ζ signaling domain. It should be noted that scFvs have a tendency to aggregate, leading to VH-VL mispairing and increased tonic signaling independent of target binding. Therefore, alternative strategies have emerged, including the utilization of VHH single domains of camelid antibodies, natural ligands, or synthetic binding domains.
Ciltacabtagene autoleucel (Cilta-cel, CARVYKTI) is produced from purified T cells isolated from autologous PBMCs. It contains a BCMA-directed CAR consisting of two distinct camelid heavy-chain-only variable domains (VHHs) and is otherwise structurally similar to Ide-cel.(Scheller L et al, 2024)
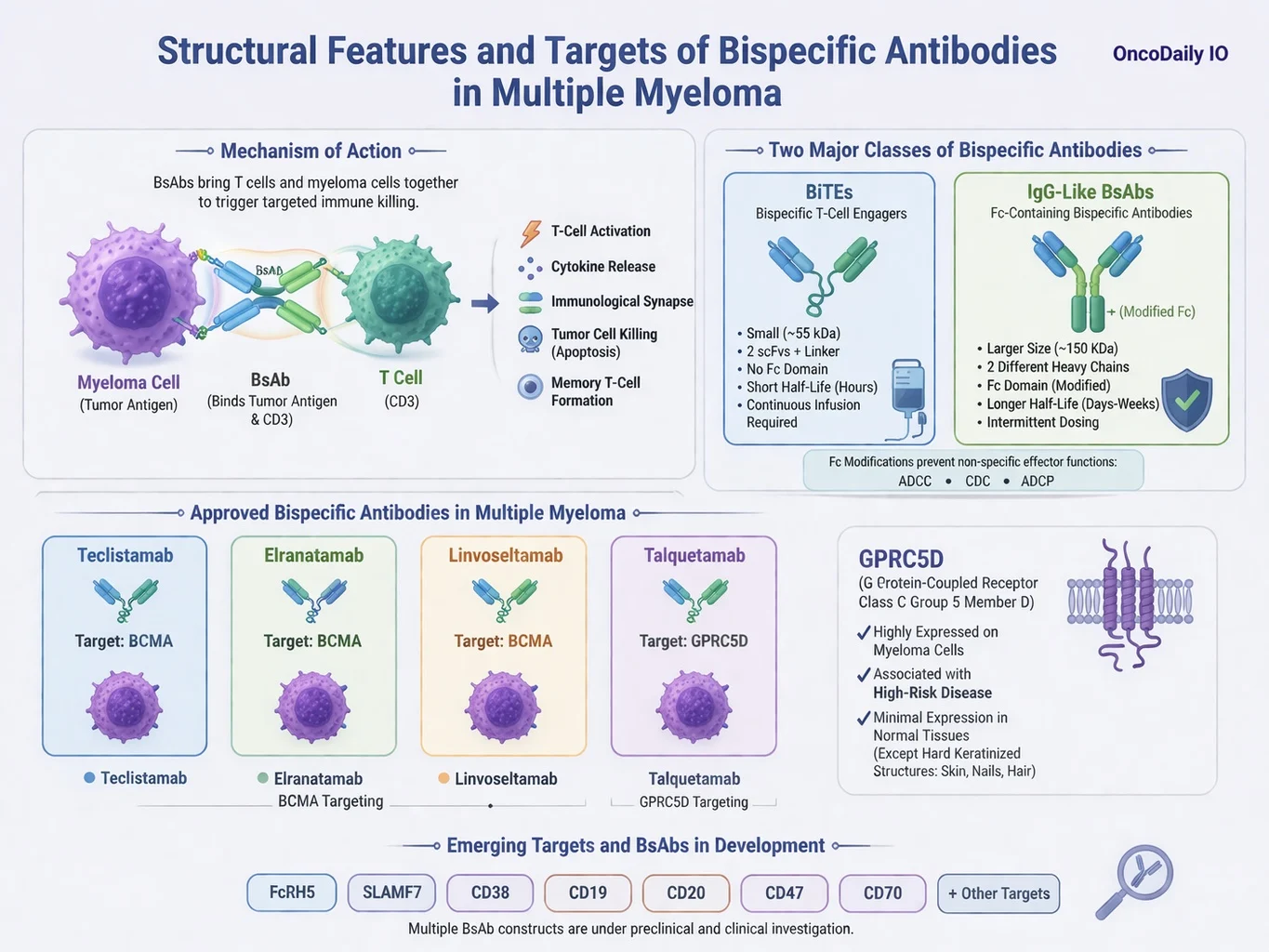
Structural Features and Targets of Bispecific Antibodies in Multiple Myeloma
BsAbs are bio-engineered antibodies. In contrast to endogenous antibodies, where the two binding sites target one specific antigen (bivalent monospecific), BsAbs can bind two distinct antigens or epitopes (bivalent bispecific). They can simultaneously bind a tumour-specific antigen on the targeted cancer cell and the effector T-cell CD3 co-receptor, triggering T-cell activation, the release of inflammatory cytokines, and the formation of an immunological synapse. Subsequent T-cell degranulation and release of perforin and granzyme B mediate the killing of target cells via apoptosis, Furthermore, these agents promote activation of naïve T cells and their differentiation into central and effector memory T-cell phenotypes.
