HER2-mutant non-small cell lung cancer (NSCLC) represents a distinct molecular subset characterized by limited targeted treatment options and historically poor outcomes following standard chemotherapy and immunotherapy. The development of antibody–drug conjugates has introduced a new therapeutic paradigm, with trastuzumab deruxtecan (T-DXd) emerging as a highly active agent in this setting. The phase 2 DESTINY-Lung02 trial evaluated two dose levels of T-DXd (5.4 mg/kg and 6.4 mg/kg) in patients with previously treated metastatic HER2-mutant NSCLC, demonstrating clinically meaningful responses and durable disease control.
A recent corrigendum to the published analysis clarified several data points related to baseline characteristics and safety outcomes, without altering the overall conclusions of the study. These corrections provide further precision in understanding patient characteristics, prior treatment exposure, and interstitial lung disease (ILD) risk, key elements in optimizing the use of T-DXd in clinical practice (Jänne et al., 2026).
Study Design and Patient Population
DESTINY-Lung02 was a dose-blinded, randomized phase 2 study enrolling patients with HER2-mutant metastatic NSCLC who had received prior systemic therapy. Patients were assigned to receive either T-DXd 5.4 mg/kg (n=102) or 6.4 mg/kg (n=50).
The patient population reflected a heavily pretreated cohort. The median number of prior lines of systemic therapy for advanced disease was 2.0 in both treatment groups, following correction from earlier reported values. Notably, 67.6% of patients in the 5.4 mg/kg group and 62.0% in the 6.4 mg/kg group had received two or fewer prior lines, while 32.4% and 38.0%, respectively, had received more than two lines of therapy (Jänne et al., 2026).
The majority of patients had adenocarcinoma histology, accounting for 98.0% in the 5.4 mg/kg cohort and 100% in the 6.4 mg/kg cohort, reflecting the known predominance of HER2 mutations in this subtype. Baseline characteristics were balanced between groups, with a median age of 59.4 years in the 5.4 mg/kg group and 61.3 years in the 6.4 mg/kg group, and a high proportion of female patients (63.7% and 68.0%, respectively).
Importantly, prior exposure to immunotherapy was common. Anti–PD-(L)1 therapy had been administered to 73.5% of patients in the 5.4 mg/kg group and 78.0% in the 6.4 mg/kg group, with approximately half receiving combination platinum and immunotherapy regimens. These data underscore the refractory nature of the population and the unmet need addressed by T-DXd (Jänne et al., 2026).
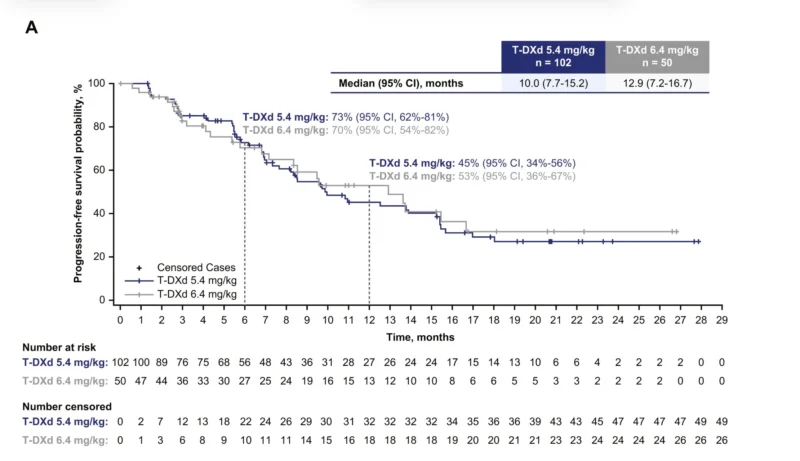
Efficacy Outcomes
The efficacy of trastuzumab deruxtecan remained robust following correction of the dataset. Progression-free survival (PFS) analysis demonstrated clinically meaningful disease control in both dosing groups.
The median PFS was 10.0 months (95% CI, 7.7–15.2) in the 5.4 mg/kg group and 12.9 months (95% CI, 7.2–16.7) in the 6.4 mg/kg group. Landmark PFS rates further illustrated durable benefit, with 73% (95% CI, 62%–81%) of patients in the 5.4 mg/kg group and 70% (95% CI, 54%–82%) in the 6.4 mg/kg group progression-free at early timepoints, declining to 45% and 53%, respectively, at later follow-up intervals (Jänne et al., 2026).
