The ASCO GU 2026 took place from February 26 to 28 in Moscone West, San Francisco. The program centered on the theme of “Patient-Centered Care: From Discovery to Delivery” and was dedicated to translate the latest research breakthroughs into improved clinical practices and patient outcomes.
Here are 15 interesting and informational posts that you shouldn’t miss from different professionals that visited and participated in the ASCO GU 2026.
15 Posts Not To Miss From ASCO GU 2026
“Again another long list of poster presentations GU26 American Society of Clinical Oncology (ASCO) by the Ospedale San Raffaele GU Oncology stars. They did it again in San Francisco, see you in 2027!”

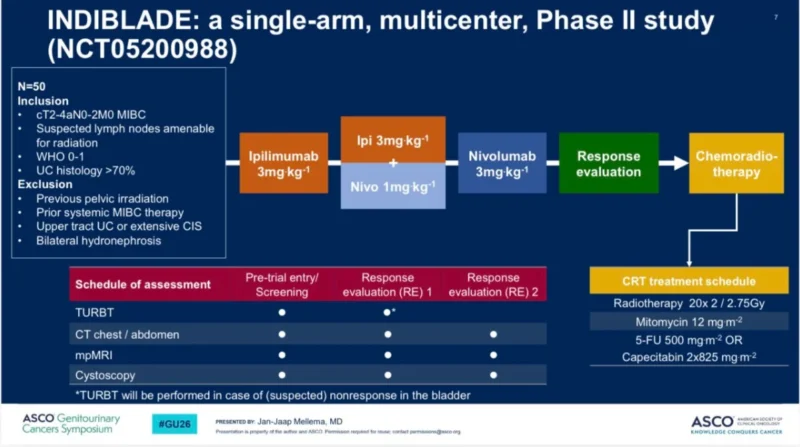
“The INDIBLADE Trial (n=50): Neoadjuvant Ipi/nivo then Chemoradiotherapy in muscle invasive bladder cancer shows 2 yr Bladder-Intact EFS of 78% and 2 yr OS of 96%. than previous chemo/rad trials. Only 39% ctDNA+ve at baseline suggests a lower risk population compared to NIAGARA (57%). CtDNA clearance with IPI/Nivo=76% is (gem/cis/durva=70%). IPI/nivo/TMT looks good. VOLGA is the last chance to answer the CTLA4 question. Outcome in MIBC is strongly influenced by the presence on MRD at baseline (2 yr DFS for IPI/nivo surgery was ~70%).”
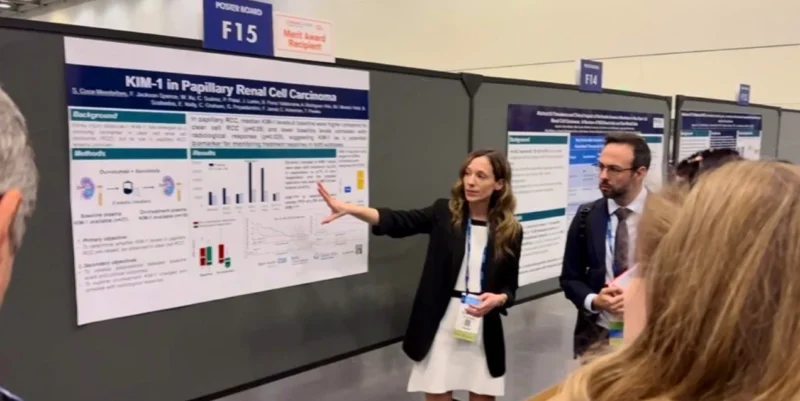
GU26 In pRCC (n=31), baseline KIM-1 was significantly vs ccRCC (7800 vs 5400) and baseline KIM-1 was associated with radiological response (p=0.025) These data support its potential prognostic value although standardization and validation are needed.
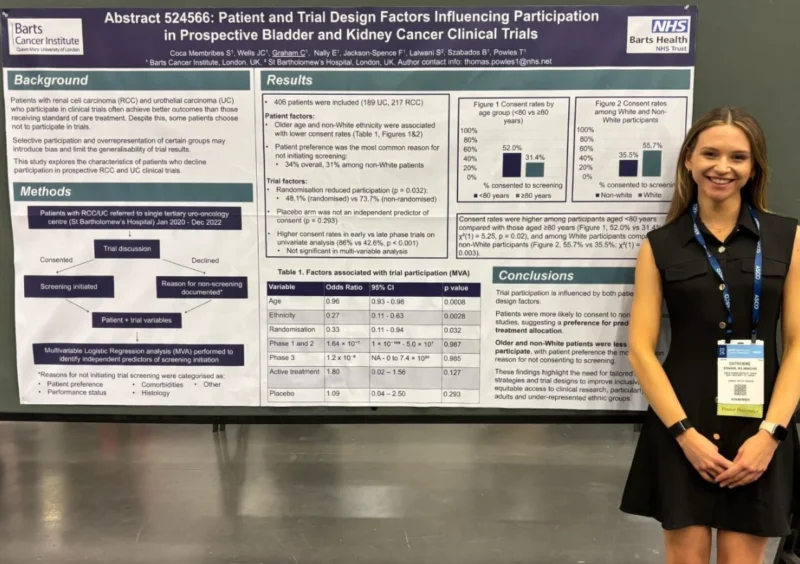
“Had the pleasure of meeting Tom Powles’ fellow Catherine Graham MD at the GU26 poster sessions. Really interesting work around characteristics driving clinical trial screening; something my group is quite interested in as well. Hats off to Tom for terrific mentoring – he is a beacon of support to investigators in our community!”
“Clinical trial populations do not always reflect real-world patients. In kidney and bladder cancer trials, older age, non-White ethnicity and randomisation reduced screening. Inclusive trial design and patient engagement are essential to improve equity and generalisability – by Catherine Graham.”
“Always fantastic to see Cristiane D Bergerot and Paulo G Bergerot. Congratulations for all the great work that you do and Happy Birthday to the little princess!”
“Always great catching up with Petros Grivas, Paulo Bergerot at ASCO GU26
Grateful for the collaboration, friendship, and shared commitment to better outcomes for our patients.”

