The role of immunotherapy in early-stage non-small cell lung cancer (NSCLC) continues to expand, particularly in the perioperative setting. The phase III KEYNOTE-671 has already established that adding Pembrolizumab to neoadjuvant chemotherapy significantly improves event-free survival (EFS), overall survival (OS), and pathological response rates compared with chemotherapy alone.
However, an important clinical question remains unresolved: what happens to patients who do not achieve a pathological complete response (pCR)? Since the majority of patients fall into this category, understanding their long-term outcomes is critical for real-world clinical decision-making.
At the ELCC 2026, Martin Reck presented a 5-year exploratory analysis focusing specifically on patients without pCR, offering important insights into the durability of benefit with perioperative immunotherapy.

Background
Pathological complete response has been widely considered a surrogate marker for long-term outcomes in several cancers, including NSCLC. Achieving pCR is associated with improved survival; however, most patients do not reach this endpoint even with modern multimodal therapy.
In the KEYNOTE-671 trial, more than 80% of patients in the pembrolizumab arm did not achieve pCR. This raises a key question: is the benefit of immunotherapy restricted to the minority who achieve deep pathological responses, or does it extend to the broader patient population?
This analysis directly addresses that gap, shifting the focus from pathological endpoints to long-term clinical outcomes.
Methods
KEYNOTE-671 is a randomized, double-blind, phase III trial enrolling patients with previously untreated, resectable stage II–IIIB (N2) NSCLC, according to the AJCC 8th edition staging system. Patients were randomized to receive neoadjuvant pembrolizumab or placebo in combination with chemotherapy for four cycles, followed by surgery and adjuvant pembrolizumab or placebo for up to 13 cycles.
The current exploratory analysis focused exclusively on patients who did not achieve pCR. With a median follow-up of approximately 60 months, the study provides one of the longest available datasets for perioperative immunotherapy in NSCLC.
The primary endpoint of interest in this subgroup was event-free survival, defined according to RECIST v1.1 criteria.

