Deficient mismatch repair (dMMR) colorectal cancers are among the most immunotherapy-sensitive solid tumors, with immune checkpoint blockade producing remarkable responses in metastatic disease and increasingly in localized settings. While dual-checkpoint approaches such as nivolumab plus ipilimumab have demonstrated exceptionally high pathologic response rates, important clinical questions remain regarding the optimal duration, intensity, toxicity profile, and practicality of neoadjuvant immunotherapy.
The RESET-C trial explored whether a simplified strategy—a single preoperative cycle of pembrolizumab—could induce meaningful tumor regression while maintaining feasibility and minimizing overtreatment in patients with localized dMMR colon cancer.
Study Design and Methods
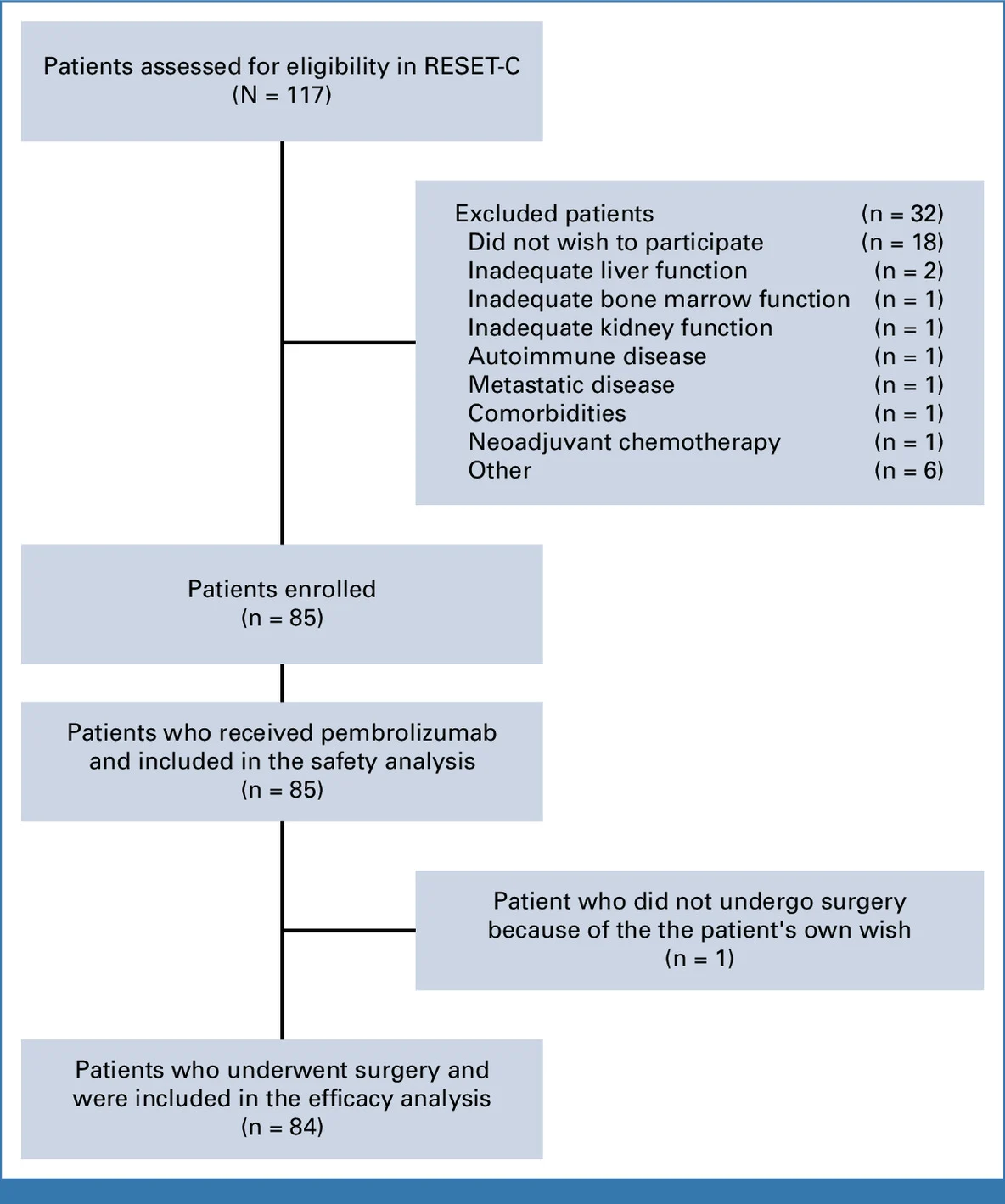
RESET-C was a Danish multicenter phase II trial enrolling 85 patients with stage I–III localized dMMR colon cancer planned for curative surgery.
