Breast cancer did not enter the ADC era gradually. It lurched into it. What once looked like a refined drug-delivery idea has become one of the most disruptive treatment shifts in modern oncology. A 2024 review in Cancer Discovery described 11 FDA-approved antibody-drug conjugates and more than 210 in clinical development, but breast oncology felt that rise in a particularly dramatic way because ADCs did more than add drugs. They redrew treatment pathways, changed how biomarkers are interpreted, and pushed clinicians to think differently about resistance, sequencing, and even what defines a targetable disease state (Colombo et al., 2024).
If you want to understand how breast cancer treatment was rewritten over the past decade, four ADCs tell that story better than anything else: trastuzumab emtansine, trastuzumab deruxtecan, sacituzumab govitecan, and datopotamab deruxtecan. They did not all change practice with the same force or in the same setting. But together they mark the moment when ADCs stopped being “promising” and became central to how metastatic and early breast cancer are treated.
One made ADCs real. One broke the HER2 binary. One made TROP2 matter. And one showed that the future is no longer about whether ADCs belong in breast cancer, but how clinicians are supposed to choose among them once several do (Verma et al., 2012; Bardia et al., 2021; Colombo et al., 2024).
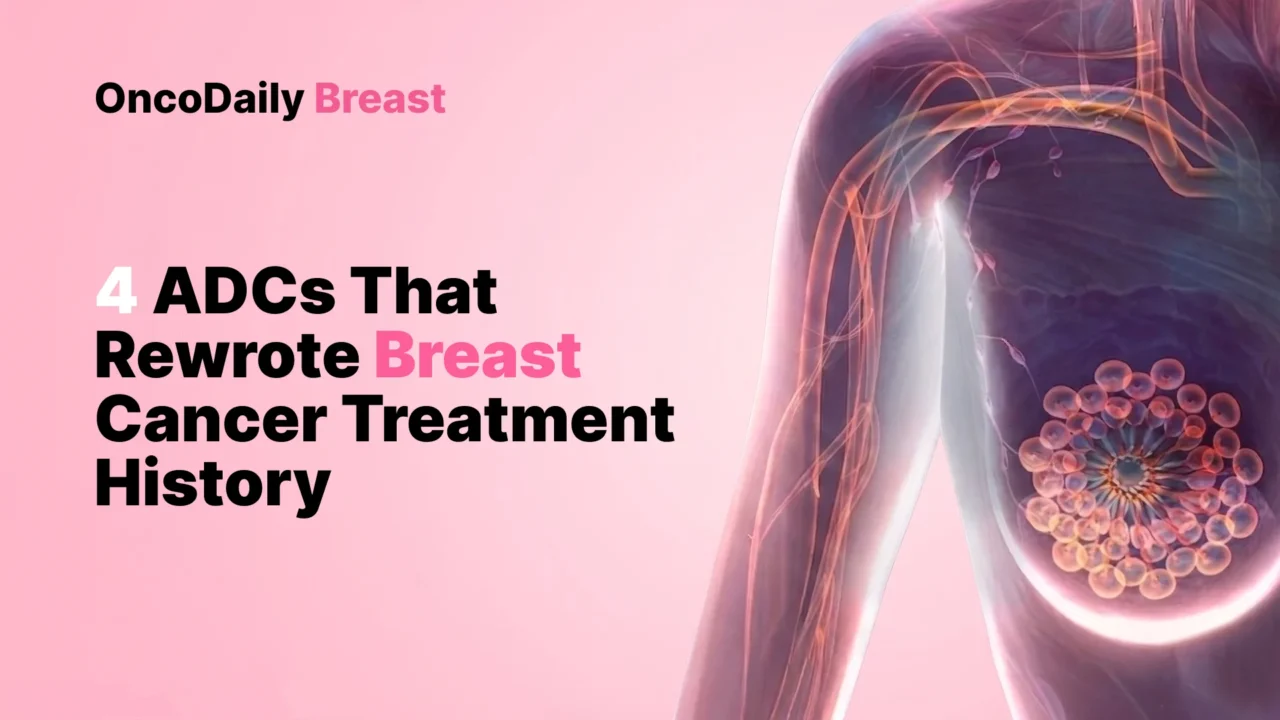
Trastuzumab Emtansine Was The Drug That Made ADCs Real
Before T-DM1, ADCs in breast cancer still carried a faint sense of theory. They were elegant on paper, interesting in development, but not yet strong enough to claim that they had changed the standard. EMILIA changed that. In previously treated HER2-positive advanced breast cancer, trastuzumab emtansine improved median progression-free survival to 9.6 months compared with 6.4 months for lapatinib plus capecitabine, and median overall survival to 30.9 months compared with 25.1 months. Those numbers mattered not only because they were statistically important, but because they proved that an ADC could beat an established HER2-directed regimen in a major metastatic setting. That was the first real moment the class stopped feeling experimental and started feeling inevitable (Verma et al., 2012).
