What happens when chronic leukemia suddenly stops behaving like one, and it is no longer just progression?
Richter Transformation (RT) refers to the evolution of Chronic Lymphocytic Leukemia (CLL) into a far more aggressive malignancy.
While CLL may remain stable for years, RT is associated with rapid progression, systemic symptoms, treatment resistance and represents one of the most complex scenarios in modern hem-oncology. Consequently, RT is a major focus of ongoing research aimed at better understanding its underlying biology and improving patient outcomes.
The Quiet Complexity of CLL
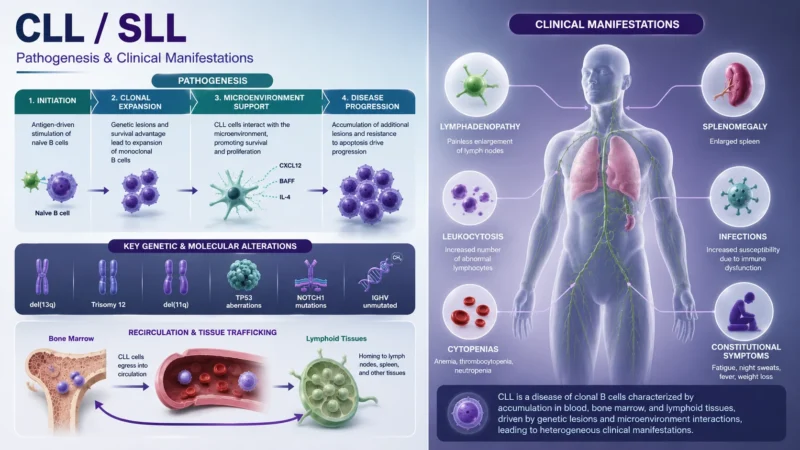
CLL is initiated by specific genomic alterations that interfere with the proliferation and apoptosis in clonal B‐cells. In the recent update of the SEER database, the age‐adjusted incidence was 4.6 per 100 000 inhabitants per year, making it the most frequent type of leukemia. The median age at diagnosis is 70 years, less than 10% are younger than 45 years. More male than female patients (1.9:1) are affected, and this gender effect seems to be stable across all ethnicities.
The diagnosis is established by blood counts, blood smears, and immunophenotyping of circulating B‐lymphocytes, which identify a clonal B‐cell population carrying the CD5 antigen as well as typical B‐cell markers.
CLL and Small Lymphocytic Lymphoma (SLL) are considered different clinical manifestations of the same mature B-cell neoplasm. The distinction mainly depends on the primary site of involvement. SLL predominantly affects lymph nodes and lymphoid tissues with minimal or absent peripheral blood involvement. The diagnosis should be confirmed by histopathological evaluation of a lymph node.
Two major clinical staging systems, Rai and Binet, provide prognostic stratification in CLL based on physical examination and blood counts. Several biological and genetic markers, in particular, chromosome 17p deletion and TP53 mutations are associated with shorter time to progression and poorer outcomes.
To integrate these variables, the CLL International Prognostic Index (CLL-IPI) combines clinical, biological, and genetic factors to classify distinct risk groups. Although the CLL-IPI remains prognostically relevant in the era of targeted agents, outcomes for patients with high-risk disease have substantially improved with modern therapies.
Timing of Therapy
Only patients with active or symptomatic disease, or those with advanced Binet or Rai stages, require therapy. When indicated, current standards include monotherapy with BTK inhibitors or venetoclax-based combinations such as venetoclax-obinutuzumab, venetoclax-ibrutinib.
At relapse, the initial treatment may be repeated if the treatment-free interval exceeds 3 years. If relapse occurs earlier, an alternative regimen should be chosen based on prior exposure and patient fitness. Options may include rituximab/lenalidomide-containing regimens, alternative targeted agents, non-covalent BTK inhibitors, PROTACs, cellular therapies, or enrollment in clinical trials.
Across studies in relapsed CLL, combinations involving targeted agents have consistently demonstrated superior efficacy compared with conventional chemoimmunotherapy, including uMRD achievement. Therapeutic options remain limited for patients with disease refractory to both BTK and BCL2 inhibitors, making this double-refractory setting particularly challenging.
