Trastuzumab deruxtecan (T-DXd) has transformed the treatment landscape of metastatic breast cancer over the past few years. Initially developed for HER2-positive tumors, the antibody–drug conjugate (ADC) has demonstrated clinically meaningful activity across a broader biological spectrum, including HER2-low and even some HER2-0 tumors. This expansion has significantly increased the population eligible for HER2-targeted therapy. However, it has also created a new challenge: conventional HER2 testing methods are no longer sufficient to predict which patients will derive the greatest benefit from T-DXd.
A new study by Tarantino and colleagues, published in npj Precision Oncology, explores whether quantitative profiling of HER2 in tumor tissue and plasma can better predict the activity of T-DXd in metastatic breast cancer (Tarantino et al., 2026). By integrating multiple molecular technologies, including proteomic, transcriptomic, genomic, and liquid biopsy platforms, the investigators demonstrate that more precise measurements of HER2 expression may significantly improve the prediction of treatment outcomes.
This work offers an important glimpse into the future of precision oncology in breast cancer, where biomarker interpretation may move beyond categorical HER2 scoring toward continuous and dynamic biological profiling.
Read About Trastuzumab Deruxtecan on OncoDaily
Why Trastuzumab Deruxtecan Requires Better Biomarkers
T-DXd has shown remarkable efficacy in multiple pivotal clinical trials. In the DESTINY-Breast03 trial, T-DXd significantly improved progression-free survival compared with trastuzumab emtansine in HER2-positive metastatic breast cancer (Cortés et al., 2024). Later, the DESTINY-Breast04 study expanded the indication to HER2-low tumors, demonstrating substantial survival benefits compared with chemotherapy (Modi et al., 2022). More recently, DESTINY-Breast06 further explored the drug in hormone receptor–positive disease with very low HER2 expression (Curigliano et al., 2024).
Despite these advances, a fundamental limitation remains. Standard HER2 assessment relies on immunohistochemistry (IHC) and in situ hybridization to classify tumors into discrete groups such as HER2-positive, HER2-low, or HER2-0. These categories were originally designed to identify tumors driven by HER2 amplification and to guide trastuzumab-based therapies. However, ADCs such as T-DXd function through a more complex mechanism involving HER2 binding, internalization, and intracellular release of a cytotoxic topoisomerase I inhibitor payload.
Because of this mechanism, the degree of HER2 expression may matter more than whether a tumor simply crosses the threshold of HER2 positivity. Subtle differences in protein abundance, gene expression, or tumor biology may influence the amount of ADC internalized and therefore the therapeutic effect.
Real-World Evidence Of T-DXd Activity In Metastatic Breast Cancer
The study retrospectively analyzed 191 patients with metastatic breast cancer treated with T-DXd. This cohort reflects real-world clinical practice rather than a highly selected trial population.
Before starting T-DXd therapy, 66% of patients had HER2-positive disease, 23% had HER2-low disease, and 11% had HER2-0 tumors. Patients were heavily pretreated, having received a median of four prior metastatic treatment lines and two prior chemotherapy regimens. Median follow-up was 16.6 months (Tarantino et al., 2026).
Clinical outcomes confirmed meaningful activity of T-DXd across HER2 subgroups, but survival durations were somewhat shorter than those reported in pivotal clinical trials. Median overall survival was 23.1 months in HER2-positive disease, 14.2 months in HER2-low disease, and 9.5 months in HER2-0 disease.
Time to next treatment (TTNT), which reflects how long patients remain on therapy before requiring another treatment, also varied by HER2 status. These results highlight an important clinical reality: outcomes observed in routine practice may differ from those seen in controlled clinical trials due to differences in patient populations, treatment histories, and disease biology.
The Dynamic Nature Of HER2 Expression In Metastatic Disease
One of the most intriguing findings of the study concerns the temporal evolution of HER2 expression.
HER2-low tumors are not biologically static. Previous research has shown that HER2 expression can change between the primary tumor and metastatic lesions or between different metastatic biopsies (Tarantino et al., 2022; Miglietta et al., 2021).