The two major classes of BsAbs of clinical relevance in MM are:
- Bispecific T-cell engagers (BiTEs)
- IgG-like bispecific antibodies
BiTEs are small (about 55 kDa) proteins composed of two scFvs connected by short linker peptide sequences. Due to their small size and absence of an Fc domain, BiTEs have a very short half-life of just a few hours, and consequently necessitate continuous infusion to sustain adequate levels in the circulation.
Conversely, IgG-like BsAbs are Fc-containing and are composed of two different heavy chains (heterodimeric) derived from different antibodies. IgG-like BsAbs have longer half-lives, given their larger size and Fc domain, and as a result, are dosed intermittently. Fc-containing T-cell engagers undergo modifications to inactivate their Fc domain to prevent non-specific effector functions, including antibody-dependent cell-mediated cytotoxicity (ADCC), complement-dependent cytotoxicity (CDC), and antibody-dependent cellular phagocytosis (ADCP).
There are currently 4 commercially approved bispecific antibodies: teclistamab, elranatamab, and linvoseltamab, which target BCMA, as well as talquetamab, which targets the GPRC5D antigen on the surface of plasma cells.
GPRC5D (G protein-coupled receptor class C group 5 member D) is an orphan receptor whose expression is absent from nearly all healthy tissues, with the exception of hard keratinized structures, it is overexpressed in malignant plasma cells and associated with high-risk disease. There are other myeloma cell targets being studied preclinically and clinically, along with their corresponding BsAb constructs and effector cell antigen targets.(Rees M et al., 2024, Devasia A. J. et al., 2024)
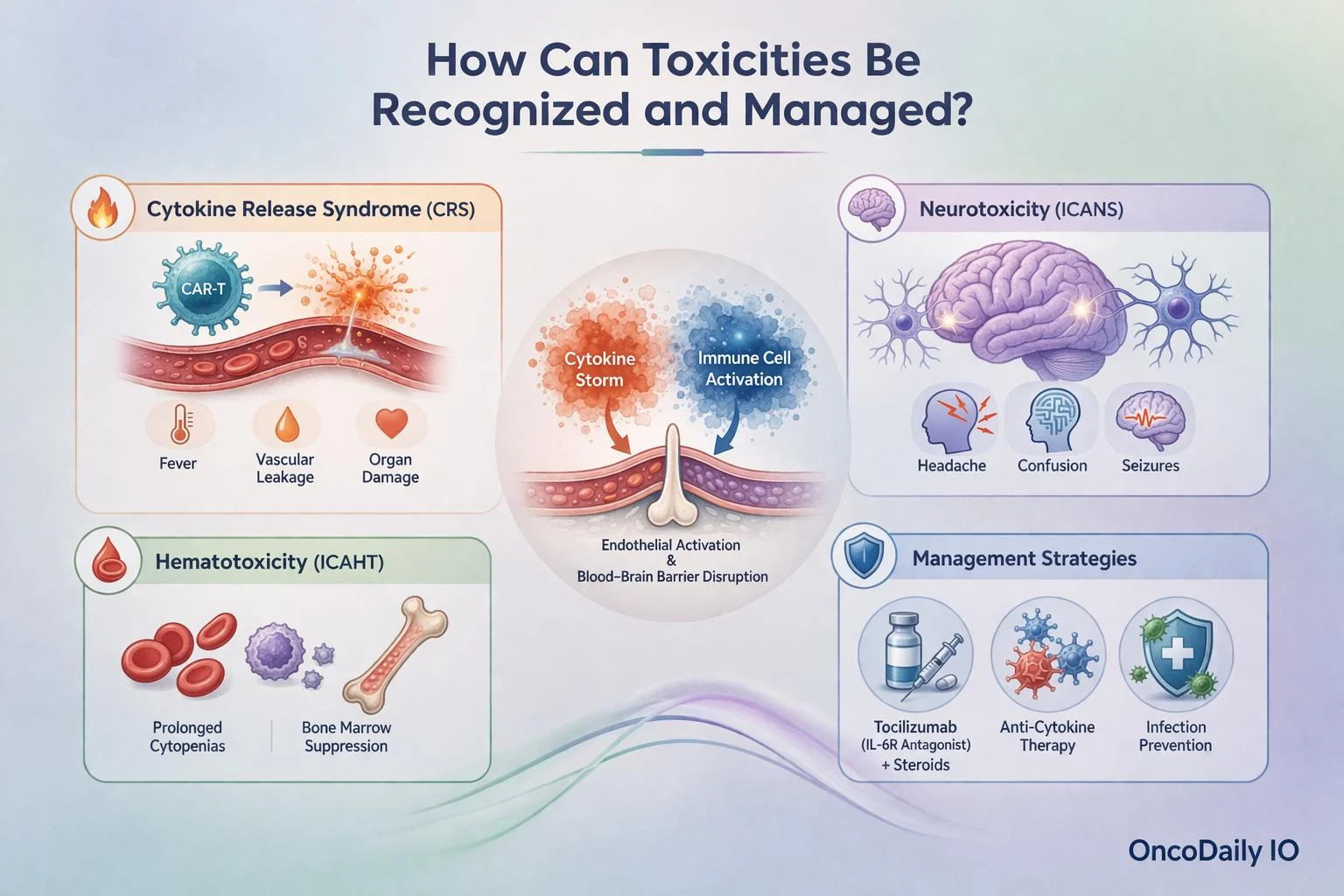
How Can Toxicities Be Recognized and Managed?
The main CAR-T–associated toxicities are:
- Cytokine release syndrome (CRS)
- Immune effector cell–associated neurotoxicity syndrome (ICANS)
- Immune effector cell–associated hematotoxicity (ICAHT)
