Breast cancer remains the most frequently diagnosed malignancy in women globally and continues to represent a major contributor to cancer-related mortality, with breast cancer metastasis accounting for the vast majority of deaths (Siegel et al., 2024). While early-stage breast cancer is often curable, the transition to metastatic disease introduces a complex biological landscape characterized by tumor heterogeneity, immune evasion, and organ-specific colonization.
Metastasis is not a single event but a multistep evolutionary process involving local invasion, intravasation into the bloodstream, survival under shear stress and immune surveillance, extravasation into distant tissues, and eventual colonization. Importantly, disseminated tumor cells may remain dormant for years or even decades before reactivation, a phenomenon particularly relevant in hormone receptor-positive (HR+) breast cancer (Park et al., 2022).
The metastatic pattern varies significantly across molecular subtypes. HR+ tumors demonstrate a strong predilection for bone metastasis, often with prolonged dormancy. HER2-positive disease frequently involves visceral organs such as liver and brain, while triple-negative breast cancer (TNBC) is associated with aggressive dissemination to lungs and central nervous system (Soni et al., 2015). These subtype-specific behaviors underscore the need for diverse and biologically relevant preclinical models.
Preclinical models have therefore become indispensable tools for dissecting the biology of metastasis, identifying therapeutic targets, and evaluating novel treatments. However, each model captures only a subset of the metastatic cascade, and no single system fully reproduces the complexity of human disease. Understanding the strengths and limitations of each model is essential for translating experimental findings into clinical advances.

Read About Breast Cancer on OncoDaily
Methods
This review critically evaluates the spectrum of preclinical models used to study breast cancer metastasis, integrating evidence from experimental oncology, molecular biology, and translational research. The models analyzed include chemically induced systems, genetically engineered mouse models (GEMMs), syngeneic models, xenograft approaches such as CDX and PDX, intraductal transplantation models, zebrafish systems, and emerging humanized platforms.
Each model is assessed based on its ability to recapitulate key biological processes, including tumor initiation, metastatic dissemination, organotropism, tumor microenvironment interactions, immune modulation, and response to therapy. Particular emphasis is placed on clinical relevance, reproducibility, and applicability to precision oncology.
Study Design
This is a comprehensive narrative review synthesizing data across multiple preclinical platforms. Rather than focusing on a single experimental system, the study adopts a comparative framework, evaluating how different models reproduce specific aspects of metastasis biology.
The analysis considers several key domains, including fidelity to human tumor biology, representation of the tumor microenvironment, capacity to model immune interactions, and translational value in drug development. By integrating findings across these domains, the review provides a structured understanding of how preclinical models contribute to advancing breast cancer research.
Results
Mutagen-Induced Models
Chemically induced models represent some of the earliest attempts to replicate breast carcinogenesis in vivo. Agents such as 7,12-dimethylbenz[a]anthracene (DMBA), N-nitrosomethylurea (NMU), and N-ethyl-N-nitrosourea (ENU) induce random mutations that lead to tumor formation, often mimicking hormone receptor-positive breast cancer.
These models offer the advantage of maintaining an intact immune system and allowing tumors to develop within their natural microenvironment. They have been particularly useful in studying hormonal influences, early tumorigenesis, and carcinogen exposure.
However, their limitations are substantial. Tumor development is unpredictable, latency periods are variable, and metastatic spread is relatively rare. Moreover, the mutational landscape generated by chemical carcinogens does not accurately reflect the genomic alterations observed in human breast cancer. Off-target tumor formation further complicates interpretation, reducing their translational applicability.
Genetically Engineered Mouse Models (GEMMs)
GEMMs have revolutionized cancer research by enabling precise genetic manipulation of oncogenes and tumor suppressors. By introducing mutations in genes such as HER2, BRCA1, or p53, researchers can study tumor development in a controlled and physiologically relevant context.
One of the key strengths of GEMMs lies in their ability to model tumor initiation and progression within an intact immune system, preserving interactions between cancer cells and the surrounding microenvironment. These models have provided critical insights into epithelial–mesenchymal transition (EMT), tumor heterogeneity, and early dissemination.
Despite these advantages, GEMMs have important limitations. Many models predominantly produce aggressive tumors that metastasize rapidly, limiting the study of long-term dormancy. Additionally, HR+ tumors are underrepresented, and metastasis often occurs primarily to the lungs rather than the broader spectrum of organs observed in patients. The time and cost associated with generating and maintaining these models further restrict their widespread use.
Syngeneic Models
Syngeneic models involve the transplantation of murine tumor cells into immunocompetent mice of the same genetic background. These models are particularly valuable for studying tumor–immune system interactions and evaluating immunotherapeutic strategies.
The 4T1 model, for example, is widely used due to its ability to spontaneously metastasize to multiple organs, including lungs, liver, bone, and brain. Its rapid growth and reproducibility make it an attractive system for preclinical testing.
However, syngeneic models are heavily biased toward aggressive, triple-negative phenotypes and often rely on artificial routes of tumor cell injection. This bypasses critical early steps of metastasis, such as local invasion and intravasation, limiting their ability to fully recapitulate the metastatic cascade.