Another critical turning point arises when a subset of patients develop RT: CLL evolves into a rapidly proliferative high-grade lymphoma, most commonly Diffuse Large B-Cell Lymphoma (DLBCL), with occasional cases of Hodgkin lymphoma or rarer variants like B-lymphoblastic lymphoma and mantle cell lymphoma. RT occurs in 2-15% of patients with an annual incidence of 0.5-1%.
What Is Known About RT?
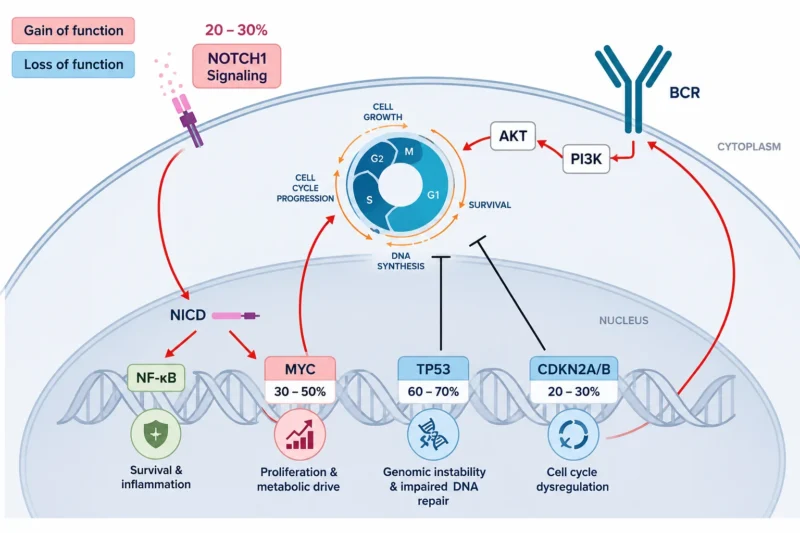
Modern studies increasingly describe RT as both a genetic and immunologic transformation process rather than a purely histologic event. Molecular analyses have identified early “seeding” subclones years before overt RT develops, suggesting that it isn’t a sudden stochastic “switch”, but rather the culmination of prolonged clonal evolution under selective pressure from both the immune microenvironment and prior therapies.
TP53 and NOTCH1 mutations have been repeatedly associated with increased transformation risk. BCR signaling, often mediated through unmutated or stereotyped IGHV receptors, activates downstream PI3K-AKT and NF-κB pathways. Concurrent loss of tumor-suppressor checkpoints (TP53 alteration, CDKN2A/B deletion), removes apoptotic and cell-cycle control. These pathways converge on MYC, promoting metabolic reprogramming and rapid proliferation. Together, chronic antigenic stimulation and cumulative genetic damage drive instability, and ultimately the emergence of RT clones.
Hypothesized therapeutic pressure includes immune dysfunction, and microenvironmental changes. Prolonged BTK inhibition may promote outgrowth of TP53-mutated clones, while venetoclax may preferentially spare apoptosis-resistant ones.
Morphologically, RT-DLBCL mostly exhibits an activated B-cell or non-germinal center phenotype, consistent with its dependence on chronic active BCR signaling. Many cases retain CLL immunophenotypic features. Compared with de novo DLBCL, RT more frequently expresses PD-1/PD-L1, facilitating immune evasion.
Clinically, transformation is marked by rapidly enlarging lymph nodes, constitutional B symptoms, high LDH levels, extranodal disease and sudden clinical deterioration. Unlike the conventional CLL with relatively slow kinetics, RT requires urgent confirmation and treatment escalation.
Is RT Always Related to the Original CLL Clone?
Approximately 80% of RT-DLBCL cases are clonally related, retaining the same IGHV rearrangements as the CLL and reflecting the stepwise acquisition of high-risk lesions. Less commonly, it is an unrelated lymphoma arising independently from the pre-existing CLL. This distinction is clinically important because clonally related RT carries worse prognosis than clonally unrelated disease.
Why Such Significance ?
One of the key reasons RT is considered so important is that secondary transformation usually confers a poorer prognosis than de novo aggressive disease. In other words, a DLBCL arising through RT is more difficult to treat than DLBCL developing independently from the outset.