T-DM1 also mattered because of how it worked. It belongs to an earlier ADC generation: a HER2-directed antibody carrying a microtubule-inhibitor payload through a non-cleavable linker. That sounds technical, but historically it matters. T-DM1 showed that targeted delivery of cytotoxic therapy could be clinically superior even without the now-famous topoisomerase I payloads and cleavable-linker strategies that dominate later ADCs. It was the bridge between the trastuzumab era and the modern ADC era. In hindsight, many of the later ADC successes look bigger and more dramatic, but without T-DM1 the class might never have had enough credibility to expand as fast as it did (Verma et al., 2012; Colombo et al., 2024).
Its second historic role came in early HER2-positive disease. KATHERINE had already changed post-neoadjuvant management, but the long-term follow-up made the magnitude of that change impossible to dismiss. Seven-year invasive disease-free survival was 80.8% with adjuvant T-DM1 versus 67.1% with trastuzumab, and seven-year overall survival was 89.1% versus 84.4%. That means T-DM1 did not merely create a new option after residual disease. It changed the meaning of residual disease itself. Residual HER2-positive disease after neoadjuvant therapy became a clear point for escalation, and that remains one of the most important standard-of-care shifts in contemporary early breast cancer (Geyer et al., 2025).
Trastuzumab Deruxtecan Was The Drug That Broke The HER2 Rules
If T-DM1 made ADCs real, T-DXd made them impossible to contain. Its first major disruption came in HER2-positive metastatic breast cancer, where DESTINY-Breast03 showed that it was not just better than T-DM1, but dramatically better. The long-term survival analysis reported median overall survival of 52.6 months with trastuzumab deruxtecan versus 42.7 months with trastuzumab emtansine, and median progression-free survival of 29.0 months versus 7.2 months. That result did not just update second-line therapy. It dethroned the prior ADC standard and replaced it with a much stronger one (Cortés et al., 2024).
But even that was not the biggest thing T-DXd did. The real historical rupture came when it made HER2-low breast cancer clinically actionable. For years, the field treated HER2 as a binary. A tumor was HER2-positive or it was not. DESTINY-Breast04 shattered that logic. In previously treated HER2-low metastatic breast cancer, trastuzumab deruxtecan produced significantly longer progression-free and overall survival than physician’s-choice chemotherapy. That was not just a positive trial. It was a conceptual shift in the biology of breast cancer treatment. The therapeutic map was suddenly no longer divided neatly into HER2-positive and HER2-negative disease. There was a new middle ground, and it mattered (Modi et al., 2022).
That conceptual break widened even further in 2025. The FDA approved trastuzumab deruxtecan for unresectable or metastatic HR-positive HER2-low or HER2-ultralow breast cancer after progression on endocrine therapy, based on DESTINY-Breast06. In that study, median progression-free survival in the HER2-low population was 13.2 months with T-DXd versus 8.1 months with chemotherapy.
The approval was also notable because it formally named HER2-ultralow disease in the label, making it the first approval to specify that subgroup. In practical terms, that means T-DXd did not only create a new treatment line. It changed the language of pathology and expanded the reach of HER2-directed therapy into patients who, a few years earlier, would have been grouped as simply HER2-negative (U.S. Food and Drug Administration, 2025a).
And T-DXd was still not finished. In December 2025, the FDA approved trastuzumab deruxtecan with pertuzumab for the first-line treatment of unresectable or metastatic HER2-positive breast cancer. The approval was based on DESTINY-Breast09, where median progression-free survival was 40.7 months with T-DXd plus pertuzumab versus 26.9 months with taxane-trastuzumab-pertuzumab, and confirmed objective response rates were 87% versus 81%. That is the kind of result that does not merely tweak sequencing. It threatens to reset the front line.
So T-DXd has now done something few breast cancer drugs ever accomplish: it changed second-line HER2-positive treatment, created HER2-low and HER2-ultralow as real therapeutic categories, and then pushed into first-line HER2-positive metastatic care as well (Tolaney et al., 2025; U.S. Food and Drug Administration, 2025b).

