Acute Lymphoblastic Leukemia (ALL) presents one of the clearest contrasts in hem-oncology. It is among the most curable cancers in children often considered one of oncology’s major therapeutic achievements. In adults, however, ALL is biologically more complex, less treatment-tolerant, and associated with poorer outcomes. Understanding why these differences exist reveals how age, genetics, and treatment strategy can reshape the course of a single leukemia.
What Is ALL?
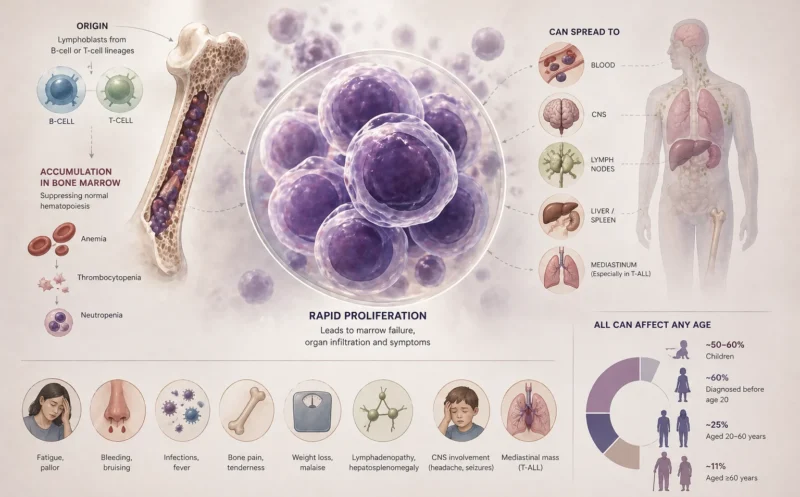
ALL is a rapidly progressing cancer characterized by the uncontrolled proliferation of lymphoblasts. Originating from B-cell or T-cell lineages, these cells lose the ability to mature into functional lymphocytes while simultaneously acquiring a survival and proliferative advantage. They accumulate within the bone marrow, suppress normal hematopoiesis, and may spread into the bloodstream and other organs.
This explains many of the classic manifestations of ALL:
- Anemia, presenting with fatigue, pallor, dizziness, reduced exercise tolerance, palpitations, or dyspnea.
- Thrombocytopenia manifesting as easy bruising, petechiae, epistaxis, gingival bleeding, or prolonged bleeding after minor trauma.
- Neutropenia and/or functionally ineffective leukocytes, with increased susceptibility to infections, slower recovery, recurrent fever, and oral ulcers.
- Bone pain or tenderness caused by marrow expansion and leukemic infiltration, involving the long bones, spine, or joints, and sometimes producing gait disturbance or limping in children.
- Constitutional symptoms such as persistent fever, night sweats, unintentional weight loss, anorexia and malaise.
- Lymphadenopathy, hepatomegaly, or splenomegaly, which may present as enlarged lymph nodes, abdominal discomfort, early satiety, or left upper quadrant pain.
- CNS involvement in a subset of patients, presenting with headache, vomiting, cranial neuropathies, visual disturbances, seizures, or altered mental status.
Mediastinal involvement, more common in T-lineage ALL, which may cause cough, chest discomfort, dyspnea, or symptoms of superior vena cava compression.
Patients with mature B-cell ALL (Burkitt’s disease) may present with massive adenopathy, particularly in the abdomen, and are at high risk of tumor lysis syndrome and acute kidney injury even before treatment initiation.
Some patients with Burkitt-like ALL may develop numbness of the chin, lower lip, and buccal or gingival mucosa due to sensory neuropathy caused by mandibular marrow expansion affecting the trigeminal nerve.
Approximately 50-60% of ALL cases occur in children, and about 60% are diagnosed before age 20. Only 25% of cases are diagnosed between ages 20-60, while adults aged 60 years and older account for ~11% of new diagnoses.
Biological Subtypes
It is broadly divided into:
- B-cell ALL (CD10 CD19 CD22 CD79a PAX5 are typical markers)
- T-cell ALL (CD2- CD8, TdT, CD34)
Modern classification further separates ALL according to cytogenetic and molecular lesions, such as:
- Philadelphia chromosome (BCR::ABL1-positive)
- Ph-like ALL
- ETV6::RUNX1 fusion
- KMT2A rearrangements
- IKZF1 alterations/deletions
- Hyperdiploidy/hypodiploidy
ETP-ALL is a distinct T-ALL subtype that frequently harbors lesions involving DNMT3A, FLT3, IDH1/2, shows few NOTCH1 mutations, and often carries activating alterations in cytokine receptor genes (Emadi A et al., 2026).
