Jeff Ryckman, Associate Professor of Radiation Oncology at WVU Medicine Camden Clark Medical Center, shared a post on X:
” New NEJM article: Perioperative enfortumab vedotin + pembrolizumab (EV+pembro) in MIBC (KEYNOTE-905).
Congratulations to the authors on an important randomized phase 3 trial in a tough, cisplatin-ineligible population.
Let’s walk through it .
Population:
- Cisplatin-ineligible (~82%) or declined (~18%)
- Majority node-negative MIBC (~95%)
- Mostly older, comorbid patients
- Standard comparator: cystectomy alone This is a group with historically poor outcomes → important unmet need
This is a group with historically poor outcomes → important unmet need.
Efficacy signals are strong:
- 2y eEFS: 39→ 75% (HR 0.40, 95CI 0.28-0.57)
- 2y eOS: 63→ 80% (HR 0.50, 95CI 0.33-0.74)
- pCR: 9→ 57%.
That pCR rate (>50%) is… eye-catching.
That pCR signal matters
We’ve long thought: High pCR rates → potential platform for organ preservation strategies.
If you can sterilize tumor pre-op, do all patients really need cystectomy?
But we have to talk safety
Grade 5 AEs: EV+pembro arm: 7.8%
Control (surgery alone): 5.7% This is not trivial
And importantly…
The control arm here is cystectomy.
We know radical cystectomy carries real perioperative risk, especially in older, frail patients.
That’s a 6-8% perioperative mortality signal across both groups, with multiple deaths tied to the surgery phase itself.
Now contrast with bladder preservation (CCRT):
Modern trimodality therapy:
- Grade 5 toxicity ~0–1% (generally speaking)
- No cystectomy upfront
- Many patients keep their bladder
Different paradigm entirely.
So we have:
EV+pembro + surgery → impressive pCR, EFS, OS → but ~6–8% treatment-related mortality range
vs
Bladder preservation → lower grade 5 toxicity → organ preservation → different selection, yes, but worth comparing
Key thought:
A pCR rate >50% should strengthen the case for organ preservation trials.
Not just better perioperative therapy.
But potentially avoiding cystectomy altogether in selected patients.
Which raises the real question
Instead of: EV+pembro → cystectomy
Should we be asking: EV+pembro → bladder preservation?
Or similarly:
Cisplatin-based + ICI combinations → CCRT platforms.
Leverage systemic control + local RT → maximize cure + preserve bladder.
People with bladder cancer care about survival… but also about keeping their bladder.
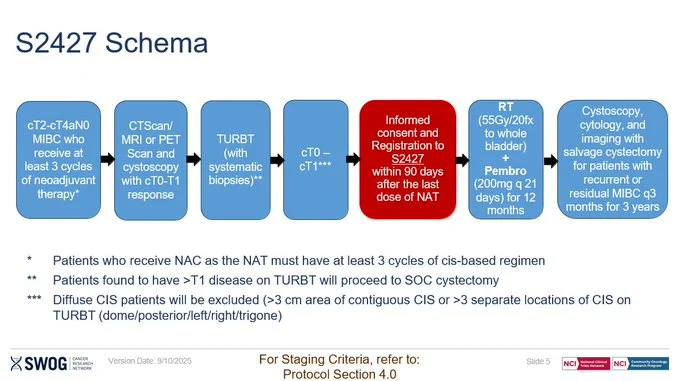
This is exactly why trials like S1806 and S2427 (BRIGHT) matter
Bladder preservation + immunotherapy → trying to define a new standard
If you believe in this question, let’s get them enrolled and move the field forward together.
Bottom line of KEYNOTE-905:
- Major step forward for cisplatin-ineligible MIBC
- Impressive efficacy signal
- Non-trivial grade 5 toxicity
- Strong biologic rationale for organ preservation
Final question:
With pCR >50% and meaningful systemic activity…
Is the next step EV+pembro with cystectomy or EV+pembro (or cisplatin-ICI) to enable bladder preservation?”
Title: Perioperative Enfortumab Vedotin and Pembrolizumab in Bladder Cancer
Authors: Christof Vulsteke, Nabil Adra, Pongwut Danchaivijitr, Maksym Sabadash, Alejo Rodriguez-Vida, Zhentao Zhang, Vagif Atduev, Y. Emre Göger, Steffen Rausch, Seok-Ho Kang, Yohann Loriot, Jens Bedke, Matthew D. Galsky, Peter H. O’Donnell, Gunhild von Amsberg, Nimira Alimohamed, Grzegorz Sulimka, Shilpa Gupta, Viktor Paramonov, Keita Nakane, Michael Mihm, Changting Meng, Caizhi David Huang, Chethan Ramamurthy, Blanca Homet Moreno, Anders Ullén

Other articles featuring Jeff Ryckman on OncoDaily.


