This week in breast oncology highlights how treatment strategies continue to evolve across targeted therapy, immunotherapy, survivorship, and precision medicine, driven by clinical trials, translational research, and real-world insights from global experts.
Patrick Aouad – Postdoc at Genentech, Discovery Oncology
I am really excited to share that our review on preclinical models of breast cancer metastasis is now out in Nature Partner Journal (NPJ) Breast Cancer! We summarize decades of experimental and translational research to provide a comprehensive overview of the major preclinical models used to study breast cancer metastasis.
I am very grateful to my co-authors, and especially thankful to have worked on this review with my past mentors, Nadine Darwiche and Cathrin Brisken MD PhD, as well as my current mentor, Ciara Metcalfe-A full circle moment for me!
Many thanks to the constructive criticism we have received from the reviewers who have helped us improve the review, and to the editorial team at NPJ Breast Cancer.
If you are interested, here’s the unedited version of the article (part of the Advances in Modeling collection!):

Read About Study on OncoDaily Breast
Evandro de Azambuja – BSMO President 2023-2026
Our pooled analysis sheds new light on the safety profile of Sacituzumab Govitecan in patients with metastatic breast cancer.
Jasmin Hundal, M.D., DipABLM, M.S., M.P.H. – Hematology/Oncology Fellow/Advancing Cancer Care Through Research, Education &Lifestyle Medicine
A clinically important question in ER-positive, HER2-negative advanced breast cancer is no longer just whether to continue targeting the estrogen receptor after progression, but which endocrine backbone may offer the greatest value when paired with CDK4/6 inhibition.
This indirect treatment comparison across EMBER-3, MONARCH 2, and postMONARCH helps address that question.
Using three complementary methods (Bucher, MAIC, and propensity score matching), imlunestrant + abemaciclib demonstrated a consistent numerical reduction in the risk of progression or death compared with fulvestrant + abemaciclib in patients previously treated with endocrine therapy ± CDK4/6 inhibitors:
• Bucher HR 0.77 (95% CI 0.58–1.04)
• MAIC HR 0.77 (95% CI 0.55–1.06)
• PSM HR 0.83 (95% CI 0.56–1.22)None reached statistical significance, and the analysis was not powered for hypothesis testing, but the consistent directionality across all three methodologies strengthens interpretive confidence.
Equally important is how we interpret cross-trial data:
The comparison underscores the need to account for baseline heterogeneity:
> postMONARCH enrolled only patients progressing on prior CDK4/6i, whereas EMBER-3 included ~40% without prior exposure
>> Despite this, fulvestrant PFS was similar across trials (~5.3 vs 5.5 months), highlighting the role of underlying prognostic differences and reinforcing the value of adjusted approaches (MAIC/PSM) and anchored comparisons when contextualizing outcomes.While limitations inherent to ITCs remain, including residual confounding, follow-up durations differed across trials (13–14 months in EMBER-3/postMONARCH vs. 80 months in MONARCH 2), and subgroup analyses were limited, the consistency of findings across methodologies supports the evolving role of oral SERDs as a meaningful endocrine backbone.
>>As more patients receive CDK4/6 inhibitors earlier in their disease course, optimizing endocrine sequencing will increasingly depend on prior therapy, disease biology, toxicity, route of administration, and patient preference.
This is where the field is moving: toward more precise endocrine backbones, more individualized sequencing, and, hopefully, more durable disease control.
Paolo Tarantino – Advanced Fellow press Dana-Faber Cancer Institute
Traditional HER2 IHC scoring was fine-tuned for trastuzumab. Predicting the efficacy of ADCs, though, may require more sensitive assays.
In this pilot study, we show that HER2 quantification with multiple proteomic (HS-HER2, RPPA), transcriptomic (HER2DX) or blood-based (DNADX) assays better predicts the effectiveness of T-DXd than IHC, and we suggest a role for payload markers (eg. TOPO1 quantification).
This is the product of 4 years of work, only made possible by Sara Tolaney’s mentoring, by the tremendous biospecimens and statistical teams of the Dana-Farber Cancer Institute Breast Oncology Center, by amazing collaborators around the globe (including David Rimm’s lab at Yale University, REVEAL GENOMICS , Ignite Proteomics, Duke Cancer Institute), by the precious advice of patients advocates, and by funding kindly committed to improving the precision of ADC therapy by the Terri Brodeur Breast Cancer Foundation (TBBCF) , Metavivor Online and the Saverin family.

Read About Study on OncoDaily
Gustavo Monnerat – Deputy Editor @The Lancet – Americas/ PhD & MBA/ Digital and Global Health / AI & Evidence Systems in Healthcare
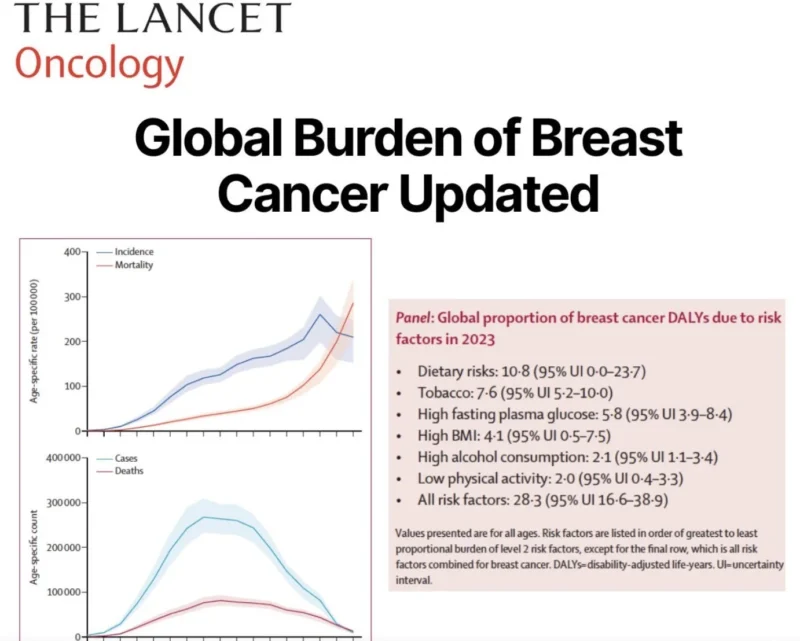
Largest study of breast cancer burden just published in The Lancet Oncology, covering 204 countries from 1990 to 2023 with forecasts to 2050.