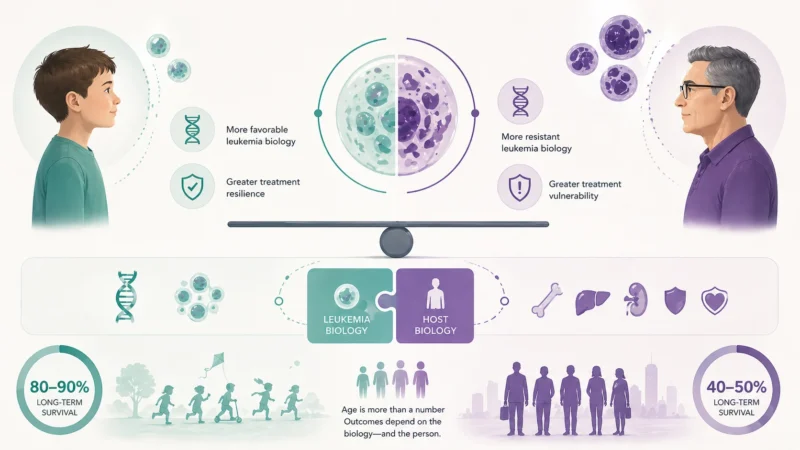
Why Age Is More Than a Number in ALL
In children, especially 1-10 years of age, ALL more often carries genetic subtypes associated with high treatment sensitivity, such as ETV6-RUNX1 fusion and high hyperdiploidy.
Adults have higher rates of Philadelphia chromosome + ALL, Ph-like ALL, IKZF1, TP53 alterations, complex cytogenetics, and cooperating therapy-resistant patterns. This means that adult ALL is not simply “childhood ALL in an older body”, it is often biologically distinct with its own patterns of resistance, vulnerability, and relapse risk.
Adult leukemias often demonstrate greater clonal diversity at diagnosis. Rather than a single dominant clone, multiple subclones may coexist, some intrinsically resistant to therapy. Under treatment pressure, resistant clones can expand, contributing to MRD persistence or relapse.
Age affects the patient’s capacity to tolerate treatment. Bone marrow reserve, hepatic metabolism, renal function, vascular integrity, and immune resilience generally decline over time, alongside higher rates of comorbidities. As a result, therapies tolerated in childhood may cause greater toxicity in adults, leading to dose reductions, treatment delays, or failure to deliver curative-intent therapy.
Thus, outcome differences arise from the interaction of two biologic systems:
1. Leukemia biology (more favorable vs more resistant disease)
2. Host biology (greater treatment resilience vs increased vulnerability)
Why Adults Lagged Behind
For decades, pediatric ALL programs were supported by large cooperative trial networks, highly standardized protocols, intensive supportive care systems, and continuous optimization. In contrast, adult ALL was historically often managed with less pediatric-inspired, less asparaginase-intensive regimens, largely limited by greater toxicity concerns.
Treatment, which relies on multiagent chemotherapy administered over 2-3 years, results in long-term survival of 80-90% in pediatric patients compared with 40-50% in adult patients. Modern ALL management increasingly depends less on chronological age alone and more on physiologic fitness, genomic risk, and therapeutic tolerance.
The AYA Interface in ALL
The AYA group lies between these worlds, both biologically and clinically. Multiple studies have suggested that pediatric-inspired regimens (PIR) achieve higher survival for AYAs than historical regimens commonly used for older adults (non-PIR).
PIR are characterized by incorporation of substantial asparaginase in combination with vincristine, corticosteroids, moderate cumulative doses of anthracyclines and cyclophosphamide, a prolonged maintenance phase using continuous thiopurine, and intermittent intrathecal methotrexate throughout treatment.
According to a 2026 retrospective longitudinal cohort study by David R. Freyer et al., survival for PIR vs non-PIR was 98.1% vs 88.3% at 1 year, 88.5% vs 69.1% at 3 years, and 87.3% vs 63.3% at 5 years, while the non-PIR group showed an early decline within the first year.