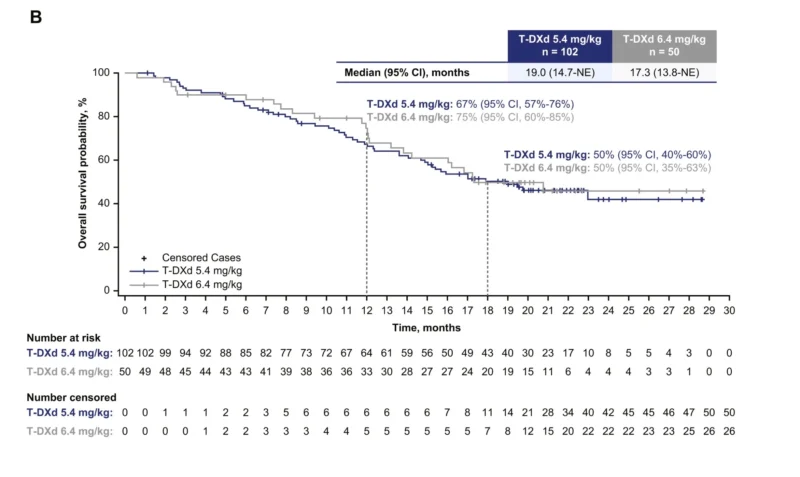
Overall survival (OS) outcomes were similarly encouraging. The median OS was 19.0 months (95% CI, 14.7–not estimable) in the 5.4 mg/kg group and 17.3 months (95% CI, 13.8–not estimable) in the 6.4 mg/kg group. Survival probabilities remained high over time, with 67% and 75% of patients alive at earlier timepoints and 50% survival observed in both groups at later follow-up (Jänne et al., 2026).
These results confirm that trastuzumab deruxtecan provides substantial clinical benefit in a heavily pretreated HER2-mutant NSCLC population, with consistent efficacy across dose levels.
Safety and Interstitial Lung Disease
Safety remains a critical consideration with T-DXd, particularly the risk of interstitial lung disease. The corrected analysis provides a detailed and more accurate understanding of ILD incidence and outcomes.
Any-grade ILD occurred in 14.9% of patients receiving 5.4 mg/kg and 32.0% of those receiving 6.4 mg/kg, highlighting a dose-dependent increase in pulmonary toxicity. Most events were low grade, with grade 1 ILD observed in 4.0% and 6.0%, and grade 2 ILD in 8.9% and 22.0% of patients, respectively.
Higher-grade events were less frequent but clinically significant. Grade ≥3 ILD occurred in 2.0% of patients in the 5.4 mg/kg group and 4.0% in the 6.4 mg/kg group, while fatal (grade 5) ILD was reported in 1.0% and 2.0% of patients, respectively (Jänne et al., 2026).
Management patterns showed that 80.0% of patients in the 5.4 mg/kg group and 81.3% in the 6.4 mg/kg group received corticosteroids, reflecting adherence to recommended treatment strategies for suspected drug-related pneumonitis. Antibiotics were also used in a subset of patients, either alone or in combination with steroids.
Outcomes of ILD events varied. In the 5.4 mg/kg cohort, 66.7% of patients recovered or resolved their ILD, while 20.0% had ongoing unresolved toxicity. In the higher-dose group, recovery was observed in 50.0% of cases, with a higher proportion (37.5%) experiencing unresolved disease at the time of analysis (Jänne et al., 2026).
These findings reinforce the importance of early recognition and prompt management of ILD, as well as careful dose selection in clinical practice.
Impact of Prior Immunotherapy
The corrigendum also clarified data regarding prior immunotherapy exposure, an important factor in interpreting both efficacy and toxicity outcomes.
In patients who developed ILD, prior immunotherapy exposure was common, with 73.3% in the 5.4 mg/kg group and 81.3% in the 6.4 mg/kg group having received prior IO therapy. Time from prior immunotherapy also varied, with 33.3% and 43.8% of patients, respectively, having completed IO therapy more than 6 months before starting T-DXd (Jänne et al., 2026).
These data suggest that prior immune checkpoint inhibitor exposure does not preclude benefit from trastuzumab deruxtecan but may contribute to the complexity of toxicity profiles, particularly in relation to pulmonary adverse events.
Clinical Interpretation
The corrected data from DESTINY-Lung02 confirm that trastuzumab deruxtecan is a highly effective treatment option for patients with HER2-mutant metastatic NSCLC. The consistency of efficacy outcomes across both dose levels, combined with manageable toxicity, supports its role as a standard therapy in this setting.
Importantly, the differences in ILD incidence between the two dosing regimens highlight the need for careful benefit-risk assessment. While the 6.4 mg/kg dose demonstrates slightly longer median PFS, it is associated with a higher rate of ILD, suggesting that the lower dose may offer a more favorable safety profile without compromising overall survival.
The corrected data also emphasize the importance of accurate baseline characterization and treatment history in interpreting trial outcomes. The adjustment in prior therapy lines and immunotherapy exposure provides a clearer understanding of the heavily pretreated nature of the study population and reinforces the clinical relevance of the observed results.
Conclusion
The DESTINY-Lung02 corrigendum refines key data points while reaffirming the central findings of the study. Trastuzumab deruxtecan demonstrates meaningful and durable efficacy in HER2-mutant NSCLC, with median overall survival approaching 19 months and progression-free survival exceeding 10 months in a previously treated population.
Safety remains manageable, although interstitial lung disease continues to represent an important risk, particularly at higher doses. The corrected analysis strengthens confidence in both the efficacy and safety profile of T-DXd and supports its integration into clinical practice.
As targeted therapies continue to evolve in thoracic oncology, the DESTINY-Lung02 study, and its refined dataset, provides a critical foundation for optimizing treatment strategies in HER2-driven lung cancer.
Full article here.


