At the European Lung Cancer Congress 2026, the ETOP-Roche i-TIMES study provided definitive evidence addressing a long-debated question in oncology:
Does the timing of immune checkpoint inhibitor (ICI) administration influence survival outcomes in lung cancer?
The concept of chronotherapy has suggested that biological rhythms could affect treatment efficacy, particularly in immunotherapy, where immune system activity varies throughout the day. However, robust clinical evidence has been lacking. The i-TIMES study represents one of the largest analyses to date evaluating whether early versus late administration of ICIs impacts overall survival.

Study Design and Statistical Framework
The i-TIMES study was a large-scale, pooled analysis of eight international randomized clinical trials, including patients with advanced or metastatic lung cancer treated with ICIs either alone or in combination with chemotherapy.
To minimize bias and ensure comparability, investigators applied a propensity score matching (PSM) approach, balancing key clinical factors such as PD-L1 expression, presence of liver and brain metastases, and gender.
Patients were categorized based on the timing of their first two immunotherapy cycles:
- Early administration: before 12:00 PM
- Late administration: after 12:00 PM
The primary endpoint was overall survival (OS), evaluated in a non-inferiority framework. The predefined non-inferiority margin was a hazard ratio (HR) of 1.18, with a one-sided alpha of 2.5% and statistical power of 80%.
In total, 3,060 patients were included in the primary cohort, with a matched analysis population of 1,550 patients (775 per group).

Overall Survival Outcomes
The study demonstrated no clinically meaningful difference in survival based on treatment timing.
In the matched cohort:
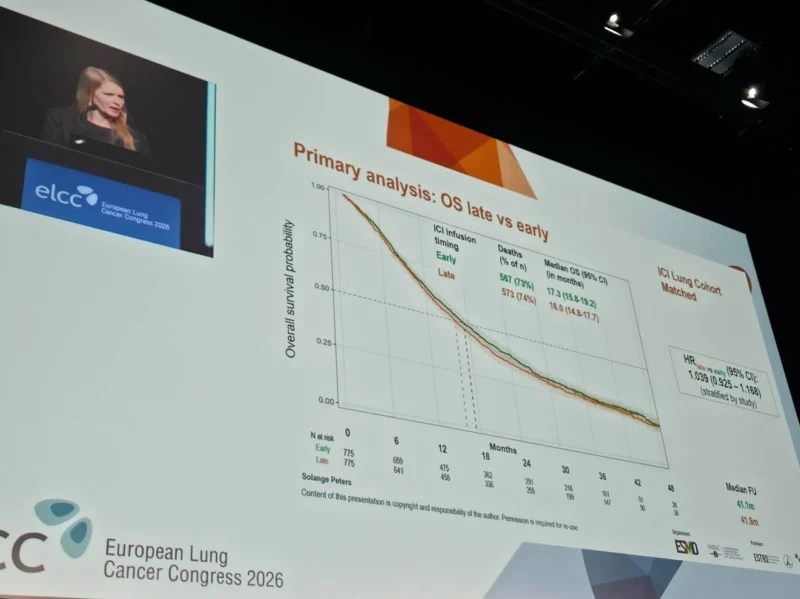
- Median OS was 17.3 months in the early administration group
- Median OS was 16.0 months in the late administration group
- Hazard ratio for late versus early administration: 1.039 (95% CI, 0.925–1.168)
Importantly, the upper bound of the confidence interval remained below the predefined non-inferiority margin of 1.18, confirming that late administration was non-inferior to early administration.
In the overall cohort, findings were consistent:
- Median OS: 18.5 months (early) vs 15.7 months (late)
- Hazard ratio: 1.088 (95% CI, 0.982–1.207)
Survival curves showed substantial overlap, with no meaningful separation between early and late treatment groups.
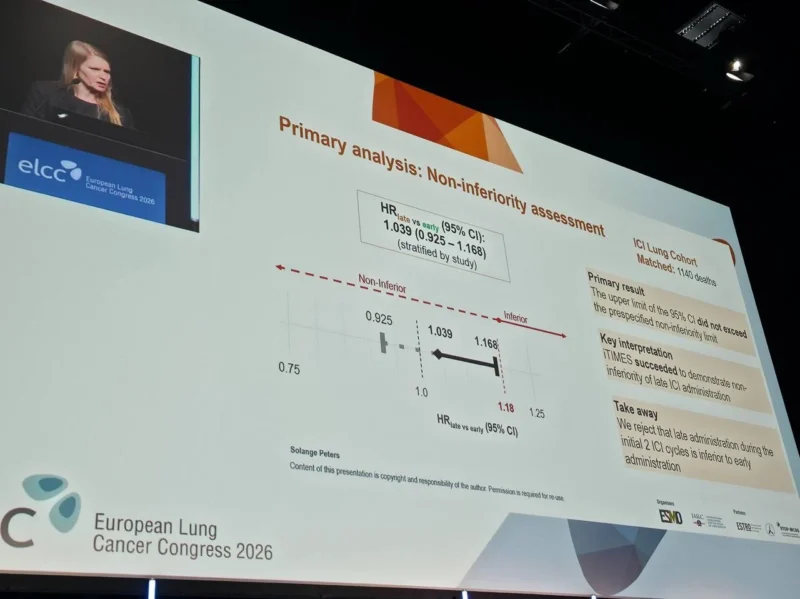
Non-Inferiority Confirmed
The statistical analysis clearly met its primary objective. The study confirmed that late administration of ICIs is not inferior to early administration, rejecting the hypothesis that treatment timing within the day significantly affects survival outcomes.
This finding directly addresses prior uncertainty generated by smaller or retrospective studies suggesting potential benefits of morning immunotherapy administration.
Clinical Interpretation
The results indicate that intravenous immunotherapy timing is unlikely to represent an independent determinant of treatment efficacy in lung cancer. While circadian biology remains a compelling area of research, its clinical relevance in this context appears limited.
The magnitude of any potential chronotherapy effect is small and does not translate into measurable survival differences in a large, rigorously analyzed patient population.
Implications for Clinical Practice
These findings have immediate and practical implications.
First, they support a flexible approach to immunotherapy scheduling. In routine clinical practice, treatment timing is often dictated by logistical considerations, including clinic workflow, infusion capacity, and patient availability. The i-TIMES data confirm that such flexibility does not compromise outcomes.
Second, the results simplify treatment delivery by eliminating the need to prioritize specific times of day for immunotherapy administration. This is particularly relevant in high-volume oncology centers where scheduling constraints are common.
Finally, the findings reinforce a patient-centered approach, allowing clinicians to align treatment timing with individual patient needs and preferences without concern for reduced efficacy.

Conclusion
The ETOP-Roche i-TIMES study demonstrates that the timing of immunotherapy administration, morning versus afternoon, does not impact overall survival in patients with advanced lung cancer.
By confirming non-inferiority of late versus early administration, the study provides strong evidence supporting flexible, pragmatic, and patient-centered scheduling of immune checkpoint inhibitors.
These results help resolve a longstanding clinical question and indicate that, in lung cancer, when immunotherapy is given during the day matters far less than ensuring timely and consistent treatment delivery.


