Immune checkpoint inhibitors have changed the treatment landscape for advanced non-small cell lung cancer, but major gaps remain in selecting the right patients and in tracking whether treatment is truly working. Established tools such as PD-L1 expression and RECIST-based imaging response often fail to align well with long-term survival, particularly in immunotherapy-treated disease. A new multicenter study now introduces a potentially important solution: the QVT score in NSCLC immunotherapy, a radiomic biomarker derived from routine CT scans that captures tumor vascular complexity and may help predict and monitor benefit from immune checkpoint blockade (Chae et al., 2026).
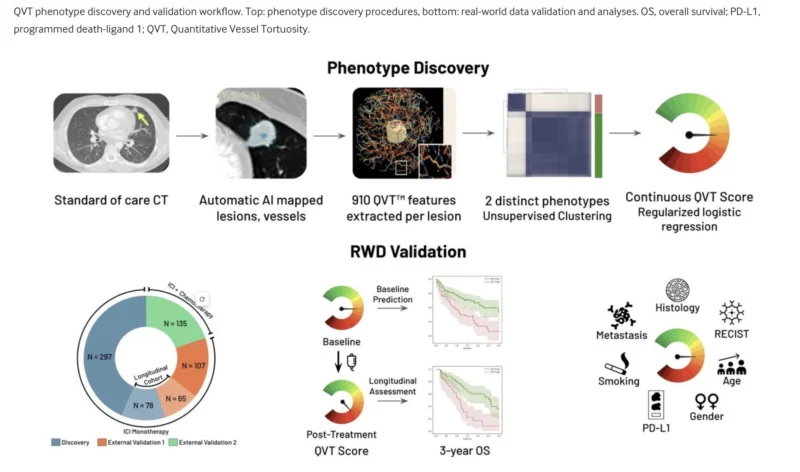
The study, evaluated 1,301 CT scans from 682 patients with ICI-treated NSCLC across multiple institutions. Investigators developed the Quantitative Vessel Tortuosity, or QVT, score as an automated, biologically interpretable imaging biomarker that reflects tumor-associated vascular architecture. Their central premise was that abnormal, chaotic tumor vasculature promotes immune evasion and treatment resistance, and that this biology can be quantified non-invasively through standard CT imaging (Chae et al., 2026).
Copyright © 2026, The Author(s). Published by BMJ Publishing Group Ltd. on behalf of Society for Immunotherapy of Cancer.
Why Tumor Vasculature Matters In Immunotherapy
Disorganized tumor vasculature is not just a structural feature of cancer. It actively shapes the tumor microenvironment. Abnormal vessel growth can worsen hypoxia, restrict immune-cell infiltration, and reinforce immune suppression, all of which may reduce the effectiveness of immune checkpoint inhibitors. This makes vascular complexity an appealing biological target for prognostic biomarker development, especially in NSCLC, where not all patients with high PD-L1 expression derive durable benefit from immunotherapy alone (Chae et al., 2026).
This is where the QVT score in NSCLC immunotherapy becomes especially interesting. Unlike many radiomic models that rely on abstract image patterns or black-box deep learning outputs, QVT was designed around a biologically meaningful concept: the complexity of tumor-associated vessels. The score summarizes 910 vessel-based radiomic features related to curvature, torsion, branching, vessel radius, vascular volume, and inflection points. In practical terms, it attempts to measure how ordered or chaotic the vascular network surrounding a tumor appears on CT (Chae et al., 2026).
Copyright © 2026, The Author(s). Published by BMJ Publishing Group Ltd. on behalf of Society for Immunotherapy of Cancer.
Study Design And Cohorts
This retrospective multicenter study included 682 patients with advanced NSCLC who received immune checkpoint inhibitors either alone or with chemotherapy. The dataset was divided into several cohorts.
The discovery cohort included 375 patients treated with ICI monotherapy. This cohort was used to identify vascular phenotypes and to build the continuous QVT score. External validation then tested the biomarker in two separate settings: an ICI monotherapy cohort of 172 patients and a chemoimmunotherapy cohort of 135 patients. A longitudinal cohort of 143 patients with both pretreatment and early on-treatment CT scans was used to evaluate whether changes in QVT score during therapy correlated with survival (Chae et al., 2026).
Across cohorts, mean age ranged from 64.6 to 69.2 years. Adenocarcinoma was the most common histology, accounting for 65.3% of the discovery cohort, 68.0% of the ICI monotherapy validation cohort, and 66.7% of the chemoimmunotherapy cohort. PD-L1 data were incomplete in many patients, with unknown expression in 50.7% of the discovery cohort and 37.8% of the ICI monotherapy validation cohort, highlighting a real-world challenge that reinforces the potential value of the QVT score in NSCLC immunotherapy as an adjunctive biomarker (Chae et al., 2026).