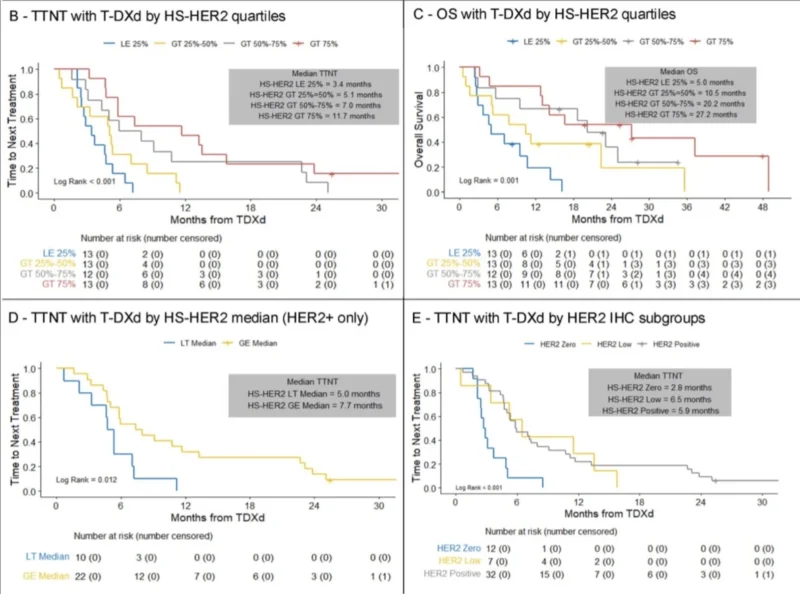
In this study, patients whose tumors maintained HER2-low status from the primary tumor to metastatic disease experienced the longest TTNT with T-DXd, with a median duration of 9.4 months. In contrast, patients whose tumors shifted from HER2-low to HER2-0 experienced much shorter TTNT of approximately 3 months.
These findings suggest that stable HER2-low expression may represent a biologically consistent population more likely to benefit from T-DXd. Conversely, tumors with fluctuating HER2 expression may have less dependency on the HER2 pathway, potentially limiting the effectiveness of HER2-targeted ADC therapy.
High-Sensitivity HER2 Protein Quantification Improves Outcome Prediction
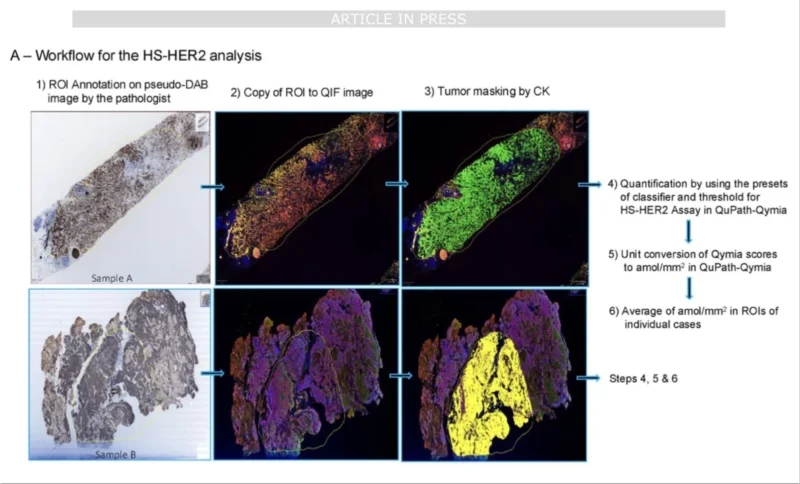
To address the limitations of standard HER2 IHC, the investigators employed a high-sensitivity quantitative HER2 assay (HS-HER2). This technique measures HER2 protein levels using immunofluorescence and converts signal intensity into precise protein concentrations.
Among 51 patients with available tissue samples, HS-HER2 levels were strongly associated with both TTNT and overall survival. As HER2 protein expression increased, clinical outcomes improved. When patients were divided into quartiles based on HER2 expression levels, those with the highest HER2 concentrations experienced the longest duration of benefit from T-DXd.
In contrast, conventional HER2 IHC categories performed poorly in predicting outcomes within the same cohort. Patients classified as HER2-low sometimes achieved outcomes comparable to those with HER2-positive disease, demonstrating the limitations of categorical HER2 scoring.
These findings suggest that quantitative protein measurement may capture biologically relevant differences in HER2 expression that are missed by standard pathology testing.
