For more than a decade, first-line treatment in HER2-positive metastatic breast cancer has opened the same way. A taxane. Trastuzumab. Pertuzumab. THP was not just a regimen. It was the regimen. It survived years of progress in later-line therapy and remained the anchor of frontline care because nothing had yet delivered a result strong enough to truly displace it.
DESTINY-Breast09 may have changed that.
In this phase III trial, trastuzumab deruxtecan plus pertuzumab did not simply perform well. It produced the kind of result that forces a field to stop, recalculate, and ask whether the standard it trusted for years is still the right one. Median progression-free survival rose from 26.9 months with THP to 40.7 months with trastuzumab deruxtecan plus pertuzumab. The hazard ratio for progression or death was 0.56, translating to a 44% reduction in risk. Response rates were higher. Complete responses were more frequent. Responses lasted far longer. In a disease where the first-line decision shapes everything that follows, this is not a routine positive study. It is a major frontline challenge to the THP era (Tolaney et al., 2026).
That is why DESTINY-Breast09 matters so much. It is not only about another active HER2-directed drug. It is about whether the most powerful antibody-drug conjugate in breast cancer is ready to move from second line into first line, and whether the center of gravity in HER2-positive metastatic care is about to shift.
Why This Trial Feels Bigger Than Most Positive Studies
HER2-positive breast cancer accounts for approximately 15% to 20% of breast cancers. The development of HER2-targeted therapy transformed what was once one of the most aggressive biologic subtypes into one of the clearest success stories in modern oncology. Yet metastatic disease remains incurable for most patients, and sequence still matters. Not every patient reaches second-line therapy. Not every patient remains fit enough to receive the most active treatments once the disease progresses. In real-world practice, attrition between lines is not theoretical. It is common enough to shape outcomes (Cottu et al., 2025; Lam et al., 2025).
That is what gives first-line trials such weight. A better regimen in later lines is important. A better regimen in the very first line may change the whole treatment arc.
Trastuzumab deruxtecan had already transformed the second-line setting. DESTINY-Breast03 showed that T-DXd was superior to trastuzumab emtansine, and that result was strong enough to rewrite the post-THP sequence almost immediately (Cortés et al., 2022; Hurvitz et al., 2023). The natural next question was unavoidable: if T-DXd is more effective in second line, should it be moved earlier? DESTINY-Breast09 was the trial built to answer that question.
Copyright © 2025 Massachusetts Medical Society.
The Trial Asked the Right Frontline Question
DESTINY-Breast09 was a global, randomized, phase III study in patients with HER2-positive advanced or metastatic breast cancer who had received no prior chemotherapy or HER2-directed therapy for metastatic disease. Patients were eligible even if they had received prior neoadjuvant or adjuvant treatment, as long as the disease-free interval exceeded 6 months. One prior line of endocrine monotherapy in the metastatic setting was allowed.
Patients were randomized in a 1:1:1 ratio to one of three groups: trastuzumab deruxtecan plus pertuzumab, trastuzumab deruxtecan plus placebo, or THP. For this prespecified interim analysis, the reported comparison was between trastuzumab deruxtecan plus pertuzumab and THP. The T-DXd plus placebo arm remains blinded until the final progression-free survival analysis, which means one of the most important unanswered questions in the study is still pending: whether pertuzumab is necessary in all patients once trastuzumab deruxtecan is brought into first-line care (Tolaney et al., 2026).
That uncertainty matters. But it does not weaken the headline result. The comparison that needed to be won was the one against THP, and that is the comparison DESTINY-Breast09 won decisively.
This Was a Real-World Frontline Population
One reason the study feels so immediately relevant is that the patient population was broad and recognizable.
