For muscle-invasive bladder cancer (MIBC), the standard approach is radical cystectomy plus cisplatin-based neoadjuvant chemotherapy. However, ~50% of patients are ineligible for cisplatin or choose to decline it. Both pembrolizumab and sacituzumab govitecan have shown activity as neoadjuvant monotherapy, so SURE-02 trial tested the combination within a perioperative strategy that could allow bladder preservation in selected patients.
Methods
Design: Single-arm, phase 2, single center (IRCCS San Raffaele Hospital, Milan, Italy).
Population: Adults with cT2–T3bN0M0 MIBC, ECOG 0–1, who were cisplatin-ineligible or refused cisplatin, and initially scheduled for cystectomy.
Treatment plan:
Neoadjuvant (4 cycles, q3w):
- Pembrolizumab 200 mg IV day 1
- Sacituzumab govitecan 7.5 mg/kg IV day 1 and day 8
- Then radical cystectomy, or for patients refusing cystectomy after MDT discussion: repeat TURBT (re-TURBT)
- Adjuvant/maintenance pembrolizumab: 13 cycles, 200 mg IV q3w after surgery (or after re-TURBT strategy)
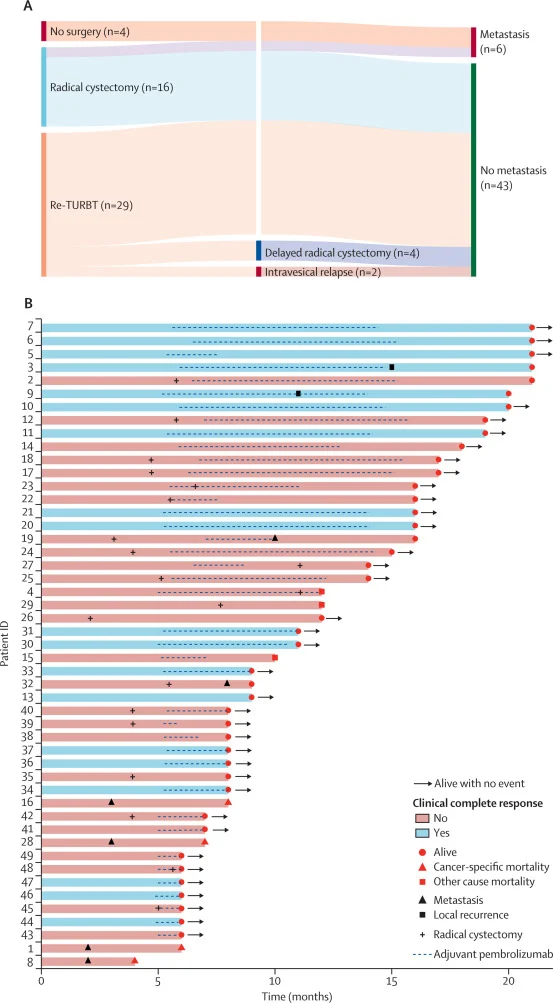
Primary endpoint: Clinical complete response (cCR) — defined for bladder-sparing patients as negative imaging + no viable tumor on re-TURBT.
Findings
- Enrollment: 49 patients (median age 66; 84% male)
- Tumor stage: 67% cT2
- Variant histology: 43% had centrally confirmed variant features
- Median follow-up: 14 months
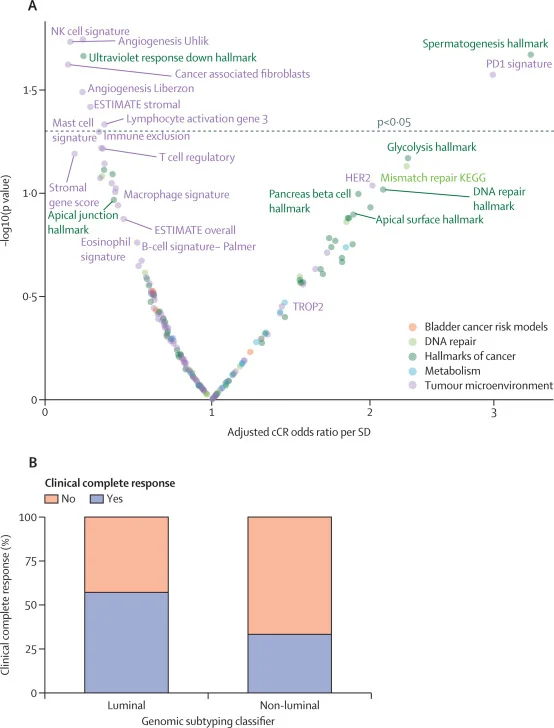
Clinical complete response (primary endpoint):
- 19/49 patients = 39% cCR (95% CI 25–54)
- All cCR patients underwent re-TURBT
- All cCR patients remained metastasis-free at cutoff
- Intravesical relapse: 2 patients had bladder relapse (local recurrence)
Safety Outcomes
Treatment-related toxicity was generally manageable. Grade 3 adverse events were observed in 16% of patients, with diarrhea representing the most frequently reported severe event (8%). Importantly, no grade 4 or grade 5 treatment-related toxicities and no treatment-related deaths were recorded. Serious treatment-related adverse events occurred in 6% of patients, consisting of two cases of bullous pemphigoid and one case of immune-mediated colitis.
Interpretation
This perioperative regimen (sacituzumab govitecan + pembrolizumab, followed by pembrolizumab) produced a promising ~40% clinical complete response rate with no grade ≥4 toxicity, supporting a feasible bladder-preservation pathway in a subset of patients who are cisplatin-ineligible or refuse cisplatin.
You Can Read Full Article Here.