Antigen-specific activation and proliferation of CAR-T cells trigger cytokine production by both CAR-T and bystander cells, particularly myeloid cells, leading to tumor cell lysis. Rapid expansion generates large amounts of proinflammatory cytokines, causing a systemic cytokine storm and CRS. This hyperinflammatory state results in endothelial dysfunction, vascular leakage, and disruption of the blood-brain barrier. ICANS develops as cytokines, CAR-T cells, endogenous T cells, and peripherally activated monocytes invade the cerebrospinal fluid and CNS. Cilta-cel has been associated with new neurotoxicities, including Parkinsonian movement and neurocognitive events, linked to BCMA expression on neurons and astrocytes in the basal ganglia.
Prolonged cytopenias characterize ICAHT and may result from immune/cytokine–mediated stem cell suppression, autoimmune cytopenias, persistent CAR-T effects, HLH, or infections. Secondary marrow neoplasms can also occur, especially in heavily pretreated patients, highlighting the need for early bone marrow evaluation. The pillars of treatment for CRS and ICANS remain tocilizumab (IL-6 receptor antagonist) and glucocorticoids. Novel strategies under investigation include:
- Anti-cytokine drugs (anakinra, emapalumab)
- Tyrosine kinase inhibitors (dasatinib, ibrutinib, ruxolitinib/itacitinib)
- T-cell–targeting approaches (antithymocyte globulin, cyclophosphamide)(Scheller L et al., 2024)
CRS and ICANS are less frequent and less severe with BsAbs (64.9% and 3.96% respectively) than with CAR-T. Ramp-up dosing and pre-emptive strategies, such as tocilizumab and corticosteroids, have further reduced these toxicities without affecting treatment response. The major drawback of BsAbs is their increased infection risk. Notably, single-target BCMA agents showed higher infection rates (21.63% grade 3-4) compared to GPRC5D therapy (14.51% grade 3-4). Current guidance recommends monthly immunoglobulin replacement for IgG < 400 mg/dL, continuing throughout immunoparesis on BCMA BsAbs, even in the absence of infections.
