The AACR Annual Meeting 2026 is taking place from April 17 to 22, 2026, at the San Diego Convention Center in San Diego, California, bringing together the global cancer research community for a comprehensive exchange of scientific knowledge and innovation. Recognized as the focal point of the cancer research community, the meeting convenes scientists, clinicians, healthcare professionals, survivors, patients, and advocates to share the latest advances in cancer science and medicine.
Here are key highlights you shouldn’t miss, featuring important insights, research developments, and perspectives shared during the AACR Annual Meeting 2026:
Stacey Tinianov, Executive Director at Advocates for Collaborative Education:
“Celebrated my birthday with a trip to American Society of Clinical Oncology (ASCO) headquarters to participate in the Spring ASCO Guidelines Evidence-Based Medicine Committee (EBMC) meeting. While we meet virtually every month to review and approve the rapidly growing library of ASCO guidelines, nothing compares to the conversastions that happen in the room with these multidisciplinary rockstars.
Nearly thirteen years after my diagnosis, it is not lost on me that I am able to continue to celebrate birthdays thanks to the dedication of clinicians and researchers like these individuals.
Our purpose may sound simple: Provide the best guidance based on the evidence.
But the execution of such an aim is not so simple.
Guidelines aren’t just about treating cancer – they are supportive guides for the treatment of a whole person. And the conversations in the room showcase that this group understands that quality of life is personal and a key priority for many people with a diagnosis or history of cancer.
Grateful to be in the room. Grateful to be on the planet.
And now on to AACR26.”
 ”
”
Phebe Mawuena Afi Havor, Research Associate at Moffitt Cancer Center:
“Excited to be attending my First AACR Annual Meeting in San Diego!
Excited to learn, connect, and contribute at the intersection of cancer research and real-world impact — from translational science to clinical development and data analytics.
At AACR26, I’ll also be presenting my Moffitt Cancer Center PhD work, “ctDNA precedes imaging: A predictive model for real-time treatment adaptation in HPV-associated anal squamous cell carcinoma,” on 4/22 from 9:00 a.m.–12:00 p.m.
If you’re attending, do stop by Poster Section 2, Board 13 and say hello. Every conversation is an opportunity to learn — always glad to connect!”

Christine M. Lovly, Division Chief of Thoracic Medical Oncology at City of Hope:
“AACR26 starts today! What makes AACR so special is the brilliant, forward-thinking individuals whose dedication, passion, and vision continue to drive meaningful innovation. This photo represents a small but impactful group of the AACR membership – the Board of Directors.
Led by current President Lillian Siu, incoming President Keith Flaherty, and our truly remarkable CEO, Margaret Foti, this group is committed to pushing boundaries, solving complex challenges, and making lasting impact for all people living with cancer.”

“City of Hope is proud to lead and contribute to 103 sessions at the American Association for Cancer Research (AACR) Annual Meeting.
View or download our complete presentation guide, add our sessions to your schedule, and visit us at Booth 4421!
Anyi Li, Chief of Computer Service at Memorial Sloan Kettering Cancer Center:
“Everyone’s talking about AI in clinical trial screening. We actually built it — taking it from idea to real-world deployment in our clinic, and scaling.
Our AI-powered screening tool is now part of our oncologists’ daily routine. Physicians check it every day, and if they don’t see the trial matches they’re expecting, they come looking for them. That’s the kind of adoption we were hoping for.
Want to learn more about agentic AI in a clinical setting? Stop by Poster 1393 on Monday morning.
Huge congratulations to Jacob, who is receiving the AACR-Agilent Scholar-in-Training Award— recognizing outstanding young investigators — for his work on “Design of a prospective implementation study to evaluate the efficacy of an AI-assisted workflow intervention to increase breast cancer clinical trial participation.”
This wouldn’t have happened without a true team effort across physicians, researchers, engineers, research coordinators, and administration. Grateful to
work alongside all of you.”
“The AACR26 Annual Meeting is starting today!
Our Molecular Oncology lab at Johns Hopkins Kimmel Cancer Center will present 15 studies covering:
- a deep multi-omics dive in immunotherapy resistance (don’t miss the Immunotherapy clinical trials plenary session on Tuesday!)
- new computational methods for determining the origin of cfDNA variants, Jenna Canzoniero, congrats, Daniel Rabizadeh, for the Margaret Foti Scholar in training award!
- liquid biopsy-informed precision oncology clinical trials Amna Jamali Jenna Canzoniero Jessica Tao Labcorp
- novel ctDNA MRD technologies Blair Landon Paul Lee
- cfDNA fragmentation-enabled tumor fraction monitoring Noushin Niknafs
- integrative cfDNA and TCR compartment analyses to interpret immunotherapy response in rate tumors Jinny Huang
- clinical validity of landmark ctDNA molecular response in the immunotherapy setting Jaime Wehr
- comprehensive sequence and structural genomic landscapes of immunotherapy-treated mesothelioma Anna Nowak Patrick Forde Alistair Cook
- serial transcriptomic analyses to capture early changes in the evolving tumor microenvironment during immune checkpoint blockade Blair Landon Vincent Lam
- Hopkins thoracic experience on clinical utility of serial liquid biopsies for oncogene-driven lung cancer William Bowers Michael Conroy Joseph Murray Julie Brahmer
- expansion of blood biomarkers to choline metabolites and immunometabolism during neoadjuvant immunotherapy Rachel Keogh Rajesh Kumar NV Chi Van Dang
- epigenetic modulation as a means to sensitize immune cold tumors to immune checkpoint blockade Blair Landon
- comprehensive temporal and spatial assessment of the evolving T cell repertoire during therapy and after mixed responses to immune checkpoint blockade Asimina Zoitou
- all you have to know about ctDNA minimal residual disease, symposium organized by Viktor Adalsteinsson and featuring presentations by Aadel Chaudhuri Viktor and me.
Take a look at the schedule embedded in this post and our concurrent publications (a couple of links will not work until the time of presentation at the AACR meeting).
Hope to engage in forward-thinking conversations at or after these sessions at the AACR meeting. Stop by!”