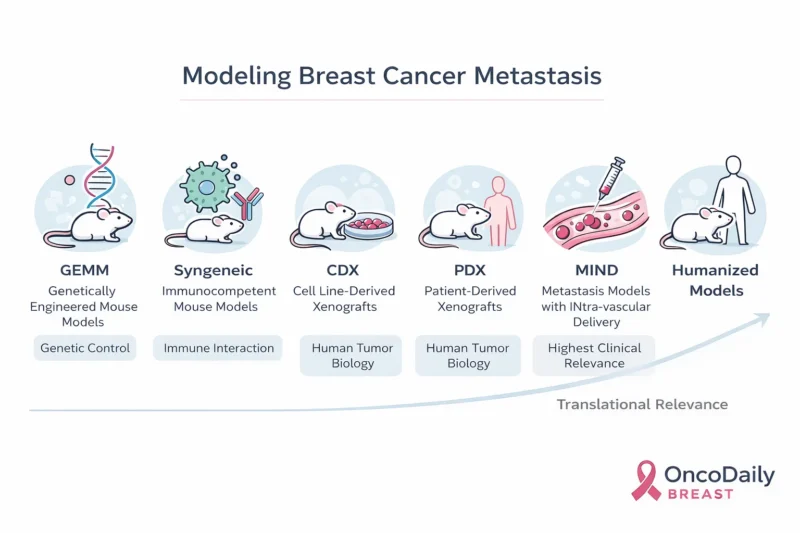
Xenograft Models
Cell Line-Derived Xenografts (CDXs)
CDXs involve the implantation of established human cancer cell lines into immunodeficient mice. These models are widely used due to their reproducibility and ease of use.
They have contributed significantly to our understanding of tumor biology and drug response, particularly through well-characterized cell lines such as MCF-7 and MDA-MB-231. However, the absence of an intact immune system represents a major limitation, as immune interactions play a critical role in metastasis and treatment response.
Additionally, prolonged culture of cell lines leads to genetic drift, reducing their resemblance to primary tumors and limiting their clinical relevance.
Patient-Derived Xenografts (PDXs)
PDXs represent a significant advancement in preclinical modeling by directly implanting tumor tissue from patients into mice. These models preserve tumor heterogeneity, architecture, and genomic characteristics, making them highly valuable for translational research.
PDXs have demonstrated strong predictive value for therapeutic response and are increasingly used in precision oncology. They are particularly useful for studying drug resistance mechanisms and evaluating novel targeted therapies.
Nevertheless, PDX models are not without challenges. Engraftment rates vary widely, particularly for HR+ tumors, and the absence of a functional immune system limits their utility in immunotherapy research. Over time, human stromal components are replaced by murine counterparts, potentially altering tumor behavior.
Intraductal Xenograft Models (MIND)
The mammary intraductal (MIND) model represents one of the most physiologically relevant systems for studying breast cancer. By injecting tumor cells directly into the mammary ducts, this approach preserves the natural architecture and hormonal environment of the breast.
MIND models uniquely recapitulate the full metastatic cascade, including spontaneous dissemination and colonization of distant organs. They also allow for the study of tumor dormancy and subtype-specific metastatic patterns, such as ovarian and gastrointestinal metastases in invasive lobular carcinoma.
This model has emerged as particularly valuable for HR+ breast cancer, addressing a major gap in traditional preclinical systems.
Zebrafish Models
Zebrafish models offer a complementary approach, enabling real-time visualization of tumor cell dissemination and metastasis. Their transparency, rapid development, and low cost make them ideal for high-throughput screening and mechanistic studies.
However, the lack of a mammalian microenvironment and hormonal system limits their ability to model complex interactions observed in human breast cancer. While useful for early-stage research, their translational relevance remains limited.
Humanized Mouse Models
Humanized mouse models represent a promising frontier in cancer research by incorporating human immune components into immunodeficient mice. This allows for the study of tumor–immune interactions and evaluation of immunotherapies in a more clinically relevant context.
These models have demonstrated that immune checkpoint inhibitors, such as PD-1 blockade, require a functional human immune system to exert their effects. However, their complexity, high cost, and technical challenges currently limit widespread adoption.
Key Findings
The analysis highlights that each preclinical model captures distinct aspects of breast cancer metastasis, but none fully replicates the complexity of human disease.
Models with intact immune systems, such as GEMMs and syngeneic systems, are essential for studying tumor–immune interactions, while xenograft-based models provide greater relevance for human tumor biology and therapeutic response. Intraductal models and PDX systems emerge as particularly valuable for bridging the gap between experimental research and clinical application.
A major limitation across models is the inadequate representation of tumor dormancy and long-term metastatic progression, which remain critical challenges in breast cancer management.
Key Takeaway Messages
Preclinical modeling of breast cancer metastasis requires a strategic, question-driven approach rather than reliance on a single system. Different models should be selected based on the biological process under investigation, whether it is tumor initiation, immune interaction, or metastatic colonization.
Among available systems, intraductal xenografts and patient-derived models provide the closest approximation to human disease, particularly for hormone receptor-positive breast cancer. Future advances will depend on integrating immune components and improving microenvironmental fidelity.
Conclusion
Preclinical models have significantly advanced our understanding of breast cancer metastasis, yet critical gaps remain in replicating the full spectrum of human disease. The inability to fully model tumor dormancy, immune interactions, and long-term progression continues to limit translational success.
Emerging approaches, including humanized models and refined intraductal systems, offer promising avenues to overcome these challenges. Ultimately, combining complementary models will be essential to develop more effective therapies and improve outcomes for patients with metastatic breast cancer.
Read full article here