Two Distinct Vascular Phenotypes Emerged
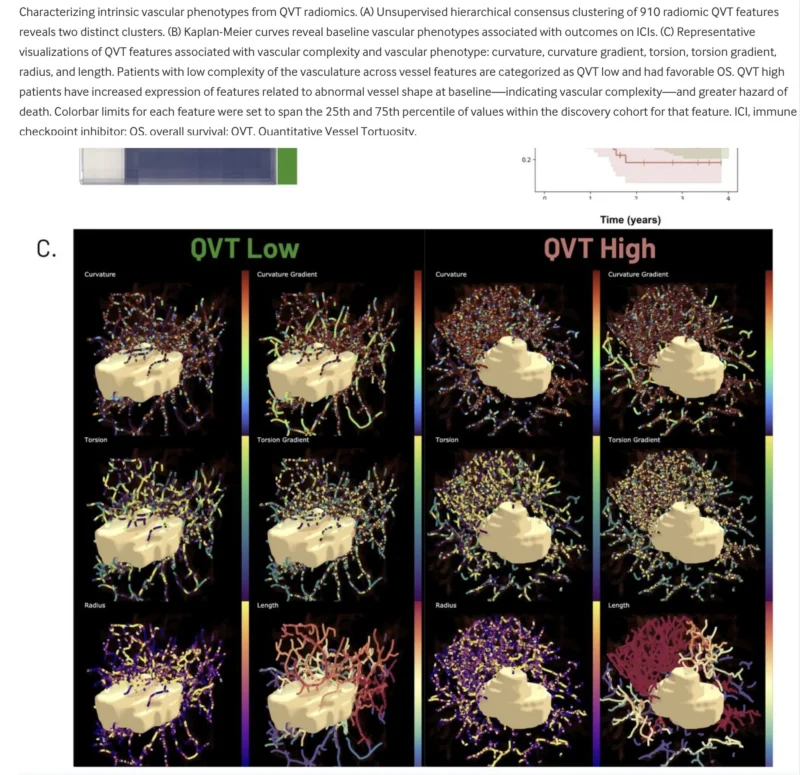
Using unsupervised clustering of all 910 QVT features in the discovery cohort, investigators identified two major vascular phenotypes. One phenotype, present in 21.6% of patients, showed highly complex, chaotic vascular architecture and was labeled “QVT High.” The other, representing 78.4% of patients, showed more normalized and ordered vascular structure and was labeled “QVT Low” (Chae et al., 2026).
These phenotypes were not merely descriptive. They were clinically meaningful. The QVT High phenotype was associated with significantly worse overall survival in the discovery cohort, with a hazard ratio of 1.65 and a p value of 0.04. This initial finding provided the biological and statistical foundation for deriving the continuous QVT score in NSCLC immunotherapy, which places patients along a spectrum between these two vascular states (Chae et al., 2026).
Copyright © 2026, The Author(s). Published by BMJ Publishing Group Ltd. on behalf of Society for Immunotherapy of Cancer.
Development Of The Continuous QVT Score
To create a clinically usable biomarker, the researchers trained a regularized logistic regression model using the full set of QVT features to distinguish the two vascular phenotypes. The resulting continuous score ranged from 0 to 1, with higher values indicating more abnormal and complex vascularity. A threshold of 0.695 best separated the phenotype groups in the discovery set (Chae et al., 2026).
Importantly, this model appeared highly robust. In fivefold cross-validation, it achieved a mean AUC of 0.9993 and a mean accuracy of 0.9813 for identifying the clustering-derived phenotype groups. The study also demonstrated practical feasibility: the cloud-based automated pipeline generated the QVT score from CT images in an average of 28.6 minutes, including DICOM ingestion, segmentation, feature extraction, and score computation (Chae et al., 2026).
This automation is one of the most attractive features of the QVT score in NSCLC immunotherapy, since it suggests that the biomarker could potentially be integrated into routine workflow without requiring additional invasive testing.
Copyright © 2026, The Author(s). Published by BMJ Publishing Group Ltd. on behalf of Society for Immunotherapy of Cancer.
Prognostic Value In ICI Monotherapy And Chemoimmunotherapy
The next question was whether the biomarker could predict outcomes in external datasets. It did.
