Paul Schwartz, Physician at Bergonié Institute, shared a post on LinkedIn:
“American Society of Clinical Oncology (ASCO) GU26 – Updates from The Prostate Cancer Working Group 4 (PCWG4)
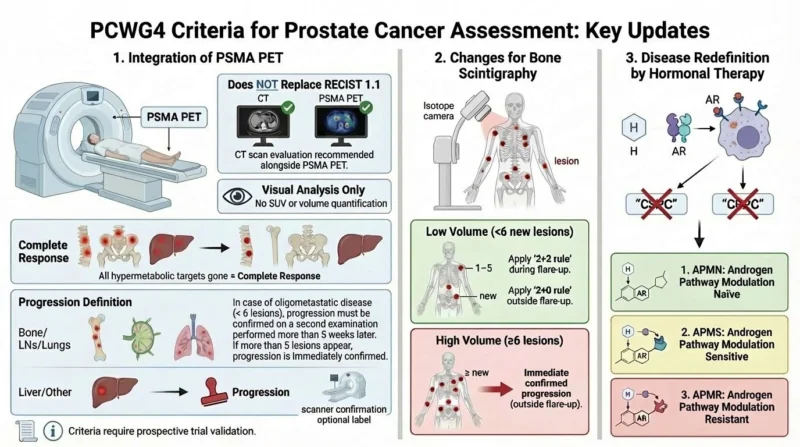
1. Integration of PSMA PET Imaging
- Does Not Replace RECIST 1.1 Evaluation: A CT scan remains recommended for disease assessment, either conducted simultaneously or separately from the PSMA PET scan.
- Visual Analysis Only: Changes in SUV (Standardised Uptake Value) or volume are not validated for response evaluation.
- Definition of Complete Response: Complete response is defined as the disappearance of all hyperfixing targets.
- Definition of Progression:
* Bone, Lymph Nodes or Lungs: In cases of oligometastatic disease (fewer than six lesions), progression must be confirmed by a second examination conducted more than five weeks later. If more than five lesions appear, progression is confirmed immediately.
* Liver or Non-Pulmonary Metastases: The appearance of a new lesion (ideally confirmed by CT) indicates progression.
- Evaluation Criteria: These criteria for complete response or progression are formalised by PCWG4 and require future validation in prospective clinical trials.
2. Changes for Bone Scintigraphy
- Low Volume Disease: For the appearance of fewer than six lesions, the 2 + 2 rule (during flare-up) or the 2 + 0 rule (outside of flare-up) is maintained.
- High Volume Disease: If more than five lesions appear (outside of flare-up), progression is confirmed immediately.
3. Redefinition of Disease in Relation to Hormonal Treatments
- Abandonment of Terms: The terms ‘castration-sensitive prostate cancer’ (CSPC) and ‘castration-resistant prostate cancer’ (CRPC) are no longer used.
- New Definitions Based on ADT/ARPI Exposure:
* Androgen Pathway Modulation Naïve (APMN)
* Androgen Pathway Modulation Sensitive (APMS)
* Androgen Pathway Modulation Resistant (APMR).”