Results
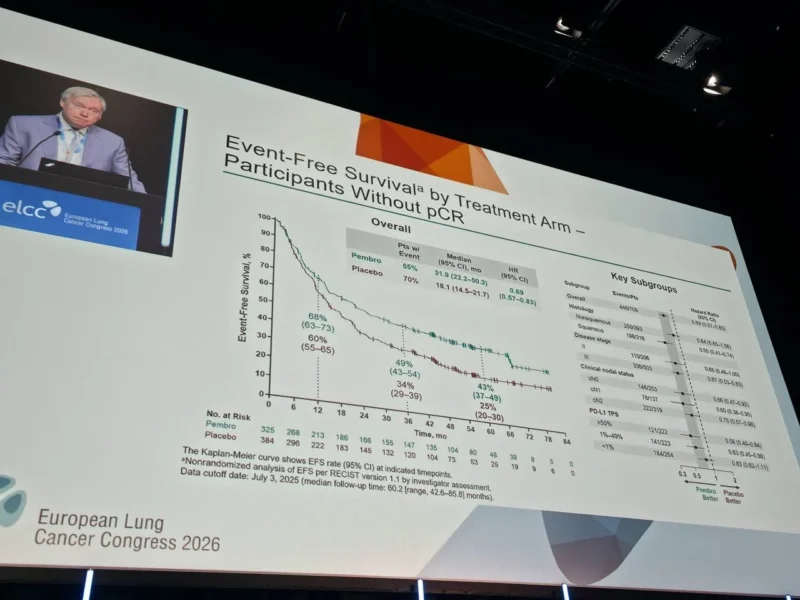
Among the randomized population, 325 patients in the pembrolizumab arm and 384 in the placebo arm did not achieve pCR. A substantial proportion of these patients proceeded to receive adjuvant therapy, and nearly half completed the full planned treatment course.
Despite the absence of pCR, pembrolizumab continued to demonstrate a significant and clinically meaningful benefit in event-free survival. The median EFS was 31.9 months in the pembrolizumab group compared with 18.1 months in the placebo group. At five years, 42.9% of patients in the pembrolizumab arm remained event-free, compared with 25.2% in the placebo arm. This translated into a hazard ratio of 0.69, indicating a 31% reduction in the risk of recurrence or death.
Importantly, this benefit was consistent across multiple subgroups. Patients with stage II and stage III disease both derived benefit, as did those with varying nodal involvement. The effect was observed regardless of PD-L1 expression, although a greater magnitude of benefit appeared in patients with higher PD-L1 levels. Histology also influenced outcomes, with a more pronounced benefit seen in squamous tumors compared with non-squamous disease.
At the time of analysis, overall survival data for this subgroup were not yet mature, and further follow-up will be required to determine the full survival impact.
The safety profile of pembrolizumab remained consistent with previous reports. Grade 3 or higher treatment-related adverse events occurred slightly more frequently in the pembrolizumab arm, and immune-related adverse events were observed in a minority of patients. No new safety signals emerged with longer follow-up.
Clinical Interpretation
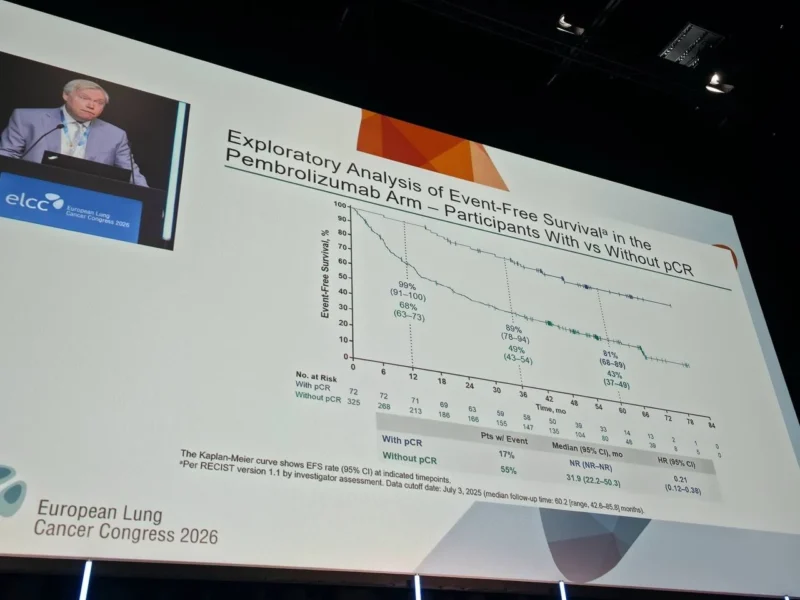
This analysis provides an important shift in how clinicians should interpret perioperative immunotherapy outcomes. While pCR has been considered a key marker of treatment success, these findings demonstrate that meaningful clinical benefit extends beyond this endpoint.
Patients with residual disease after neoadjuvant therapy still experienced a substantial improvement in event-free survival with pembrolizumab. This suggests that immunotherapy contributes to systemic disease control in ways that are not fully captured by pathological response alone.
The results challenge the notion that failure to achieve pCR represents treatment failure. Instead, they support a broader understanding of benefit, where delayed recurrence and prolonged disease control are equally important endpoints.
Implications for Practice
The findings reinforce perioperative pembrolizumab plus chemotherapy as a standard of care for resectable early-stage NSCLC. Clinicians should be cautious not to rely solely on pCR when evaluating treatment effectiveness or making subsequent management decisions.
Rather, these data support continuing immunotherapy strategies even in patients with residual disease, as they may still derive long-term benefit.

Conclusion
The 5-year exploratory analysis of the KEYNOTE-671 confirms that perioperative pembrolizumab improves event-free survival in early-stage NSCLC, even among patients who do not achieve pCR.
These findings highlight a critical message for clinical practice: while pathological response is important, it does not fully define treatment benefit. Immunotherapy provides durable clinical advantages that extend beyond early tumor eradication.
As perioperative strategies continue to evolve, this study underscores the importance of focusing on long-term outcomes rather than relying solely on surrogate markers.
You can read full abstract here