Proteomic Analysis Reveals Additional Biological Insights
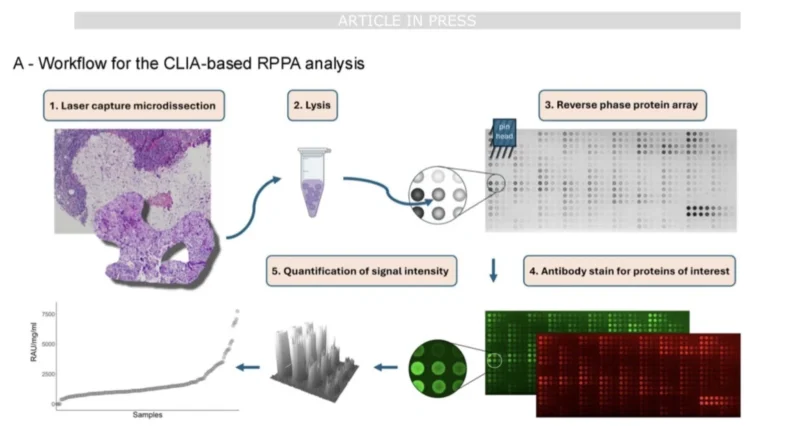
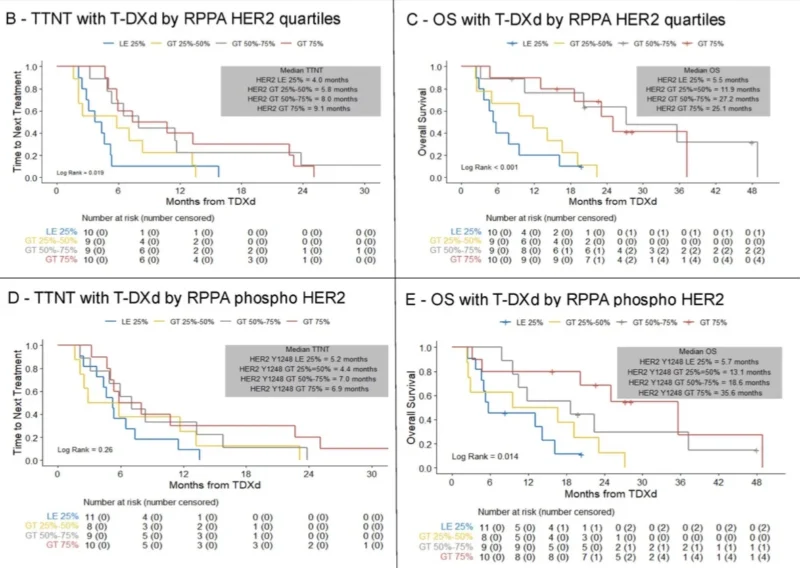
The investigators further explored HER2 signaling using reverse phase protein array (RPPA) technology, a proteomic platform capable of simultaneously measuring multiple proteins and post-translational modifications.
In 38 tumor samples analyzed with RPPA, higher HER2 protein levels and increased HER2 activation, measured through phosphorylation at tyrosine residue Y1248, were associated with improved survival outcomes. These findings support the hypothesis that both HER2 abundance and pathway activation influence T-DXd activity.
Interestingly, the analysis also evaluated proteins related to the ADC payload. T-DXd delivers a topoisomerase I inhibitor, prompting the investigators to examine the expression of TOPO1 and SLFN11, two markers associated with DNA damage response.
In patients with HER2-negative tumors, higher TOPO1 expression was paradoxically associated with worse outcomes with T-DXd. Although this observation requires validation in larger cohorts, it suggests that payload-related biology may influence ADC efficacy independently of HER2 expression.
Importantly, proteins unrelated to T-DXd targeting, such as Trop2, phosphorylated EGFR, and phosphorylated HER3, did not correlate with treatment outcomes, supporting the specificity of the HER2-related findings.
Transcriptomic HER2 Signatures Provide Additional Predictive Value
The study also examined HER2 gene expression using the HER2DX assay, which analyzes multiple gene signatures related to HER2 signaling, immune activity, tumor proliferation, and luminal differentiation.
Among 41 tumor samples analyzed, higher expression of the HER2 amplicon gene module, which includes ERBB2, GRB7, STARD3, and TCAP, was significantly associated with longer TTNT and overall survival. ERBB2 mRNA expression alone also correlated with improved outcomes.