Cytopenias are another common adverse event, with grade ≥3 neutropenia observed in 35.17% of patients (meta-analysis). The use of G-CSF is recommended for grade 3–4 neutropenia, along with consideration of antibacterial, antiviral, and antifungal prophylaxis. Modifying the BsAb maintenance schedule can also reduce infectious and myelosuppressive side effects. Switching to a biweekly schedule with elranatamab in MagnetisMM-3 maintained treatment responses while lowering grade 3-4 adverse events from 59% to 47%.
Toxic effects on skin, nails, and oral mucosa are common with GPRC5D-targeting BsAbs due to receptor expression in keratinized structures. In the phase 1 MoumenTAL-1 study, talquetamab caused nail- and skin-related adverse events in 27–57% and 67–70% of patients, respectively. Most events were mild, and no grade 3 rash was reported.(Liang X et al., 2024, Raza S et al., 2025)
Behind the Scenes of Therapeutic Failure
Resistance to BCMA-directed CAR-T therapy in MM can arise from:
- T-cell-intrinsic factors
- Tumor-intrinsic factors
- Tumor microenvironment (TME)-intrinsic factors
Preexisting terminally differentiated or exhausted T cells may lead to poor CAR-T expansion and limited persistence after infusion. Tumor-intrinsic factors, such as extramedullary disease, high-risk cytogenetics, and clonal heterogeneity, can reduce treatment efficacy, antigen escape through downregulation or loss of BCMA has also been observed following therapy. Additionally, an immunosuppressive tumor microenvironment, characterized by regulatory T cells and myeloid-derived suppressor cells, may hinder CAR-T cell activity.
Mechanisms of resistance of BMCA- and GPRC5d-targeting bispecific antibodies in RRMM to date have been characterized by high disease burden and sBCMA “sink effect”, T-cell exhaustion and antigen escape. Gamma-secretase directly sheds membrane-bound BCMA from plasma cells, leading to increased levels of circulating soluble BCMA. The “sink effect” created by high levels of sBCMA acts as a decoy for BsAb binding.
Target antigen loss can happen due to rare events of biallelic deletions or mutations affecting the BCMA gene. This has been reported rarely in the context of BCMA CAR T-cell therapy and more commonly in the context of BCMA-BsAbs. Dual-antigen targeting to overcome BCMA attenuation, as seen in the RedirecTT-1 trial with teclistamab and talquetamab, appears to be effective.(Rees M et al., 2024)
Correlative studies from MajesTEC-1 and MagnetisMM-1 indicate that the baseline T-cell landscape strongly influences BsAb efficacy. Non-responders exhibit a depleted immune profile, including reduced CD8+ naïve T cells, higher frequencies of Tregs, MHC class I loss, target antigen downregulation (even selective outgrowth of preexisting antigen-negative clones), and abundant CD8+ terminally exhausted cells. Preclinical studies show that combining bispecific antibodies with IMiDs enhances T-cell activation but may worsen exhaustion, whereas cyclophosphamide reduces tumor burden, depletes regulatory T cells, and limits exhaustion. Studies are underway combining talquetamab or teclistamab with a PD-1 inhibitor (TRIMM3 trial).(Chari A et al., 2024)
Translational Horizons in T-Cell Redirecting Therapy
Several strategies can be utilized to target multiple antigens via CAR-T therapy. Tandem or bivalent CAR constructs harbor two distinct antigen-binding sites on a single extracellular domain. Bicistronic CAR-T products express distinct, unique CARs from a single vector to allow dual targeting through separate extracellular motifs. Co-transduction of different CAR vectors results in a heterogeneous product with subsets harboring one of each individual CAR construct, in addition to a third subset harboring both CARs.
