Despite decades of research, extensive-stage small cell lung cancer remains one of the most aggressive malignancies, characterized by rapid progression and limited long-term survival. While chemo-immunotherapy, including PD-1 inhibitors such as serplulimab, has improved outcomes modestly, most patients still relapse early, highlighting the need for innovative therapeutic strategies.
At European Lung Cancer Congress 2026, new data from the phase II BL-B01D1-204-01 study provide promising evidence for a novel approach combining a bispecific antibody-drug conjugate with immunotherapy in extensive-stage small cell lung cancer.

Study Design and Treatment Strategy
This phase II study evaluated iza-bren (BL-B01D1), a potential first-in-class bispecific antibody-drug conjugate targeting EGFR and HER3, linked to a topoisomerase I inhibitor payload. The agent was combined with serplulimab, an anti–PD-1 monoclonal antibody, in patients with extensive-stage small cell lung cancer.
The trial included two stages, with the current analysis focusing on treatment-naïve patients enrolled in stage II. Patients received iza-bren at either 2.5 mg/kg or 2.75 mg/kg on days 1 and 8 every three weeks, in combination with serplulimab administered every three weeks.
A total of 82 patients with extensive-stage small cell lung cancer were enrolled in this cohort, with 77 evaluable for response. The median follow-up duration was 10.5 months.

Key Efficacy Results
The combination demonstrated remarkably high response rates in extensive-stage small cell lung cancer, exceeding those typically observed with standard therapies.
The overall response rate reached 88.3%, with a confirmed response rate of 77.9%, indicating that a substantial proportion of patients experienced meaningful tumor shrinkage. Disease control was achieved in 94.8% of patients, reflecting broad clinical benefit.
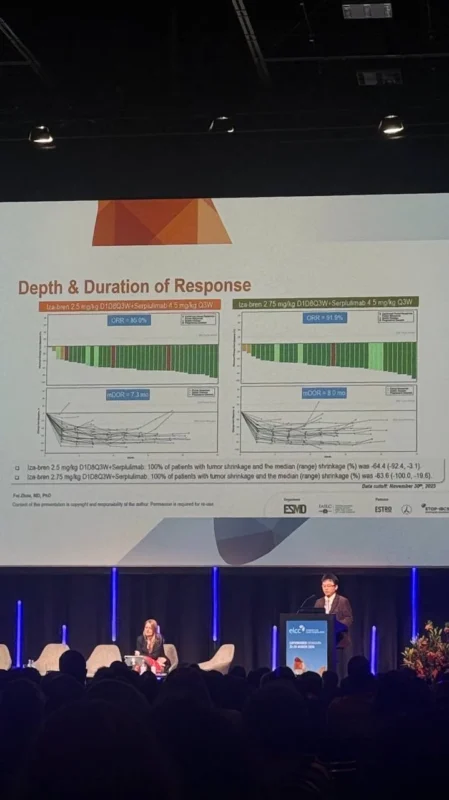
Responses were consistent across both dose levels. At the 2.5 mg/kg dose, the objective response rate was 85.0%, while at 2.75 mg/kg it reached 91.9%, suggesting robust activity across dosing strategies.
Median progression-free survival was 8.2 months, which compares favorably with historical outcomes in extensive-stage small cell lung cancer. The median duration of response ranged from 7.3 to 8.0 months, supporting the durability of treatment effect.
In addition, early survival signals were encouraging, with a 12-month overall survival rate of 80.8% in the overall cohort, highlighting the potential impact of this approach in extensive-stage small cell lung cancer.
Safety and Tolerability
The safety profile of the combination was manageable and consistent with expectations for antibody-drug conjugates and immunotherapy.
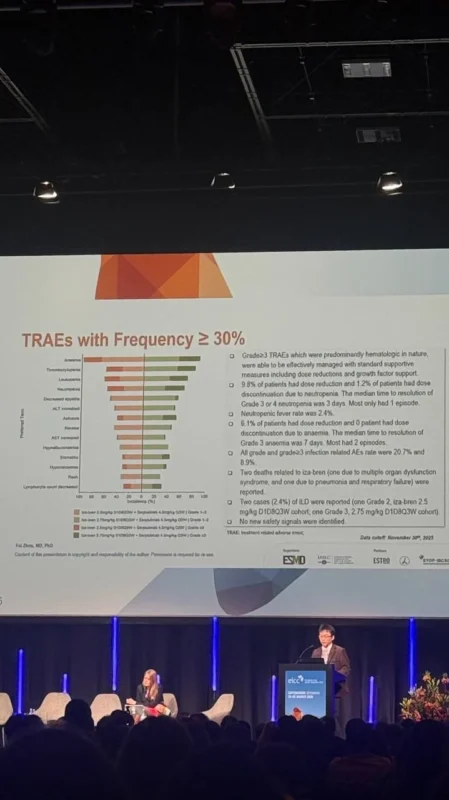
Hematologic toxicities were the most common adverse events, including anemia, thrombocytopenia, leukopenia, and neutropenia. These events were frequent but generally manageable with supportive care.
Among non-hematologic adverse events, decreased appetite was the most commonly reported. Grade 3 or higher toxicities were primarily hematologic in nature and were considered controllable within the clinical setting.
The rate of treatment discontinuation due to adverse events was low at 7.3%, suggesting acceptable tolerability. Two deaths (2.4%) were reported as possibly treatment-related, emphasizing the need for continued monitoring in further studies. Importantly, no new safety signals were identified.
Mechanistic Rationale
The combination of a bispecific antibody-drug conjugate with immunotherapy represents a novel therapeutic strategy in extensive-stage small cell lung cancer.
Iza-bren is designed to simultaneously target EGFR and HER3, delivering a cytotoxic payload directly to tumor cells, while serplulimab enhances antitumor immune responses through PD-1 blockade. This dual mechanism may provide both direct tumor killing and immune-mediated activity, potentially overcoming resistance mechanisms associated with conventional therapies.
Such approaches are particularly relevant in extensive-stage small cell lung cancer, where rapid tumor proliferation and early resistance limit the durability of existing treatments.
Clinical Implications
The results of this study suggest that combining ADCs with immunotherapy may represent a promising new direction in extensive-stage small cell lung cancer treatment.
The high response rates and encouraging survival outcomes observed in this trial exceed those typically seen with standard chemo-immunotherapy, raising the possibility that this strategy could redefine first-line treatment in selected patients.
However, these findings are based on a non-randomized cohort and require confirmation in larger, randomized studies to establish their role in clinical practice.
What Are Experts Saying About Trial?
Mario Balsa shared on X
Proud moment: my first paper as first author 🎉 frontiersin.org/journals/oncol… @FrontOncology
🎯 Real-world data on immunotherapy & TKIs in advanced chordoma
👥 Patients treated at a EURACAN sarcoma reference center
💥 Selected patients achieved meaningful disease control in this ultra-rare disease
Grateful to @ICO_oncologia and @hbellvitge (EURACAN centers), to all co-authors, and especially to Dr. Juan Martin-Liberal for his mentorship and endless patience 🙏
From chordoma to core data: rare tumors, real teamwork 🧡
Key Takeaway
The phase II BL-B01D1 study demonstrates that iza-bren combined with serplulimab achieves high response rates, durable activity, and manageable safety in extensive-stage small cell lung cancer.
This novel ADC-immunotherapy combination represents a potential breakthrough approach, warranting further investigation in randomized trials.
You can read full abstract here