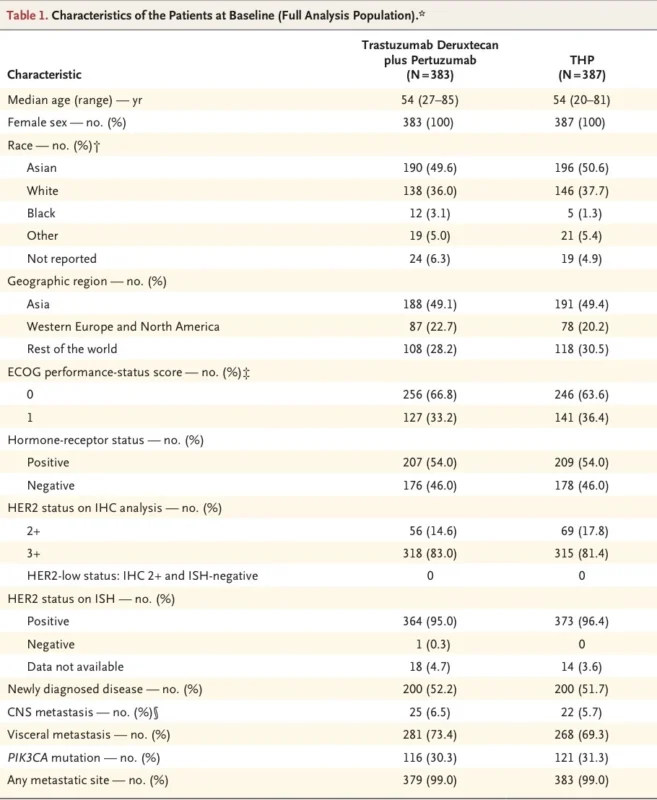
Among the patients included in the current analysis, 383 received trastuzumab deruxtecan plus pertuzumab and 387 received THP. The median age was 54 years, with a range from 20 to 85. About 52% had newly diagnosed metastatic disease, 54% had hormone receptor-positive tumors, and 30.8% had PIK3CA mutations. Roughly 70% to 73% had visceral metastases, and approximately 6% had brain metastases at baseline. This was not a heavily filtered trial population made artificially favorable by design. It looked like the modern HER2-positive metastatic clinic (Tolaney et al., 2026).
Previous treatment exposure also reflected real practice. Around 43% had received prior neoadjuvant or adjuvant therapy. Approximately 28% had prior trastuzumab exposure, and just over 7% had previously received pertuzumab in the curative-intent setting. That matters because this is increasingly the patient population being seen in daily practice: not only de novo metastatic disease, but also relapse after modern early-stage HER2-directed treatment.
The Progression-Free Survival Result Was Impossible to Ignore
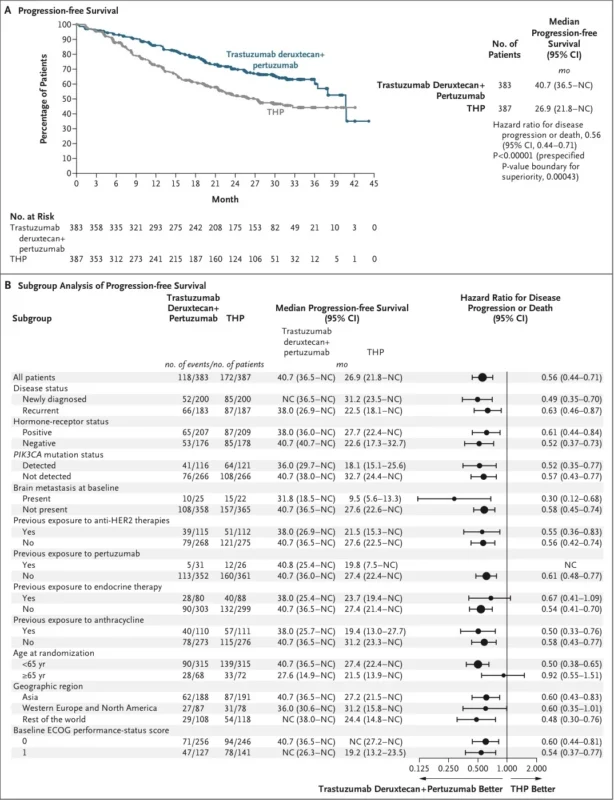
At a median follow-up of 29.2 months, the primary endpoint was clearly positive.
Median progression-free survival, assessed by blinded independent central review, was:
- 40.7 months with trastuzumab deruxtecan plus pertuzumab
- 26.9 months with THP
The hazard ratio for progression or death was 0.56 with a 95% confidence interval of 0.44 to 0.71. The superiority boundary was crossed with P<0.00001. At 24 months, an estimated 70.1% of patients in the T-DXd plus pertuzumab group remained free of progression or death, compared with 52.1% of patients in the THP arm (Tolaney et al., 2026).