The CALGB 10403 major international trial provides an important benchmark for T-ALL outcomes, demonstrating that 295 patients aged 16–39 years treated with a pediatric-inspired regimen achieved 3-year overall survival and event-free survival rates of 73%.
Collectively, these findings have helped shape expert recommendations favoring pediatric-inspired therapy for Ph- ALL in AYAs (Azza E et al., 2025).
How Adult ALL Therapy Is Evolving
Modern adult ALL therapy is progressively narrowing the survival gap. Key advances include:
1.Pediatric-inspired regimens in younger fit patients
2.TKIs for Ph+ ALL
3. Blinatumomab
4. Inotuzumab ozogamicin
5. CAR-T Cell Therapy
6. Stem cell transplantation in some high-risk patients
7. MRD-guided intensification or de-escalation
The TKI Era
The combination of a TKI with a backbone of multiagent chemotherapy (often Hyper-CVAD), or more recently blinatumomab, remains the mainstay of therapy for Ph+ B-cell ALL. Regardless of the backbone used, emphasis should be placed on continuous TKI use.
The UKALLXII trial showed that adding Imatinib to intensive chemotherapy improved CR rate (92% vs 82%), 4-year OS (38% vs 22%), and the proportion of patients proceeding to allogeneic SCT (71% vs 50%), with greater benefit when imatinib was introduced during induction.
The EWALL-PH-01 trial evaluated a lower-intensity regimen combining Dasatinib with vincristine and dexamethasone in newly diagnosed patients aged ≥55 years. Complete remission was achieved in 96% of patients, and the 5-year OS rate was 36%. Among tested relapses, 75% harbored the T315I mutation.
Ponatinib has potent activity against Ph+ leukemias with both wild-type and mutated ABL1 kinase domains, including the T315I mutation. The multicenter phase 3 randomized PhALLCON trial showed that Ponatinib achieved higher MRD-negativity at the end of induction than Imatinib (34% vs 17%), as well as higher deep MRR (42% vs 21%).
These findings suggest that some patients who achieve sustained responses may not require allogeneic transplant. Asciminib, which acts through the ABL myristoyl pocket rather than the ATP-binding site, and Olverembatinib have both demonstrated promising early activity, with asciminib achieving a 3-month MR4 rate of 26% and a 2-year OS of 75%.
In newly diagnosed Ph-negative ALL, nearly half of patients deemed MRD-negative by MFC were still MRD-positive by NGS. In Ph+ ALL, persistent low-level BCR::ABL1 transcripts despite NGS negativity may not always represent true residual lymphoblastic disease, emphasizing the need for assay-specific interpretation.
You Can Watch More on OncoDaily TV
Further Advances
Blinatumomab, a BiTE targeting CD3 and CD19, has shown activity in both Ph− and Ph+ ALL. In 45 heavily pretreated patients with Ph+ ALL, 36% achieved a response, median OS was 7.1 months. Because continuous IV infusion is cumbersome, a subcutaneous formulation is under investigation.
Overall, a chemotherapy-free approach with Blinatumomab plus Ponatinib has improved outcomes, achieving high CMR rates, reducing toxicity. However, omission of high-dose chemotherapy has been associated with increased CNS relapse risk, prompting greater use of IT therapy and reconsideration of CNS-penetrating regimens in high-risk patients.
Ph-like ALL is associated with GATA3 polymorphism. More than 50% of cases harbor CRLF2 rearrangement or overexpression, and activating JAK mutations, which are associated with poorer prognosis.
Blinatumomab and Inotuzumab ozogamicin (an anti-CD22 ADC carrying calicheamicin) have shown efficacy in Ph-like ALL in the phase 3 TOWER and INO-VATE trials, potentially overcoming the impact of these alterations. Their incorporation into frontline therapy may further improve outcomes. Early phase 1 data with Venetoclax plus chemo are also promising.
InO’s major toxicity is sinusoidal obstruction syndrome (SOS), particularly in patients proceeding to allogeneic SCT or receiving dual-alkylator conditioning. Fractionated weekly dosing reduced SOS risk without compromising efficacy.