“Here is our first onsite edition of AACR Annual Meeting News written by our Science Communications team previewing our American Association for Cancer Research Annual Meeting in San Diego with approximately 22,000 attendees.”

American Association for Cancer Research:
“The AACR Board of Directors is meeting today as we prepare for the start of AACR26 this afternoon. We are grateful to our Board members for their visionary leadership and their commitment to advancing our mission to prevent and cure all cancers.”

Jennifer Brain, PhD Candidate at Fred Hutch:
“AACR2026 kicks off this weekend! I’ve been fortunate to receive a Scholar-in-Training award to support my travel to San Diego. I’ll be presenting 2 posters about my favorite amino acid, cysteine – come check them out!
Constitutive NRF2 activation drives excess cysteine stress
Sunday April 19th, 2-5pm
Poster 537, Section 22, Board 3
Cysteine as a driver of resistance to Bortezomib (in collaboration w/ Matthew Vander Heiden MD, PhD)
Monday April 20th, 9am-12pm
Poster 1788, Section 16, Board 8.”
Ingo Hartung, Executive Director, Head of Medicinal Chemistry & Drug Design at Merck Healthcare:
“Only a few hours until AACR26 starts. As every year, the program is packed and it is challenging to make choices. Here are my top session choices for each day.
Friday:
This is an easy choice – Kevan Shokat‘s educational session about mechanism of resistance to targeted & IO therapies. As I have written many times: Kevan Shokat is one of those speakers you can not listen too often to because you will always learn something new.
Saturday:
The selection becomes more challenging! However, the Chemistry to the Clinic series of educational sessions – organized by the Chemistry in Cancer Research Working Group of American Association for Cancer Research – are Do-Never-Miss sessions. I am especially looking forward to part 3 which is focusing on innovative ways of drugging oncogenic pathways. We will learn more about Circle Pharma’s macrocyclic cyclin A/B inhibitors, about Delphia Therapeutics’ approach to kill cancer cells by overactivation of oncogenic pathways. And Rob Van Montfort will disclose work on a novel way to modulate splicing which was pursued in a collaboration between The Institute of Cancer Research & Merck Group.
I am still puzzling about how to clone myself to cover Frank McCormick‘s KRas inhibitor session as well as the New Frontiers in Drug Discovery session going head-to-head in the afternoon.
Sunday:
Now it gets really difficult with New Drugs on the Horizon 1 & 2, New Frontiers in Precision Oncology and so much more!
But I will go with the Opening Plenary as my top choice. Charles Sawyers, Carl June, Georg Winter & Regina Barzilay in back-to-back talks. Come on, this is too good to be true.
Monday:
Almost impossible to choose one session. But now I need to go with the 3rd part of New Drugs on the Horizon. Obviously, I am not going to miss any of the three sessions. The mix of modalities covered by these 12 first disclosures (glue degraders, T cell engagers, bispecific antibodies, highly selective inhibitors, dual payload ADCs, mycrocyclic peptides, bifunctional degraders) is a true representation of how modality innovation is starting to drive clinical progress.
I am not going to miss the AACR Award for Outstanding Achievement in Chemistry in Cancer Research (Congrats, Cheryl!) and the joint CICR-HMWG townhall in the late afternoon. Well, I have a reason to be there… plus free drinks.
Tuesday:
My top choice is Timothy Yap‘s session about synthetic lethality. With all the innovation around new modalities, we are starting to see how leveraging synthetic lethality translates into clinical impact. But I will for sure also learn about resistance mechanisms to ADCs and count kilometers in the poster sessions on tuesday.
Wednesday:
Do not even consider skipping Wednesday! The final plenary session is dedicated to innovative treatment modalities and how they shape the future of oncology. Featuring 4 excellent speakers including Raffaele Colombo discussing the evolution of ADCs.”