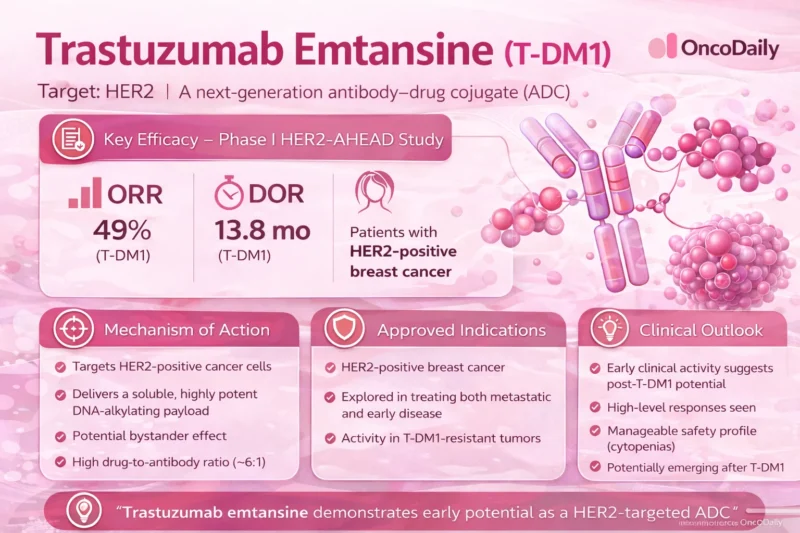
Sacituzumab Govitecan Was The Drug That Made TROP2 Matter
Sacituzumab govitecan changed history in a different corner of breast oncology, one where progress has often been harder to secure. In metastatic triple-negative breast cancer, ASCENT showed that sacituzumab govitecan improved median progression-free survival to 4.8 months versus 1.7 months with chemotherapy and median overall survival to 11.8 months versus 6.9 months. In a disease long associated with rapid progression and disappointing post-chemotherapy durability, that was a true change in expectation. It meant that an ADC could produce a meaningful survival gain in metastatic TNBC, not just a response-rate signal or a short-lived benefit (Bardia et al., 2021).
But the impact of sacituzumab govitecan was larger than TNBC alone. What it really did was establish TROP2 as a clinically serious target in breast cancer. Before SG, TROP2 was biologically interesting but not central. After ASCENT, it became a therapeutic axis the field had to take seriously. Mechanistically, SG also helped define the newer ADC generation: a TROP2-directed antibody, a cleavable linker, and a topoisomerase I inhibitor payload. That matters because it pushed the class away from the first-generation design of T-DM1 and toward the more aggressive bystander-effect logic that later ADCs would continue to exploit (Colombo et al., 2024).
Its reach then extended beyond TNBC. In 2023, the FDA approved sacituzumab govitecan for HR-positive, HER2-negative unresectable or metastatic breast cancer after endocrine therapy and at least two additional systemic therapies in the metastatic setting. In TROPiCS-02, median progression-free survival was 5.5 months versus 4.0 months with single-agent chemotherapy, and median overall survival was 14.4 months versus 11.2 months. That was important not only because it added another option in heavily pretreated HR-positive disease, but because it proved that TROP2 ADC benefit was not confined to triple-negative biology. Sacituzumab govitecan helped move ADC therapy into a broader part of metastatic breast oncology (U.S. Food and Drug Administration, 2023).
Sacituzumab govitecan also helped create one of the most pressing problems of the current ADC era: sequencing. Once SG became standard in TNBC and then relevant in HR-positive/HER2-negative disease, the field had to confront a more difficult question. What happens when one topoisomerase I–based ADC is followed by another? Are these drugs complementary, partially redundant, or cross-resistant? The modern ADC era became more powerful because of sacituzumab govitecan, but also more complicated. That complication is part of its historical importance. It did not simply improve outcomes. It forced clinicians to start thinking at the class level, not just at the single-drug level (Colombo et al., 2024).
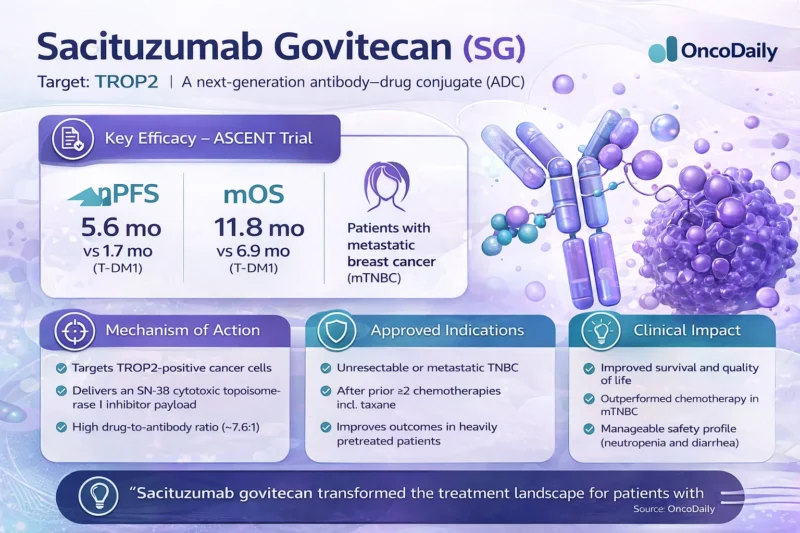
Datopotamab Deruxtecan Opened The Next ADC Chapter
Datopotamab deruxtecan is the newest of the four, but that is exactly why it belongs in this story. Its January 2025 FDA approval for unresectable or metastatic HR-positive, HER2-negative breast cancer followed TROPION-Breast01, where median progression-free survival was 6.9 months with Dato-DXd versus 4.9 months with chemotherapy. By itself, that is a meaningful approval. But historically, Dato-DXd matters for a different reason: it marked the moment when breast oncology stopped asking whether TROP2 ADCs belong and started asking how more than one TROP2 ADC should coexist in the same treatment space (U.S. Food and Drug Administration, 2025c; Royce et al., 2025).