Emerging trials such as STELLAR now stratify patients distinguishing between treatment-naïve and previously treated populations. This trial is scheduled for completion in 2027 and reflects a broader shift toward risk-adapted approaches in RT.
Current Options and the Search for More Effective Therapies
Historically, RT has been treated similarly to de novo DLBCL, using conventional CIT such as R-CHOP. More intensive regimens, including hyper-CVAD, platinum-based combinations, and dose-adjusted R-EPOCH, have produced higher response rates in some studies, reaching up to 67%, however, remissions are often short-lived, particularly in patients with complex karyotypes.
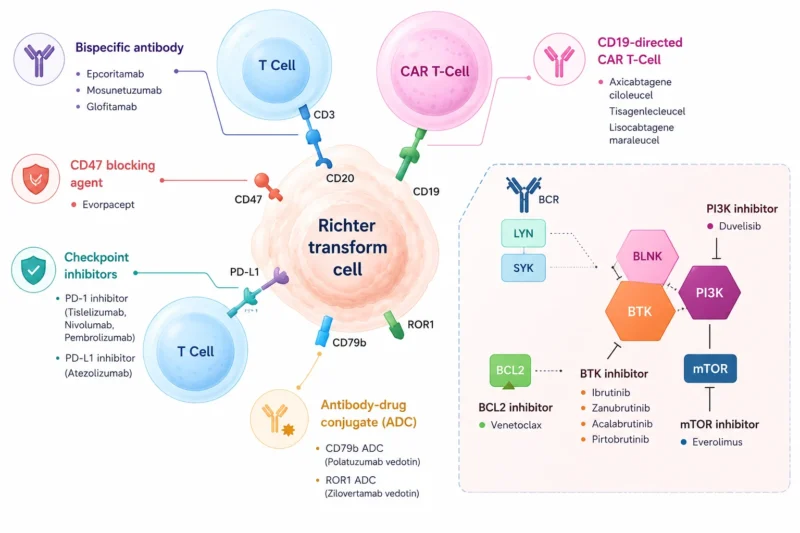
BTKi have shown limited efficacy as monotherapy in RT. Zanubrutinib combined with tislelizumab improved ORR to 58%, while the non-covalent BTKi pirtobrutinib demonstrated an ORR of 50% yet median PFS remained short.
Venetoclax-based combinations have demonstrated greater activity, particularly when combined with CIT. Chemotherapy-free approaches have also shown efficacy, the MOLTO trial combining venetoclax, obinutuzumab, and atezolizumab reported an ORR of 67.9% and a CR rate of 28.6%.
Although most evidence comes from retrospective or early-phase studies, venetoclax-based combinations currently represent one of the most promising strategies in RT, particularly as a bridge to stem cell transplantation or cellular therapies.
Allo-HSCT remains the only potentially curative therapy for RT and is generally reserved for fit patients who achieve remission after induction. While MRD negativity pre-allo-HSCT correlates with superior outcomes in aggressive lymphoma cohorts, specific data in RT are lacking.
Patients achieving complete metabolic remission on PET/CT demonstrate the best survival. Retrospective studies report 3-year OS rates of 40-60%, supporting a clinically meaningful graft-versus-lymphoma effect. In contrast, autologous transplantation is associated with limited long-term benefit.
Allo-HSCT is feasible in only a minority of patients because RT often occurs in older, heavily pretreated individuals with significant comorbidities. Though reduced-intensity conditioning regimens have expanded eligibility, many patients never reach transplant.
Where Ongoing Research Is Heading
Although RT patients were excluded from pivotal CAR T-cell trials in DLBCL, multiple retrospective studies suggest anti-CD19 CAR T-cell therapy may be a promising option in this setting. Across international cohorts, ORRs of 57-65% and CR rates of 46-50% have been reported.
Toxicities were generally manageable and comparable to those observed in aggressive B-cell lymphomas, though long-term neurocognitive and psychosocial complications were relevant. Importantly, it can also eradicate residual underlying CLL in a substantial proportion of patients. Durability remains a major limitation. Median PFS is only approximately 4-5 months, and median OS generally remains below 10 months, indicating a high risk of early relapse.