That is not the kind of PFS gain that lives only in a Kaplan–Meier curve. It is clinically loud. It is visible. It is hard to dismiss.
And it is especially striking because THP is an excellent control arm. CLEOPATRA established THP as one of the most successful standards in metastatic breast cancer, with durable benefit and long survival follow-up (Baselga et al., 2012; Swain et al., 2020). DESTINY-Breast09 did not beat an obsolete regimen. It beat a frontline standard that had defined the field for years.
Copyright © 2025 Massachusetts Medical Society.
The Depth of Response Strengthened the Story
DESTINY-Breast09 was not just about delaying progression. It also showed a deeper antitumor effect.
The confirmed objective response rate was 85.1% with trastuzumab deruxtecan plus pertuzumab compared with 78.6% with THP. More importantly, complete responses occurred in 15.1% of patients receiving the T-DXd combination and 8.5% of those receiving THP. Median duration of response was 39.2 months versus 26.4 months, respectively (Tolaney et al., 2026).
That pattern matters because it reinforces that the T-DXd regimen is not only postponing radiographic progression through marginal disease control. It is producing more profound and more durable responses. In frontline metastatic care, that matters for symptoms, organ preservation, treatment confidence, and the broader trajectory of the disease.
The Benefit Was Broad, Not Confined to a Niche Subgroup
One of the first questions clinicians ask when a trial is this positive is whether the benefit was driven by one especially favorable group. DESTINY-Breast09 does not appear to support that kind of narrowing.
The progression-free survival benefit favored trastuzumab deruxtecan plus pertuzumab across most major subgroups, including patients with newly diagnosed and recurrent metastatic disease, hormone receptor-positive and hormone receptor-negative tumors, patients with and without PIK3CA mutations, and those with and without prior anti-HER2 exposure in the early-stage setting. Even the small subgroup with brain metastases at baseline showed a favorable signal, although the numbers there were limited and should be interpreted carefully (Tolaney et al., 2026).
That consistency is one reason this trial will likely change practice quickly. Clinicians do not need to squint at the data and search for the one subgroup in which the result truly matters. The signal looks broad enough to force a frontline conversation in most patients with HER2-positive metastatic disease.
The Safety Story Is Good, but Not Comfortable
Now comes the harder part.
The efficacy results are strong enough to generate excitement. The safety profile is strong enough to demand discipline.
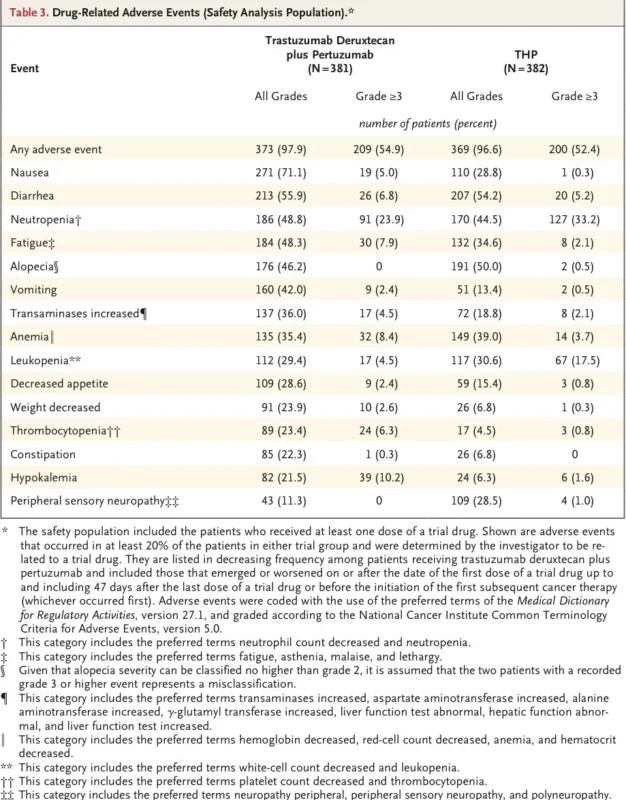
Grade 3 or higher adverse events occurred in 63.5% of patients treated with trastuzumab deruxtecan plus pertuzumab and 62.3% of patients treated with THP. Serious adverse events occurred in 27.0% and 25.1%, respectively. Adverse events leading to discontinuation of any trial treatment occurred in 20.7% with the T-DXd combination and 28.3% with THP (Tolaney et al., 2026).