Early anti-CD19 CAR T-cell successes with tisagenlecleucel and brexucabtagene autoleucel demonstrated high remission and MRD-negativity rates in heavily pretreated children and adults, establishing cellular therapy as a major salvage option. Newer platforms such as Obecabtagene autoleucel aim to reduce toxicity while preserving efficacy, but relapse remains common, often through CD19 loss.
Patients treated with low-burden or MRD-only disease show better survival and lower rates of severe CRS/ICANS. These findings support earlier use in MRD-positive or other high-risk settings (Kantarjian H et al., Ramos Peñafiel et al., 2025).
What Future Strategies May Bring
Future progress is likely to come from earlier integration, dual-target constructs, and personalized use of allo-SCT, rather than as a routine default:
1. The incorporation of multiple available antibodies into one regimen, including newer formulations.
2. Less chemotherapy and a shorter overall treatment duration.
3. The replacement of chemotherapy with targeted agents (blinatumomab and ponatinib in Ph-positive ALL, and blinatumomab and menin inhibitors in KMT2A-rearranged ALL).
4. The use of sequential CAR T-cell therapies in patients with positive MRD or high-risk disease to hopefully replace allogeneic SCT in this setting.
Where a Child Is Treated Still Influences ALL Survival
Although the survival of children and adolescents with ALL has significantly improved in high-income countries, patients in low-income countries still face poor prognoses, often due to delayed diagnosis, treatment abandonment, limited supportive care, shortages of essential medicines, and reduced access to molecular testing or stem cell transplantation.
According to an analysis of global trends by Ding F et al. (2025), approximate 5-year survival rates for childhood ALL are 85-95% in high-income countries (HICs), 60-85% in upper-middle-income countries (UMICs), 35-70% in lower-middle-income countries (LMICs), and often below 50%, sometimes under 20% – in low-income countries (LICs).
A child with ALL in a low-resource setting may have worse outcomes than an adult treated in a modern tertiary center, showing that the survival contrast can also operate in the reverse direction and is shaped not only by age, but also by the healthcare system into which that patient is diagnosed.
You can also read: Acute Myeloid Leukemia (AML) in 2026: The Current Landscape and Future Perspectives
Written by Susanna Mikayelyan, MD
FAQ
Why ALL behaves so differently in children and adults?
Although ALL carries the same name across age groups, the underlying genetics, clonal complexity, treatment tolerance, and relapse biology can differ substantially. In many ways, age changes both the leukemia and the host treating it.
Why is ALL considered one of pediatric oncology’s success stories?
Childhood ALL became a landmark success through decades of cooperative trials, risk-adapted therapy, strong supportive care, and continuous protocol refinement. Cure rates in many high-income countries now exceed those seen in most adult populations.
Why does ALL sometimes spread beyond the bone marrow?
Lymphoblasts can circulate through the bloodstream and infiltrate other tissues such as the central nervous system, lymph nodes, liver, spleen, or mediastinum, creating symptoms beyond low blood counts alone.
What makes adult ALL harder to treat than childhood ALL?
Adults more often present with high-risk subtypes such as Philadelphia chromosome-positive or Ph-like ALL, and may have lower tolerance for intensive chemotherapy. Comorbidities and organ reserve also become more important with age.
Why are adolescents and young adults (AYAs) a unique group in ALL?
Adolescents and young adults often fall between pediatric and adult treatment systems. Many studies suggest they may benefit from pediatric-inspired regimens while still requiring adult-specific toxicity management.
Can modern therapies close the survival gap between adults and children?
The gap is narrowing. Advances such as tyrosine kinase inhibitors, Blinatumomab, Inotuzumab ozogamicin, CAR-T Cell Therapy, and MRD-guided strategies have significantly improved adult outcomes.
What is MRD, and why do leukemia specialists care so much about it?
MRD (minimal residual disease) refers to tiny amounts of leukemia that remain after treatment. It is one of the strongest predictors of relapse and increasingly guides decisions about intensification, transplantation, or therapy de-escalation.
Does every patient with ALL need a stem cell transplant?
No. While transplantation remains important for some high-risk patients, modern targeted therapy and deeper molecular remissions may allow selected patients to avoid routine allogeneic transplant.
What may future ALL treatment look like?
Future strategies may include less chemotherapy, more targeted combinations, dual-target cellular therapies, earlier immunotherapy use, and increasingly personalized decisions based on genetics and MRD rather than age alone.