That transition is more significant than it first appears. When Dato-DXd arrived, clinicians already had sacituzumab govitecan in the broader TROP2 conversation. That immediately raised new questions about toxicity, payload overlap, prior ADC exposure, and whether the class could be sequenced rationally. The field was no longer celebrating a lone breakthrough. It was now facing a crowded ADC battlefield. Dato-DXd therefore belongs in the history of breast cancer treatment not because it single-handedly changed every standard, but because it made clear that the ADC era has matured. The difficult question now is not whether ADCs matter. It is how to choose among them once several matter at once (Colombo et al., 2024; Royce et al., 2025).
It also sharpened another modern reality: not every ADC success looks the same. Some approvals are backed by dramatic survival gains. Others are built on progression-free survival improvements with more nuanced overall survival data. That distinction matters clinically, because it means the field can no longer treat “ADC” as a single category with predictable value. Dato-DXd helped expose that complexity. It arrived not as the drug that launched the class, but as the drug that proved the class is now broad enough to require internal comparison, strategy, and restraint (Royce et al., 2025; Colombo et al., 2024).
The Bonus Lesson They Left Behind
The biggest lesson from these four drugs is not that ADCs improve progression-free survival, raise response rates, or extend overall survival, although they do all of those things. Their deeper impact is that they changed definitions. T-DM1 changed what to do with residual HER2-positive disease after neoadjuvant therapy. T-DXd changed what HER2-negative means by making HER2-low and HER2-ultralow therapeutically actionable. Sacituzumab govitecan changed what TROP2 means by turning it into a survival-relevant target across more than one subtype. Datopotamab deruxtecan changed what ADC success means by proving that the class has become crowded enough for real competition inside it (Geyer et al., 2025; U.S. Food and Drug Administration, 2025a; U.S. Food and Drug Administration, 2025c).
That success, however, has created new problems. The same Cancer Discovery review that celebrates the rise of ADCs also emphasizes the next frontier: toxicity, sequencing, and biomarker selection. ADC toxicities still often resemble the payloads they carry more than the elegance of their targeting strategy. Predictive biomarkers remain weak or inconsistent. And sequencing is increasingly messy as more agents use overlapping targets or payload classes. In other words, breast oncology is now rich in ADCs but still poor in precise rules for who should receive which one first, and why. That is the real bonus lesson. These drugs did not merely solve old problems. They created smarter, harder new ones (Colombo et al., 2024).
What These Four Drugs Actually Changed
The most important thing these ADCs changed is not a single trial result. It is the logic of breast cancer treatment itself. After them, HER2 is no longer a simple yes-or-no variable. Post-neoadjuvant escalation in HER2-positive disease is no longer optional when residual invasive disease remains.
TROP2 is no longer a side target discussed mostly in translational circles. And ADC selection is no longer just a matter of whether a drug works, but which target, which linker, which payload, and which prior exposure matter most for a specific patient. These four drugs did not merely rewrite standards. They rewrote the architecture of decision-making in breast cancer (Verma et al., 2012; Modi et al., 2022; Bardia et al., 2021; Colombo et al., 2024).
That is why they belong in breast cancer history. T-DM1 proved ADCs could be a standard. T-DXd turned HER2 from a binary into a spectrum. Sacituzumab govitecan gave TROP2 survival relevance. Datopotamab deruxtecan showed that the ADC era has already become too big for one-drug thinking. Taken together, they did not just extend old maps. They forced the field to draw new ones. And that is the clearest sign that they changed treatment history for good (Tolaney et al., 2025; U.S. Food and Drug Administration, 2025a; U.S. Food and Drug Administration, 2025c; Colombo et al., 2024).