Key findings from GBD 2023:
-> 2.30 million new cases, 764,000 deaths, and 24.1 million DALYs among women globally in 2023. By 2050, cases are projected to reach 3.56 million with 1.37 million deaths.
-> In low-income countries, age-standardised incidence surged 147.2% since 1990 versus just 1.2% in high-income countries.
-> Risk factors including dietary risks, tobacco, and high fasting plasma glucose accounted for 28.3% of breast cancer DALYs in 2023.
These numbers demand urgent action on early detection and treatment access, though sparse registry data in these settings means the true burden may vary.
GBD 2023 Breast Cancer Collaborators. Global, regional, and national burden of breast cancer among females, 1990–2023, with forecasts to 2050: a systematic analysis for the Global Burden of Disease Study 2023 . Lancet Oncol 2026
Professor Kefah Mokbel – Chief of Breast Cancer Surgery @ London Breast Institute
The updated PHILA trial reinforces an important point in HER2-positive metastatic breast cancer:
More complete HER-family blockade translates into better outcomes.
Adding pyrotinib (an irreversible pan-HER TKI) to trastuzumab and docetaxel significantly improved both progression-free and overall survival—likely reflecting dual extracellular and intracellular pathway inhibition.
So why isn’t pyrotinib widely used in first-line practice?
Lack of direct comparison with established standards (e.g. pertuzumab-based regimens)
Evolving landscape with antibody-drug conjugates moving into first line
Toxicity profile—particularly diarrhoea
Geographic/regulatory differencesTake-home:
Pyrotinib is a highly active option that may be under-used in clinical practice —but its place in global first-line therapy remains limited by comparative evidence and rapidly shifting standards.
Joseph Steward – Medical & Marketing Writer/ Biotech, Genomics, Oncology & Regulatory
Trastuzumab deruxtecan (T-DXd) is an antibody-drug conjugate successfully used to treat HER2-low and HER2-positive metastatic breast cancer, but resistance consistently develops. Using multivariate Cox proportional hazards in a real-world cohort of 2,799 patients with breast cancer, we aimed to identify clinically relevant T-DXd resistance mechanisms.
In patients with samples collected prior to T-DXd treatment, higher expression of ERBB2 (HER2) and lower expression of ABCC1 (an ATP-binding cassette transporter involved in drug efflux) were significantly associated with longer T-DXd-related overall survival (OS); ABCC1 predicted OS independently of HER2.
Furthermore, mutations in several genes were enriched in post-T-DXd samples compared to unmatched T-DXd-naïve samples, including ERBB2, NFE2L2 (a transcriptional activator of ABCC1), and KEAP1 (a negative regulator of NFE2L2), indicating plausible resistance mechanisms related to HER2 target levels and ABCC1-mediated drug efflux.
Identifying such resistance mechanisms might lead to improved methods of precision oncology and novel therapeutic approaches to overcome resistance.
Jeff Legos – Chief Oncology Officer at Pfizer
Today Pfizer announced positive topline Phase 2 results for our next generation CDK4 inhibitor in HR-positive, HER2-negative advanced or metastatic #BreastCancer, showing a statistically significant and clinically meaningful improvement in progression free survival with a manageable safety profile:
These results deepen our confidence in this differentiated approach as a potential new cell cycle inhibitor backbone for HR+ breast cancer, and we are continuing to accelerate its development in earlier lines of therapy where it may provide the greatest benefit.
My sincere thanks to the patients, investigators, and Pfizer teams whose commitment made this progress possible.
Komal Jhaveri, Md, FACP, FASCO – Patricia and James Cayene Chair for Junior Faculty, Breast Medicine and Early Drug Development Service at Memorial Sloan Kettering Cancer Center
DB-08 provided safety data for various strategic combinations including TDXd plus ET, certainly relevant given the recent DB-05 data in the residual disease setting wherein we can use this combination for triple positive breast cancer Memorial Sloan Kettering Cancer Center
Lorenzo Scardina – MD, PhD candidate, FEBS, BRESO Certified Breast Surgeon, Fondazione Policlinico Universitario Agostino Gemelli IRCCS
Finally out our new publication in Annals of Surgical Oncology!
Proud to share our results on axillary nodal positivity in early-stage invasive lobular carcinoma and its implications for the omission of sentinel lymph node biopsy.
In this retrospective analysis, approximately 1 in 5 patients had nodal metastases at final pathology.
These results highlight the biologic distinctiveness of invasive lobular carcinoma.Even in small, luminal tumors with negative imaging, the risk of axillary disease remains clinically relevant.
Grateful to collaborate with the colleagues of the Breast Unit at Fondazione Policlinico Universitario Agostino Gemelli IRCCS Università Cattolica del Sacro Cuore.