Patients received a single cycle of pembrolizumab (4 mg/kg, maximum 400 mg), followed by repeat endoscopy with biopsies and surgery approximately 3–5 weeks later. The primary endpoint was pathologic complete response (pCR), while secondary endpoints included major pathologic response (MPR), safety, disease-free survival, overall survival, ctDNA dynamics, and endoscopic response assessment.
The study population reflected a real-world elderly dMMR cohort, with a median age of 74 years and 60% of patients presenting with stage III disease.
Results
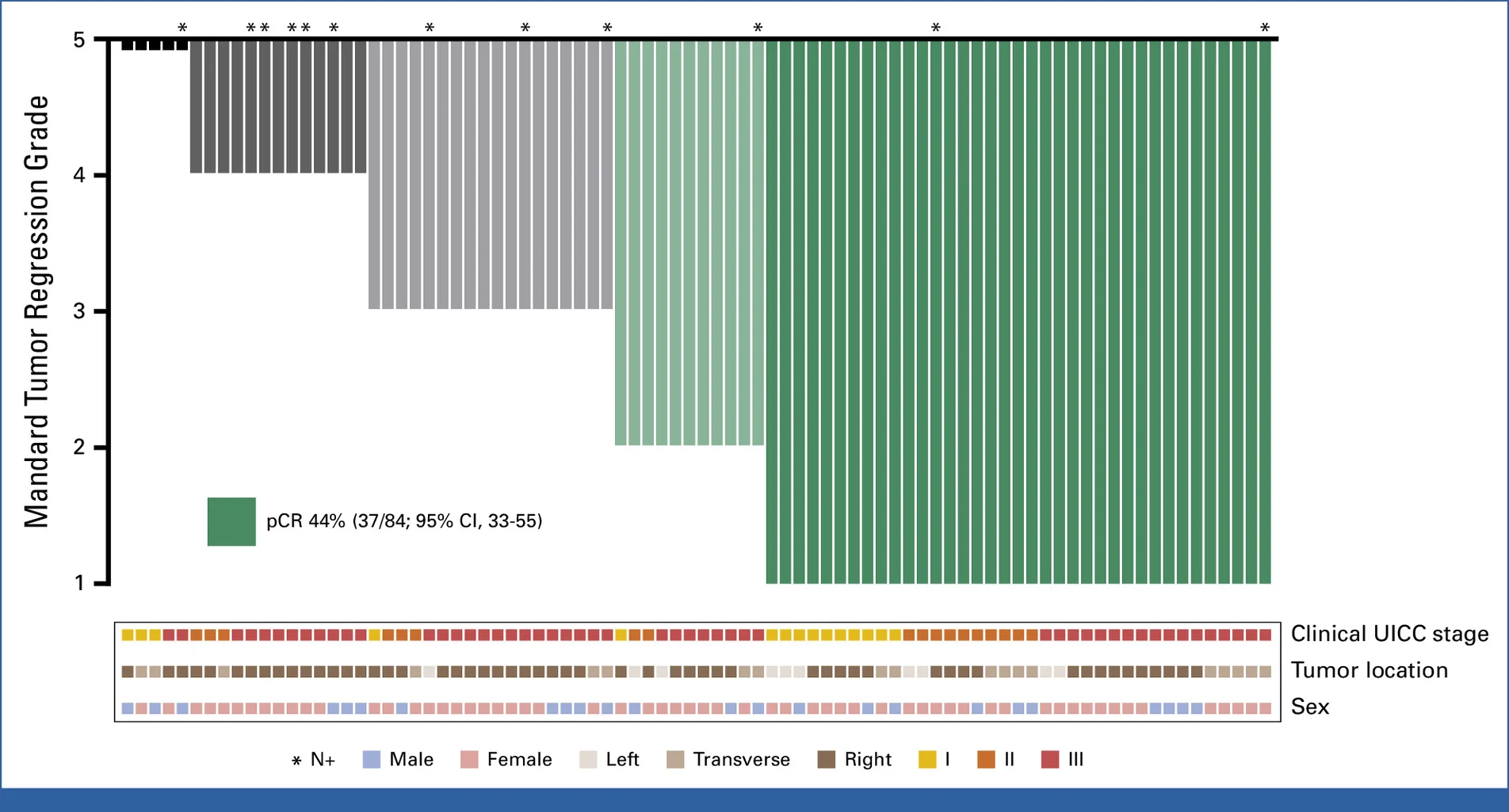
RESET-C demonstrated that even a single dose of pembrolizumab could induce profound tumor regression in localized dMMR colon cancer.