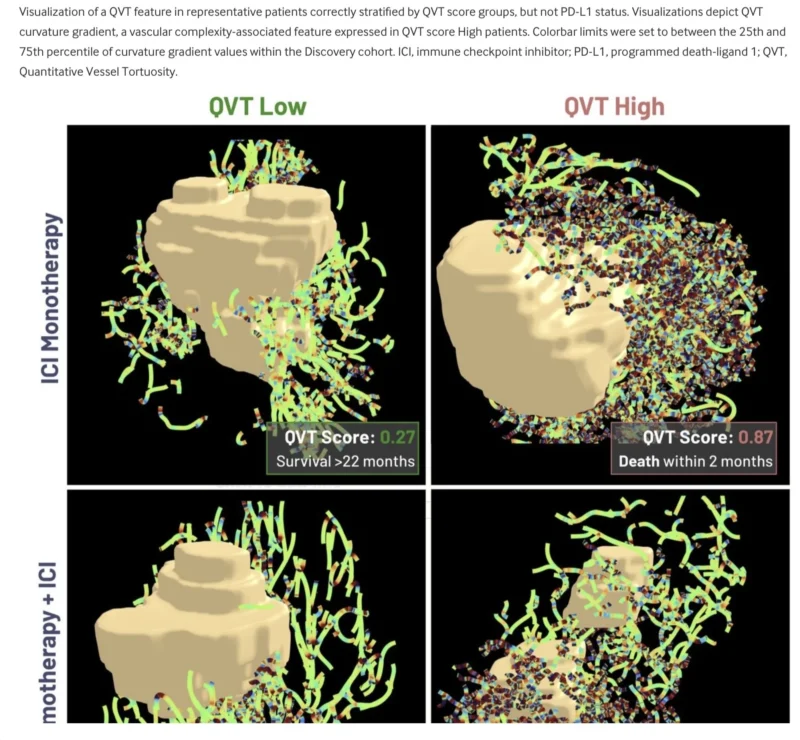
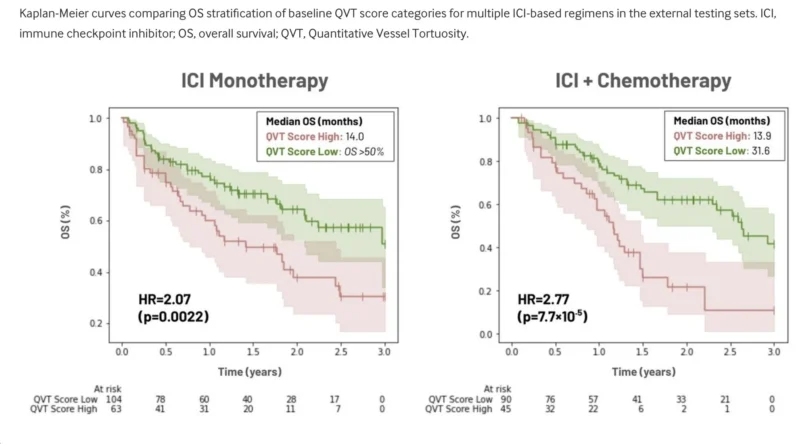
In the external ICI monotherapy validation cohort, the continuous QVT score was significantly associated with overall survival, with a hazard ratio of 1.17 per 0.1 increase in score and a p value of 0.0028. Patients classified as QVT High had significantly worse survival than QVT Low patients, with a hazard ratio of 2.07 and a p value of 0.0022. Median overall survival was 14.0 months in the QVT High group, while median overall survival was not reached in the QVT Low group within the 3-year analysis window (Chae et al., 2026).
The results were even stronger in the chemoimmunotherapy cohort. There, the continuous QVT score in NSCLC immunotherapy showed a hazard ratio of 1.23 per 0.1 score increase, with a p value of 4.9×10⁻⁵. The QVT High group had a hazard ratio of 2.77 compared with the QVT Low group, with a p value of 7.7×10⁻⁵. Median overall survival was 13.9 months for QVT High patients versus 31.6 months for QVT Low patients (Chae et al., 2026).
When all validation patients were combined, the QVT High group had a hazard ratio of 2.26, and median overall survival was 14.0 months versus 35.1 months for QVT High versus QVT Low groups, respectively (Chae et al., 2026).
Independent Of PD-L1 And Clinical Variables
One of the most important findings was that the QVT biomarker remained independently prognostic after adjustment for baseline variables including PD-L1 expression. In multivariable analyses, the QVT High group retained significance across treatment settings, with adjusted hazard ratios ranging from 2.13 to 2.38 and p values of 0.002 or less (Chae et al., 2026).