Yan et al. published phase-II data on simultaneous co-administration of anti-CD19 CAR-T along with anti-BCMA CAR-T for the treatment of RRMM. Overall, 21 patients were treated, with a ORR of 95%. Among responders, 85% had durable responses without relapse at the time of data cut-off. Grade 1-2 CRS occurred in 86% of patients, while 5% developed grade 3 CRS, and 10% experienced CAR-related encephalopathy syndrome.
Utilizing nonviral transfection can reduce costs and does not require S2 Good Manufacturing Practice (GMP) laboratories which could increase availability of CAR T therapy to more clinical centers and patients. Currently, both preclinical and clinical studies have demonstrated stable gene transfer efficiency in the manufacturing of CAR-T using transposon systems. Transposon systems prove more suitable for multi-targeted CAR-T production when compared to viral vectors, owing to their reduced complexity.
When using patient T cells in the manufacturing process, three major challenges arise:
- Long vein-to-vein time
- T-cell dysfunction
- High costs associated with individualized production
Allogeneic CAR-T could potentially address these challenges. Nonetheless, it faces the risks of life-threatening graft-versus-host disease (GVHD) and rapid elimination of infused T cells by the host immune system. Interim safety and tolerability results from phase-I UNIVERSAL in RRMM that used allogeneic BCMA CAR-T engineered to limit expression of αβ TCR and lymphodepletion of CD52+ cells showed promising results.
Trispecific antibodies targeting two malignant plasma cell antigens and a T-cell or NK-cell antigen to stimulate cytotoxicity are in clinical development for RRMM.(De Marco et al., 2023, Tebuka E et al., 2024)
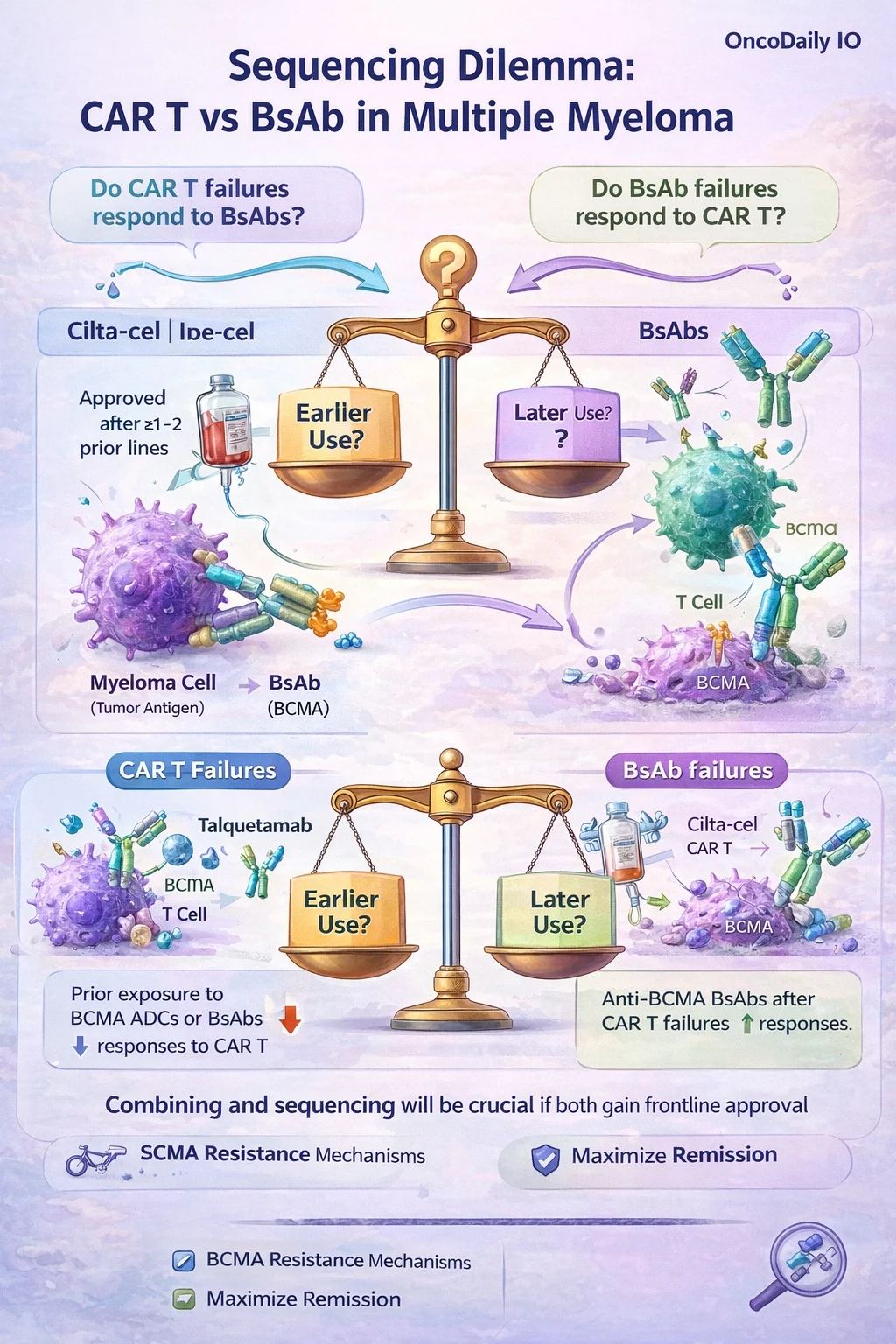
Is There Clarity on How to Sequence BsAbs and CAR T-Cell Therapy?
Even in the relapsed setting, in which both modalities are approved, there is a paucity of data defining ideal positioning. Cilta-cel and ide-cel are approved for earlier use, after 1 and 2 prior lines, respectively, based on phase 3 data, unlike bispecific antibodies, which are approved after ≥4 prior lines based on single-arm studies. There are many ongoing and upcoming trials testing the combination of various BsAbs (alone or in combination) with other agents in the newly diagnosed setting.