“Beyond proud of this team. Watching our team grow and present at ASCO GU26 is the most rewarding part of this work. Grateful for each one of you.”
“11 abstracts. 1 amazing team. Memories for life. So proud to be part of Beacon Riazlab presenting at ASCO GU’26. None of this would be possible without our mentors Irbaz Riaz, Zakharia Yousef, Parminder Singh. Thank you for believing in us.”

“A sincere thank you to the American Society of Clinical Oncology (ASCO) for the invitation to speak as part of a multidisciplinary panel at the 2026 ASCO Genitourinary Cancers Symposium, GU26, on multidisciplinary approaches to bladder preservation, highlighting the role of imaging, including VI-RADS MRI, in clinical management. Thank you and congratulations to our excellent co-moderators, Drs. Leslie Ballas and Pooja Ghatalia and to my co-presenters, Drs. Marie-Pier St. Laurent and Andrea Necchi and congrats to our session abstract presenter, Dr. Jan-Jaap Mellema.”

“GU26 provided several noteworthy readouts for ProstateCancer.
PEACE3 — Enza + Radium-223 (mCRPC, bone-dominant)
- Design: Ph 3, enza +/- 6 cycles Ra-223; ARPI-naive, no visceral disease
- Results: Final OS 38.2 vs 32.6 mo (HR 0.76); rPFS HR 0.71.
- Takeaway: OS is noteworthy but fits narrow CRPC population in ARPI era. Radium is probably overlooked with all our novel options.
CAPItello-281 — Capivasertib + Abiraterone (PTEN-deficient mHSPC)
- Design: Ph 3, abi + ADT +/- capi; PTEN-deficient by IHC (n=1012)
- Results: rPFS 33.2 vs 25.7 mo (HR 0.81); AKT AEs present but manageable, QOL slightly worse early then similar.
- Takeaway: PTEN is a bad marker and intensifcation is needed. Need clarity in depth of loss, 100% looks most relevant.
BRCAAway — Abi + Olaparib vs monotherapy (1L BRCA-altered mCRPC)
- Design: Ph 2, abi vs ola vs combination; BRCA1/2 or ATM-altered, ARPI-naive.
- Results: mOS 68 mo (combo) vs 37 mo (ola) vs 28 mo (abi). Sequential therapy did not catch up.
- Takeaway: BRCA patients should get both PARP and ARPI at some point. Still questions around 1L combo.
PSMAddition — Lu-PSMA-617 + ADT + ARPI (PSMA+ mHSPC, PRO update)
- Results: No clinically meaningful worsening in HRQoL or pain; G3+ AEs slightly higher (50.7% vs 43%)
- Takeaway: Reassuring QOL data for frontline RLT. More efficacy data still needed before changing mHSPC SOC.
PEACE2 — ADT + RT +/- Cabazitaxel (Very high-risk local PCa)
- Design: Ph 3, 3 years ADT + RT +/- cabazi (n=761).
- Results: No improvement in cPFS, bPFS, MFS, or PCSS but more tox; 9-yr OS ~90% both arms.
- Takeaway: Chemo doesn’t help here. Abiraterone remains the only proven intensification strategy. Good news: even very high-risk disease has excellent long-term outcomes.
GUNS — Genomic Umbrella Neoadjuvant Study (High-risk localized PCa)
- Design: Biomarker-based cohorts; ARPI doublet vs triplet with docetaxel based on genomics.
- Results: MRD rate higher with triplet in TP53, PTEN, RB1, AKT-altered cancers (39% vs 0%, p=0.016)
- Takeaway: Genomics likely impact chemo-sensitivity, supports NGS being part of docetaxel mHSPC decision.
POSEIDON — ADT with post-op RT for PSA-recurrent PCa (Meta-analysis)
- Design: Individual patient meta-analysis, 6 RCTs (n=3938); RT +/- ADT in PSA-recurrence post-RP.