- Pathologic complete response (pCR): achieved in 44% of patients (37/84), while major pathologic response (MPR) occurred in 57%
- Stage-dependent efficacy: pCR rates reached 67% in stage I disease, 56% in stage II disease, and 33% in stage III disease
- Survival outcomes were highly encouraging: at a median follow-up of 18.4 months, overall survival was 98% and disease-free survival was 96%
- Treatment-related toxicity remained manageable: grade 3 adverse events occurred in 11% of patients, with only three grade 3 events considered pembrolizumab-related and no grade 4–5 immune-related toxicities observed
- Endoscopic response assessment showed strong predictive value: endoscopic imaging predicted pCR with 77% sensitivity, 93% specificity, and 86% overall accuracy, while the positive predictive value reached 90%
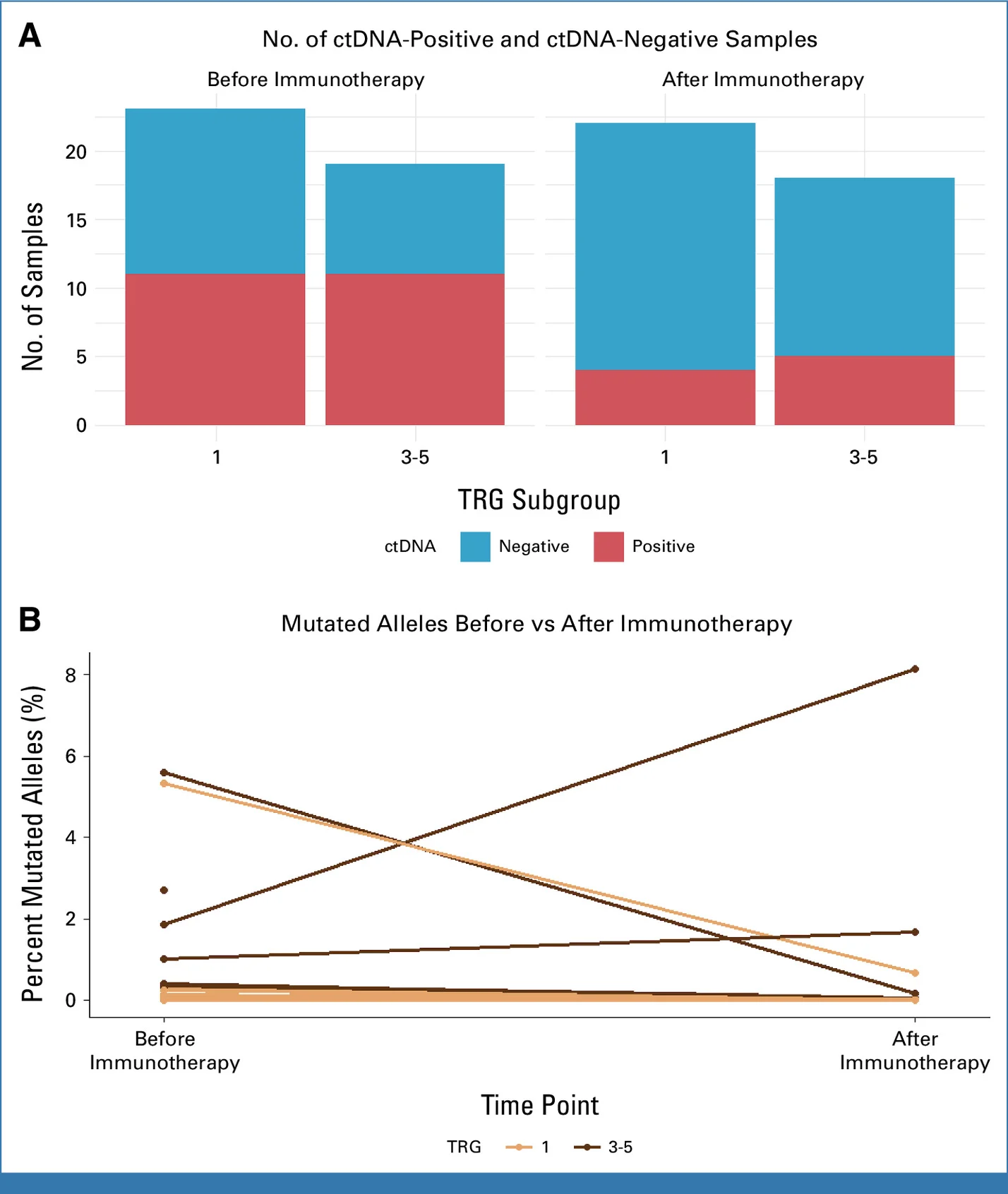
Importantly, ctDNA analyses demonstrated biologic activity after only one cycle of therapy, with ctDNA positivity decreasing from 52.4% pre-treatment to 22.5% post-treatment, supporting a rapid systemic immune response.
Scientific Interpretation
RESET-C is important because it demonstrates that meaningful pathologic regression can occur after only a single cycle of PD-1 blockade in localized dMMR colon cancer. This challenges the assumption that prolonged or dual-agent immunotherapy is always necessary to achieve deep responses.
Although pCR rates were lower than those reported in dual-checkpoint studies such as NICHE-2, the simplified single-agent strategy offers potential advantages in toxicity, logistics, accessibility, and cost-effectiveness—particularly for older patients or those with significant comorbidities.
One of the most clinically relevant findings was the strong performance of centralized endoscopic response assessment. Accurate prediction of pCR remains one of the greatest barriers to organ-preservation strategies in colon cancer. The high specificity and positive predictive value observed in RESET-C suggest that advanced endoscopic evaluation may become an essential component of future nonoperative management pathways.