Sub-analyses of the KarMMa and CARTITUDE studies showed that prior exposure to anti-BCMA ADCs or BsAbs reduced responses to CAR T products. Conversely, anti-BCMA BsAbs after CAR T failure demonstrated favorable responses, albeit with short PFS.
In MonumenTAL-1, talquetamab’s ORR was 75% in patients with prior BCMA CAR T with a median DOR of 12.3 months. Conversely, for patients with prior BCMA BsAb therapy, talquetamab’s ORR was 52%, which fell further to an ORR of 29% in patients who received a BsAb as the immediate prior line.
Additional attempts to utilize better T cell function at the precursor stages of the disease are also underway. The randomized phase 2 Immuno-PRISM study has shown significant superiority of teclistamab when compared with lenalidomide and dexamethasone in patients with high risk smoldering myeloma. Overall response rate and MRD negativity was seen in 100% of patients and this also included CR in patients with high risk FISH.
If CAR T and BsAb therapies both gain frontline approval, sequencing will be crucial, guided by resistance mechanisms from prior treatments to maximize remission.(Jurgens E et al., 2026, Dima D et al., 2025)
Bispecific Antibodies vs CAR-T: Key Considerations
Although CAR-T is a one-time therapy, the production time of a minimum of three weeks, along with the need for GMP-grade facilities and trained personnel, makes its manufacturing both time-consuming and costly.
Off-the-shelf availability of BsAbs offers superior availability and reliability for patients who are rapidly progressing. While rates of CRS and neurotoxicity appear to be lower with BsAbs than with CAR T cells, morbidity from infection is high, and novel pathways of treatment resistance arise from the longitudinal selection pressure of chronic BsAb therapy.
Despite exhibiting somewhat lower efficacy compared with CAR-T therapy, bispecific antibody, with its procedural simplicity and reduced side effect profile, could be a more viable option for patients with compromised baseline health and patients who are not candidates for CAR-T treatment.
The significant difference in CR rates between CAR-T therapies and BsAbs can be attributed to differences in treatment mechanisms, including the persistence and potency of CAR-T cells versus the chronic administration and reliance on endogenous T cells in BsAbs. These factors, combined with biological differences in the tumor microenvironment, help explain the observed variance in efficacy.
You Can Watch More on OncoDaily Youtube TV
Written by Susanna Mikayelyan MD