That matters because current biomarkers often fall short in clinical practice. PD-L1 helps guide treatment selection, but it does not consistently predict durable benefit. The QVT score in NSCLC immunotherapy may therefore complement PD-L1 by capturing a different biological dimension: the vascular environment that shapes immune accessibility and response.
Early On-Treatment Changes Also Predicted Survival
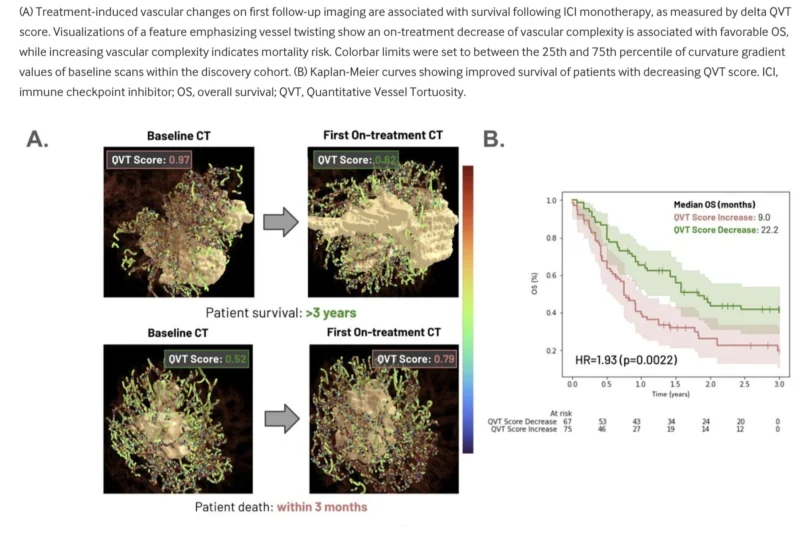
The study also evaluated whether QVT could serve as a dynamic monitoring biomarker. In the longitudinal cohort of 143 patients, early on-treatment changes in score were significantly associated with overall survival.
An increase in QVT score after treatment initiation was associated with worse survival, with a hazard ratio of 1.11 per 0.1 increase and a p value of 0.012. When patients were grouped by median score change, those with increasing QVT score had significantly worse survival than those with decreasing score, with a hazard ratio of 1.93 and a p value of 0.0022. Median overall survival was 9.0 months in the increasing-QVT group versus 22.0 months in the decreasing-QVT group (Chae et al., 2026).
This suggests that falling QVT score may reflect vascular normalization during therapy, a biologically favorable process that corresponds with better long-term outcomes. Notably, these associations remained significant even after accounting for tumor volume change and RECIST best overall response. In multivariable comparisons, longitudinal QVT change retained prognostic significance with hazard ratios of 1.55 to 1.84 depending on the comparator model, while RECIST and tumor volume were also significant but did not eliminate the independent contribution of the QVT score in NSCLC immunotherapy (Chae et al., 2026).
Copyright © 2026, The Author(s). Published by BMJ Publishing Group Ltd. on behalf of Society for Immunotherapy of Cancer.
What This Means For Precision Oncology
This study positions QVT as more than another radiomics experiment. Its strength lies in combining interpretability, automation, and applicability across multiple immunotherapy regimens. Because it uses standard CT imaging and a biologically grounded model, it may be more clinically deployable than many previous radiomic or deep learning approaches.
For clinicians, the potential uses are clear. Baseline QVT score may help identify patients at higher risk of poor outcomes despite ICI-based treatment. Early changes in QVT may offer a way to detect benefit or resistance before conventional radiographic response becomes obvious. For researchers and drug developers, QVT may also serve as a non-invasive endpoint for studying angiogenesis-related biology and treatment-induced vascular remodeling (Chae et al., 2026).
The Bottom Line
The QVT score in NSCLC immunotherapy is a novel CT-based imaging biomarker that captures tumor vascular complexity, predicts survival across ICI monotherapy and chemoimmunotherapy, and tracks early treatment-related vascular remodeling associated with outcome. In this multicenter study, higher baseline QVT score consistently identified patients with worse overall survival, while early decreases in score were linked to improved survival independent of RECIST response and tumor volume change (Chae et al., 2026).
If validated further in prospective trials, QVT could help fill one of the most important unmet needs in thoracic oncology: a practical, non-invasive biomarker that improves both risk stratification and early response monitoring in immunotherapy-treated NSCLC.
Read full article here