Emil Lou, Professor with Tenure at University of Minnesota:
“Today is Superman Day, and the first full day of AACR26. You don’t have to leap tall buildings in a single bound to make a difference. Supporting or otherwise contributing to research in cancer biology and in oncology clinicaltrials makes a difference. Up up and away AACR.”
Pavlos Msaouel, Physician-Scientist, Oncologist at MD Anderson:
“For the 2nd year in a row, two talented Msaouel Lab post-docs MD Anderson, Pankaj Chauhan and Kai Yu, co-mentored with Linghua Wang, Nizar Tannir&JJ Gao each received an AACR scholar-in-training award for Kidney cancer research AACR26 honoring their dedication to Cancer Research.”

“Fascinating AACR26 talk by Dr. Abu-Remaileh
VHL loss → ↑HIF2 → ↑PTHrP in ccRCC PTHrP drives cachexia + hypercalcemia — tumor-size independent Belzutifan ↓PTHrP & rescues body composition even WITHOUT tumor shrinkage.”

” Clinical trials mini symposium AACR26 Dr. Garralda
DKY709: IKFZ2 degrader +/- PS1 in pretreated ICI Novartis
3PR and 1CR 1CR beyond 2.5y DVR 41% Most common: ALT, rash and diarrhea, 2DLT in comb Peripheral neuropathy + QTc.” 
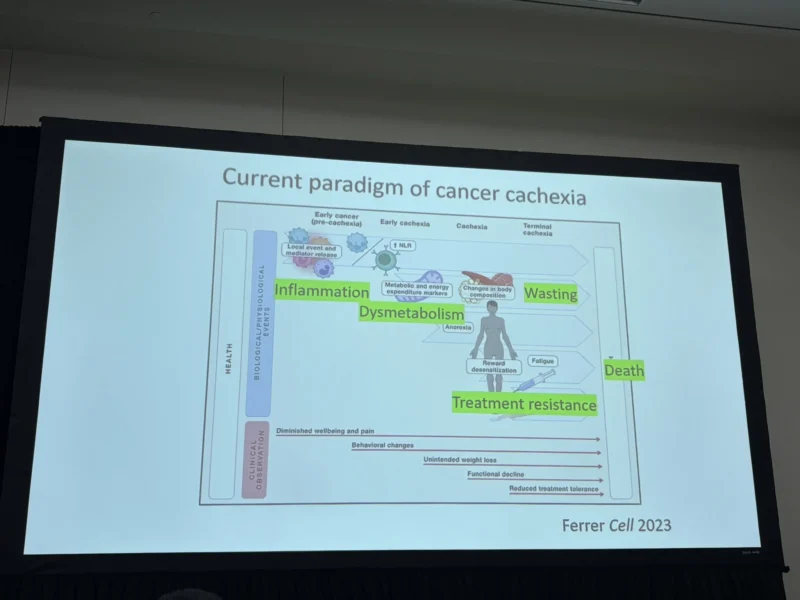
“Cancer cachexia isn’t just wasting—it’s a systemic “identity crisis” of host tissues.
AACR26 Dr. Teresa Zimmers highlights a 3-axis paradigm:
- Inflammation
- Dysmetabolism
- Cell Identity
Regression Time to target identity loss, not just metabolic symptoms.”
Raffaele Colombo, Associate Director of Medicinal Chemistry at Zymeworks:
“Superstar Elena Garralda presenting DKY709A12101C study DKY709, an IKZF2 degrader, alone or in combination with spartalizumab (PD-1).”
Ravali Reddy, Gyn Onc Fellow at MD Anderson:
” Let’s not confuse outcomes with impact. They are related but they are not the same.
A powerful reminder from Dr. Robert A. Winn, director of VCU Massey Comprehensive Cancer Center, in this morning’s AACR session on advancing patient-centered clinical trials.”
Tanya Bondar, Deputy Editor at Blood Cancer Discovery:
“The research article reporting final efficacy outcomes of the allogeneic CAR-NK cell product TAK-007 in non-Hodgkin lymphoma published in sync with Rizwan Romee commentary and AACR26 talk.”
Vivek Subbiah, Chief of Early-Phase Drug Development at the Sarah Cannon Research Institute:
“So nice to meet with Dr. Anthony Letai
We need an all‑hands‑on‑deck approach from everyone to beat cancer. These are the best times in oncology research – we’re finally going after the “undruggable,” targets and it’s incredible to have in the mission to cure cancer.”

Oriol Mirallas, Clinical Fellow at UT MD Anderson:
“Clinical trials mini symposium
Dr. Garralda
DKY709: IKFZ2 degrader +/- PS1 in pretreated
- 3PR and 1CR
- 1CR beyond 2.5y
- DVR 41%
- Most common: ALT, rash and diarrhea, 2DLT in comb
- Peripheral neuropathy + QTc.”

Jian Carrot-Zhang, Assistant Professor at Memorial Sloan Kettering Cancer Center:
“Thrilled to be selected as one of the NextGen Stars at the AACR Annual Meeting 2026!
I’ll be speaking on April 22 in the Major Symposium “Genetic and Environmental Determinants of Cancer,” where I’ll present our work on germline determinants of cancer progression using multi-ancestral real-world data.
Excited to be part of such an inspiring program—hope to see many of you there!”
See other articles on AACR 2026.