Other gene modules within HER2DX, including immune-related and proliferation signatures, did not demonstrate strong predictive value. These results reinforce the central importance of HER2 signaling intensity rather than broader tumor biology in determining T-DXd responsiveness.
Liquid Biopsy May Offer A Non-Invasive Biomarker Strategy
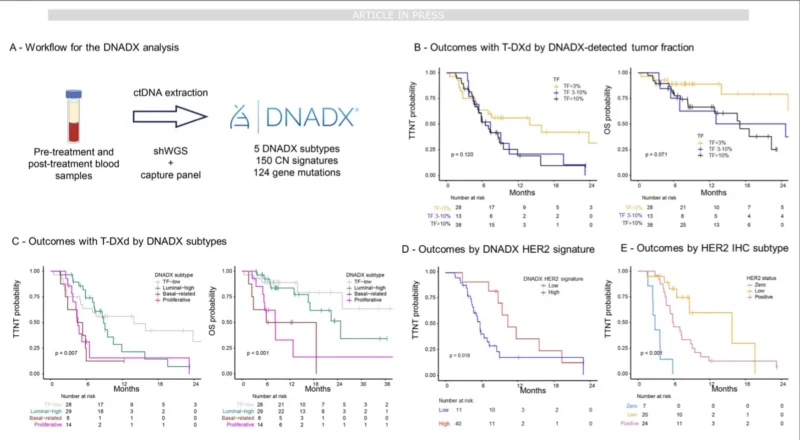
While tissue-based assays provide valuable biological information, they require tumor biopsies that may be difficult or risky to obtain. For this reason, the investigators also evaluated circulating tumor DNA (ctDNA) using the DNADX platform.
The analysis included 140 plasma samples from 98 patients. Higher tumor DNA fraction in plasma was associated with worse outcomes, reflecting the known prognostic significance of ctDNA burden.
More importantly, a plasma-based HER2 signature derived from DNADX showed significant association with T-DXd activity. Patients with higher HER2 signature scores experienced longer TTNT compared with those with low scores.
This finding suggests that liquid biopsy could potentially serve as a non-invasive method for predicting T-DXd response, allowing clinicians to monitor tumor biology dynamically during treatment.
Genomic Alterations Associated With Resistance
Genomic analysis also provided insight into potential resistance mechanisms.
Among patients with HER2-negative metastatic breast cancer, approximately 15% harbored ERBB2 heterozygous loss. These patients demonstrated numerically shorter TTNT and overall survival compared with those without the alteration.
Baseline TP53 mutations were also associated with poorer outcomes, while mutations in ARID1B appeared enriched in ctDNA after T-DXd treatment, suggesting possible therapy-driven clonal evolution.
Although these findings require further validation, they provide important clues about the molecular mechanisms that may influence sensitivity or resistance to ADC therapy.
What This Study Means For Clinical Practice
This study highlights several important lessons for oncologists managing metastatic breast cancer.
First, HER2 expression should be viewed as a continuous biological variable rather than a simple categorical classification. Quantitative assays measuring HER2 protein or gene expression may offer more accurate prediction of ADC activity.
Second, HER2 status can evolve over time. Rebiopsy or longitudinal monitoring may provide additional insights into tumor biology and treatment sensitivity.
Third, liquid biopsy technologies may soon allow non-invasive prediction of ADC efficacy through ctDNA-based HER2 signatures.
Finally, ADC activity likely depends not only on target expression but also on payload sensitivity and tumor genomic context.
The Future Of Precision Oncology In The ADC Era
The expansion of antibody–drug conjugates across oncology has outpaced the development of predictive biomarkers. As more ADCs enter clinical practice, the need for more sophisticated biomarker strategies will become increasingly urgent.
This study represents one of the most comprehensive attempts to address that challenge for T-DXd. By integrating proteomic, transcriptomic, genomic, and liquid biopsy approaches, the investigators demonstrate that quantitative HER2 profiling can meaningfully predict treatment outcomes.
If validated in larger prospective studies, these approaches could reshape how oncologists select and sequence ADC therapies in metastatic breast cancer.
In the future, treatment decisions may no longer rely solely on HER2-positive or HER2-low classifications. Instead, clinicians may use integrated molecular profiling to determine the degree of HER2 dependency, identify resistance mechanisms, and tailor therapy accordingly.
For patients with metastatic breast cancer, such advances could translate into more personalized treatment strategies and improved outcomes.
Full article is available in Nature


