Optimizing first-line treatment in metastatic colorectal cancer remains a key challenge, particularly when balancing treatment intensity with tolerability. While triplet chemotherapy can increase response rates in selected patients, its added value in combination with antiangiogenic agents beyond bevacizumab is still not clearly defined.
Understanding how newer agents such as ramucirumab perform across different chemotherapy backbones is therefore essential to refine treatment strategies.
This study was published in the European Journal of Cancer on March 19, 2026.
Title: Randomized phase II study of FOLFIRI plus ramucirumab versus FOLFOXIRI plus ramucirumab as first-line treatment for metastatic colorectal cancer: WJOG9216G (RECAST)
Authors: Yosuke Kito, Kentaro Yamazaki, Hirokazu Shoji, Takeshi Yamada, Takahiro Tsushima, Seiichiro Mitani, Kazuhiro Shiraishi, Hisateru Yasui, Hiroki Hara, Kenro Hirata, Taito Esaki, Yudai Shinohara, Takao Tsuzuki, Shinya Kajiura, Toshiki Masuishi, Naoki Izawa, Eishi Baba, Kohei Murata, Naoya Akazawa, Yozo Suzuki, Hironaga Satake, Narikazu Boku, Ichinosuke Hyodo, Kenichi Yoshimura, Hisato Kawakami, Shuichi Hironaka, Kei Muro.
Background
In current practice, first-line treatment for metastatic colorectal cancer generally consists of a chemotherapy doublet such as FOLFOX or FOLFIRI combined with a biologic agent. In selected patients with good performance status, triplet chemotherapy with FOLFOXIRI is sometimes used to increase response rates, although at the cost of higher toxicity.
Ramucirumab, a monoclonal antibody targeting VEGFR-2, has demonstrated benefit in multiple tumor types and is approved in combination with FOLFIRI in the second-line setting for metastatic colorectal cancer. However, its role in the first-line setting remains less clearly defined.
Study design and methods
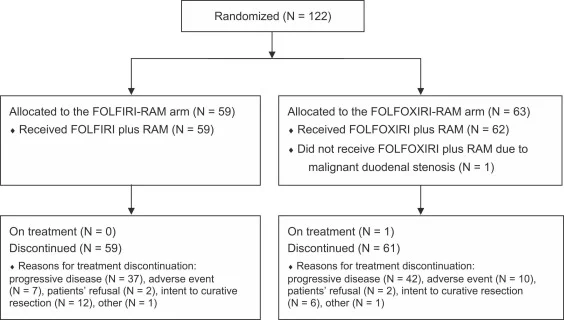
RECAST was a multicenter, open-label, randomized phase II trial conducted across 26 Japanese institutions.
A total of 122 patients with previously untreated, unresectable metastatic colorectal cancer were randomized:
- 59 patients to FOLFIRI plus ramucirumab
- 63 patients to FOLFOXIRI plus ramucirumab
The primary endpoint was confirmed objective response rate (ORR). Secondary endpoints included early tumor shrinkage (ETS), progression-free survival (PFS), PFS2, overall survival (OS), depth of response (DpR), R0 resection rate, and safety.
Median follow-up was 42.1 months in the FOLFIRI arm and 40.4 months in the FOLFOXIRI arm.
Results
The study did not meet its primary endpoint. Confirmed ORR was 59.3% (95% CI 45.7–71.9) vs 60.3% (95% CI 47.2–72.4; P = 0.91), and the primary endpoint was not met.
Despite this similarity in response rates, differences emerged in other clinically relevant endpoints. Early tumor shrinkage at week 8 was significantly higher in the FOLFIRI arm, with 71.2% of patients achieving at least a 20% reduction in tumor burden compared with 52.4% in the FOLFOXIRI arm (P = 0.033). Depth of response was similar between the two groups (41.3% vs 41.7%; P = 0.97).
Progression-free survival was comparable, with a median of 11.5 vs 10.5 months (HR 1.00; P = 0.99). Similarly, PFS2 did not differ significantly between the two strategies (HR 1.27; P = 0.22).
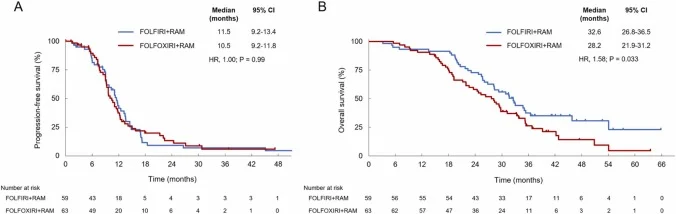
In contrast, overall survival was longer in the FOLFIRI-based regimen. Median overall survival was 32.6 vs 28.2 months (HR 1.58; P = 0.033), although the study was not powered for definitive survival comparisons. At the time of final analysis, a higher proportion of deaths had occurred in the FOLFOXIRI group (64.4% vs 79.4%).
Resection outcomes also numerically favored FOLFIRI plus ramucirumab, with R0 resection achieved in 18.6% of patients compared with 11.1% in the triplet arm. Across both treatment groups, patients who underwent R0 resection had markedly improved survival, with a median overall survival of 54.1 vs 26.8 months compared with those who did not undergo resection (HR 0.14; P < 0.001).
Safety
The safety analysis highlighted a clear difference between the two regimens. The FOLFOXIRI-based strategy was associated with substantially higher toxicity, particularly hematologic adverse events. Rates of grade 3 or higher neutropenia were notably higher, and febrile neutropenia, anorexia, and diarrhea were also more frequent in the triplet arm. Grade ≥3 neutropenia occurred in 44.1% vs 72.6%, and febrile neutropenia in 3.4% vs 11.3%.
Serious adverse events requiring prolonged hospitalization occurred much more often with FOLFOXIRI plus ramucirumab. In addition, two treatment-related deaths were reported in this group, due to pulmonary infection and colonic perforation. While hypertension and proteinuria were observed in both arms, consistent with the known profile of ramucirumab, no unexpected safety signals emerged.
Interpretation
Several factors may explain the absence of benefit with the more intensive regimen. Dose intensity during the early treatment period was lower in the FOLFOXIRI arm, suggesting reduced tolerability that may have impacted efficacy. In addition, a higher proportion of patients with liver-limited disease in the FOLFIRI arm may have contributed to higher resection rates and improved outcomes.
The findings are consistent with the hypothesis that ramucirumab may be more effective in irinotecan-based combinations than in oxaliplatin-containing regimens, a concept that has been suggested in prior studies.
Importantly, as a phase II selection trial, RECAST was not designed to definitively compare survival outcomes, and these results should be interpreted within that context.
Conclusion
The RECAST trial demonstrates that FOLFOXIRI plus ramucirumab does not improve efficacy compared with FOLFIRI plus ramucirumab in the first-line treatment of metastatic colorectal cancer, while leading to increased toxicity.
FOLFIRI plus ramucirumab achieved similar response rates, comparable progression-free survival, higher early tumor shrinkage, longer overall survival, and a more favorable safety profile. These results support further investigation of this regimen and highlight the importance of optimizing treatment intensity based on both efficacy and tolerability.
The study is available in the European Journal of Cancer.