Bispecific T-cell engagers show potential in RT, given the T-cell exhaustion commonly observed in CLL. By simultaneously targeting CD3 on T-cells and CD20 on malignant B-cells, these agents redirect endogenous cytotoxic immunity without the need for individualized cell manufacturing. Among them, epcoritamab achieved an ORR of 50%, CR of 35%, and median PFS of 12.8 months in heavily pretreated RT patients, with several patients achieving MRD-negative remission.
mdpi.com
Mosunetuzumab and glofitamab, have also demonstrated meaningful activity, with manageable CRS and limited neurotoxicity. Though response rates remain variable, these therapies appear capable of inducing durable remissions in patients refractory to CIT or ineligible for CAR T therapy.
Phase II trial is evaluating the addition of polatuzumab vedotin (ADC) to R-CHOP based on the frequent expression of CD79b in RT cells. Early evidence suggests that combining PD-1 blockade with BTK inhibition may be more effective than checkpoint inhibition alone.
Current research is focused on CAR T-cell combinations with BTKis, BsAbs, checkpoint inhibitors, alongside development of “armored” CAR Ts engineered to resist immune exhaustion. Macrophage checkpoint inhibitors targeting CD47 and next-generation engineered cellular therapies are being actively explored.
Trials are also evaluating time-limited triplet regimens: pirtobrutinib, venetoclax and obinutuzumab, as well as investigational BTKi-based triplets incorporating mTOR inhibitors or immunomodulatory agents. Toxicity and trial feasibility remain limitations in this rare disease.
Finally, several novel molecular targets are under investigation. ROR1-directed therapies such as zilovertamab vedotin have shown activity in related B-cell malignancies and may extend into RT. Approaches targeting MYC, BCL6, CDK9, and MCL1-related resistance pathways reflect a broader shift toward biomarker-driven and biologically tailored therapies in RT. At the clinical level, trials should integrate treatments targeting both the aggressive lymphoma component and the underlying CLL.
You can also read: BCR::ABL1-Rearranged Chronic Myeloid Leukemia: Molecular Basis, TKI Resistance and Emerging Therapies
Written by Susanna Mikayelyan, MD
FAQ
Why is Richter transformation biologically different from ordinary CLL progression?
Unlike gradual progression of Chronic Lymphocytic Leukemia, RT involves acquisition of aggressive molecular features such as TP53 disruption, MYC activation, and complex karyotypic changes. The disease no longer behaves like an indolent leukemia and instead resembles high-grade lymphoma biology.
Could Richter transformation happen without prior CLL treatment?
Yes. Although prior therapy and selective therapeutic pressure may contribute to clonal evolution, Richter Transformation can also occur in treatment-naïve patients. This suggests that intrinsic disease biology, genetic instability, and microenvironmental interactions play central roles independent of therapy exposure.
Can PET/CT distinguish Richter transformation from active CLL?
PET/CT may strongly raise suspicion for RT, particularly when lymph nodes demonstrate high FDG uptake, but imaging alone cannot confirm the diagnosis. Histopathologic biopsy remains essential because aggressive symptoms and radiographic progression may also occur in advanced CLL without transformation.
Why does clonally related Richter transformation carry a worse prognosis?
Clonally related RT evolves directly from the original CLL clone through cumulative genetic damage and therapy resistance mechanisms. These tumors often retain the molecular complexity and immune evasion patterns of heavily evolved CLL, making them more difficult to eradicate than clonally unrelated lymphomas.
Why is Richter transformation such an important area for CAR T-cell research?
RT combines aggressive lymphoma biology with profound immune dysfunction from underlying CLL, creating a setting where conventional therapies often fail. CAR-T Cell Therapy offers the possibility of targeting both the transformed lymphoma and residual CLL simultaneously.
Could bispecific antibodies become more practical than CAR T-cell therapy in RT?
Possibly. Unlike CAR T-cell therapy, bispecific antibodies do not require individualized cell manufacturing and can often be administered more rapidly. This may be particularly valuable in RT, where disease acceleration frequently demands urgent treatment initiation.