The trial also highlights an important paradox in modern colorectal immunotherapy: surgery-related morbidity may become increasingly difficult to justify in patients achieving profound immune-mediated tumor eradication. Two patients died from postoperative complications despite excellent oncologic outcomes overall, emphasizing the urgent need to evaluate carefully selected watch-and-wait approaches in future studies.
Clinical Implications
RESET-C strengthens the growing movement toward response-adapted treatment strategies in dMMR colorectal cancer.
The study supports universal mismatch repair testing at diagnosis and reinforces the concept that even short-duration immunotherapy can produce major biologic and clinical responses in localized disease.
At the same time, the findings raise several important future questions:
- Can surgery safely be omitted in selected patients with convincing clinical complete response?
- Is single-agent PD-1 blockade sufficient for lower-stage disease?
- Which biomarkers best identify patients requiring more intensive therapy?
- What is the optimal interval between immunotherapy and surgery?
These questions are likely to define the next generation of neoadjuvant immunotherapy trials in colorectal cancer.
Key Takeaway Message
RESET-C demonstrates that a single cycle of neoadjuvant pembrolizumab can induce substantial pathologic regression in localized dMMR colon cancer, with nearly half of patients achieving pCR and excellent early survival outcomes. The study also provides important evidence supporting endoscopic response assessment as a potential foundation for future organ-preserving and watch-and-wait strategies in dMMR colon cancer.
You Can Read Full Article Here