Arndt Vogel, Head of the Center for Personalized Medicine, MHH at Medical University of Hanover, shared on X about a recent paper by Van K. Morris et al. published on Cancer Cell:
“Phase 1/2 trial of encorafenib, cetuximab, and nivolumab in MSS BRAF V600E mCRC
ORR 42%, mPFS 7.2 mo, mOS 22mo
Extracellular vesicle and tumor RNA profiling to identify immune signatures
BRAF+EGFR+PD-1 shows some clinical efficacy > biomarker.”
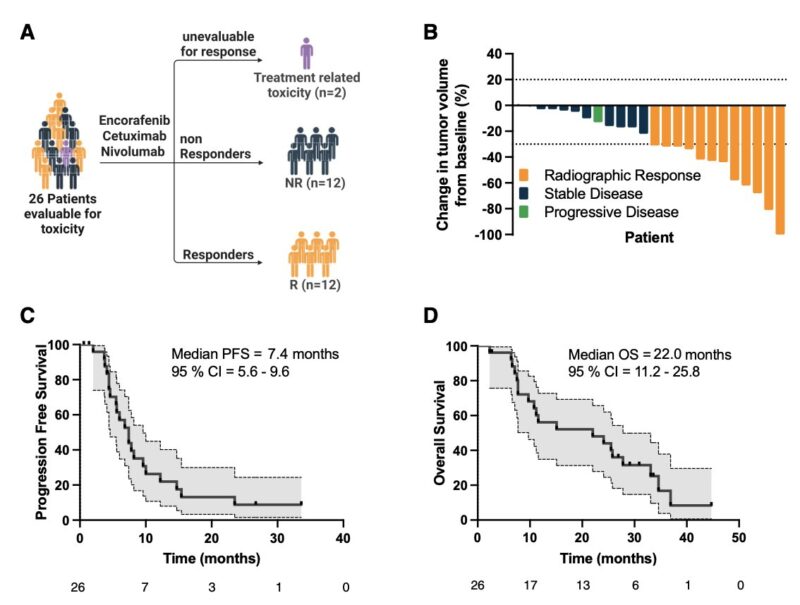
Title: Phase 1/2 trial of encorafenib, cetuximab, and nivolumab in microsatellite stable BRAFV600E metastatic colorectal cancer
Authors: Van K. Morris, Christine M. Parseghian, Vahid Bahrambeigi, Nourhan Abdelfattah, Lianchun Xiao, Anjali Agrawal, Kangyu Lin, Kanwal P.S. Raghav, Robert A. Wolff, Arvind Dasari, Ryan W. Huey, Bryan K. Kee, Michael J. Overman, Jason A. Willis, Phat H. Le, Michelle Escano, Yunyu C. Baig, Kelsey Pan, David Menter, Alda L. Tam, Wai C. Foo, Li Shen, Hey Min Lee, Thomas D. Gallup, Cori Margain, Dave Gallup, Kimal I. Rajapakshe, Paola A. Guerrero, Jing Wang, Ryan B. Corcoran, Anirban Maitra, Kyuson Yun, Scott Kopetz.

More posts featuring Arndt Vogel.