So at the highest level, the global burden of major toxicity was not dramatically different between arms. But the type of toxicity was different, and that difference is clinically meaningful.
With trastuzumab deruxtecan plus pertuzumab, the most common drug-related adverse events included nausea, diarrhea, fatigue, alopecia, vomiting, transaminase elevation, anemia, thrombocytopenia, and hypokalemia. With THP, the expected taxane-heavy pattern remained prominent, including neutropenia, leukopenia, diarrhea, and peripheral neuropathy (Tolaney et al., 2026).
The adverse event that continues to define trastuzumab deruxtecan, however, is interstitial lung disease or pneumonitis. In DESTINY-Breast09, adjudicated drug-related ILD or pneumonitis occurred in 12.1% of patients in the trastuzumab deruxtecan plus pertuzumab arm. Most events were grade 1 or 2, but there were 2 grade 5 events, meaning fatal toxicity. In the THP arm, ILD or pneumonitis occurred in 1.0%, and all cases were grade 1 or 2 (Tolaney et al., 2026).
This cannot be softened. T-DXd is highly effective, but ILD remains a real and potentially lethal risk. That risk is not a reason to avoid the drug in every setting. It is a reason to use it with vigilance, experience, and strict monitoring.
Why the Trial May Change the Sequence of HER2-Positive Disease
DESTINY-Breast09 is not only a positive first-line study. It is a sequence-changing study.
For years, HER2-positive metastatic breast cancer has been managed with a fairly stable order. THP first. T-DXd second. T-DM1 historically before that, and then newer agents layered later. DESTINY-Breast09 now raises the possibility that the sequence itself has to be rebuilt around the most potent drug much earlier in the course of disease.
That would be a major shift.
Moving T-DXd into first line does not only affect the THP standard. It affects what second line becomes, whether pertuzumab is needed beyond T-DXd, how endocrine therapy combinations are used in hormone receptor-positive disease, and how clinicians think about CNS control in HER2-positive metastatic care. The implications go well beyond one regimen. They touch the full architecture of HER2-directed sequencing.
This is also happening within a broader movement in breast oncology, where antibody-drug conjugates are steadily moving upstream. DESTINY-Breast09 is one of the clearest examples yet of that shift.
What the Trial Still Does Not Tell Us
For all the excitement, several important questions remain open.
The T-DXd plus placebo arm remains blinded, so the field still does not know whether trastuzumab deruxtecan alone may be sufficient for some patients. That question matters because de-intensifying treatment while preserving efficacy would be enormously valuable in metastatic care.
Overall survival is also not yet mature. The progression-free survival benefit is large enough to be practice-relevant, but in HER2-positive metastatic breast cancer, where multiple effective later-line therapies exist, overall survival remains a crucial endpoint.
The intracranial efficacy story is also still developing. The subgroup signal in patients with baseline brain metastases is encouraging but small. Given how clinically important CNS disease is in HER2-positive metastatic breast cancer, future analyses will be closely watched.
And finally, the generalizability of the trial, while broad, is not perfect. The study authors note that Black patients were underrepresented, which remains an important limitation in interpreting global metastatic breast cancer data (Tolaney et al., 2026).
Copyright © 2025 Massachusetts Medical Society.
The Bottom Line
DESTINY-Breast09 is one of the most important HER2-positive breast cancer trials in years.
Trastuzumab deruxtecan plus pertuzumab extended median progression-free survival to 40.7 months, increased the confirmed objective response rate to 85.1%, nearly doubled complete responses, and prolonged median duration of response to 39.2 months. Those are not incremental gains. They are the kind of gains that force reconsideration of an entrenched frontline standard (Tolaney et al., 2026).
The caution is real. Interstitial lung disease remains the defining toxicity. Fatal cases still occurred. Overall survival is immature. The monotherapy arm remains blinded. But none of that changes the central fact that this trial delivered a major efficacy result in the exact setting where the field had the most to gain from one.
THP built the frontline era of HER2-positive metastatic care.
DESTINY-Breast09 may be the trial that ends it.

Read About CLEOPATRA Trial on OncoDaily