- Results: ADT w/ minimal OS benefit overall; benefit suggested only w/ PSA >1; potential harm at PSA <0.2; short-term ADT sufficient if given.
- Takeaway: Common clinical decision. We’re probably treating too many of these men with ADT, but PSA isn’t only relevant factor.
Great podium talks from Andrew Armstrong on PCWG4 and Daniel George on CAPItello-281, representing our Duke Cancer Institute team. The Keynote highlighted AI impacts now on daily practice and expedited timelines to the next generation of better treatments for patients.”

“Progress in cancer care is built in moments of shared curiosity.
Dinner meeting with Oliver Sartor my VISION co-PI and pioneer of PSMA radioligand therapy, discussing how Lutetium-177 is becoming the Model T of radioligand therapy — the first scalable platform for precision systemic radiation in prostate cancer.
Walking back, we ran into Dr. Jonathan Rosenberg my EV-301 co-PI and global leader in antibody–drug conjugate development, he discussed Enfortumab vedotin, KEYNOTE-B15, and the convergence of targeted payload therapies.
Not different drugs.
Different chapters of the same scientific story.
Radioligands.
Antibody–drug conjugates.
Precision targeting is reshaping cancer therapy.
The future of oncology will be written in better precision development — and in the patients who benefit from it!”

“We are proud to present at ASCO GU 2026 our work “Profiling BCRAness phenotype in public prostate cancer datasets without homologous repair gene mutations”, providing evidence for better molecular stratification in prostate cancer.”
“Great overview by Pavlos Msaouel at GU26 on clinical trial design. Highlighting the work of Think Tank group at IKCS!”

“Being in ASCO GU26 in San Francisco and being able to contribute, even if it is with a small grain of sand, to the knowledge of bladder cancer is a real privilege.
On this occasion we present an analysis of almost 4,000 patients from the American database Flatiron. What we found was relevant: approximately 28% of the patients had comorbidities that would make it difficult to receive standard treatment with enfortumab vedotin + pembrolizumab. This is not just a statistic. It means a worse prognosis, a higher risk of progression and, above all, the real need to think about therapeutic alternatives with different tolerability profiles.
Large trials change clinical practice, but real life reminds us every day that not all patients fit into the “ideal patient” of studies. And that is where we have to continue researching, understanding and refining.
I confess that, after so many years, I still get nervous before each presentation. Even when it’s in poster format. I suppose that’s a good sign. It means that I still care.
This analysis has been a logistical challenge of bringing several pharmaceutical companies to an agreement, attracting resources and interpreting a huge amount of data.
And I was also extremely lucky to accompany my friend Félix Guerrero-Ramos MD PhD FEBU in his oral presentation. Seeing the room full and seeing how far it is going filled me with pride, not only professionally, but personally. How wonderful to see friends grow like this.
Thank you to all of you who make it possible for us to continue moving forward. The cáncerdevejiga needs science, but also equipment.”

More posts about ASCO on OncoDaily.


