Immune checkpoint inhibitors have fundamentally reshaped the treatment of advanced malignancies, yet durable benefit remains limited to a subset of patients. Increasing evidence indicates that this variability cannot be explained by tumor genomics alone. Host factors, particularly the gut microbiome, have emerged as major determinants of response and resistance to immunotherapy. Experimental and translational studies have shown that microbial composition can influence antigen presentation, T-cell activation, cytokine signaling, and the broader immune tone of the tumor microenvironment. In some settings, fecal microbiota transplantation from ICI responders has even restored sensitivity in previously refractory disease, underscoring the causal relevance of the microbiome in anti-tumor immunity.
Against this background, concomitant medications that perturb the gut microbiome have become a major area of interest in immuno-oncology. Among them, proton pump inhibitors (PPIs) are particularly important because they are widely prescribed, often chronically used, and frequently taken without strong oncologic consideration. PPIs are known to alter gastric acidity and thereby reshape intestinal microbial ecology. However, although retrospective studies have suggested that PPI exposure may correlate with poorer outcomes in ICI-treated patients, the underlying biologic basis has remained insufficiently defined, and the interaction with other treatments—especially chemotherapy—has not been fully characterized.
This study addressed that gap by examining whether PPI exposure is associated with inferior survival in patients treated with ICIs and whether this association is accompanied by measurable changes in the gut microbiome and peripheral immune cell composition.
Gut Microbiome as a Predictor of Dual Checkpoint Inhibition Benefit in Non–Small Cell Lung Cancer
Study Design
The investigators used a multi-cohort strategy that combined large-scale retrospective clinical analysis with smaller prospective translational cohorts.
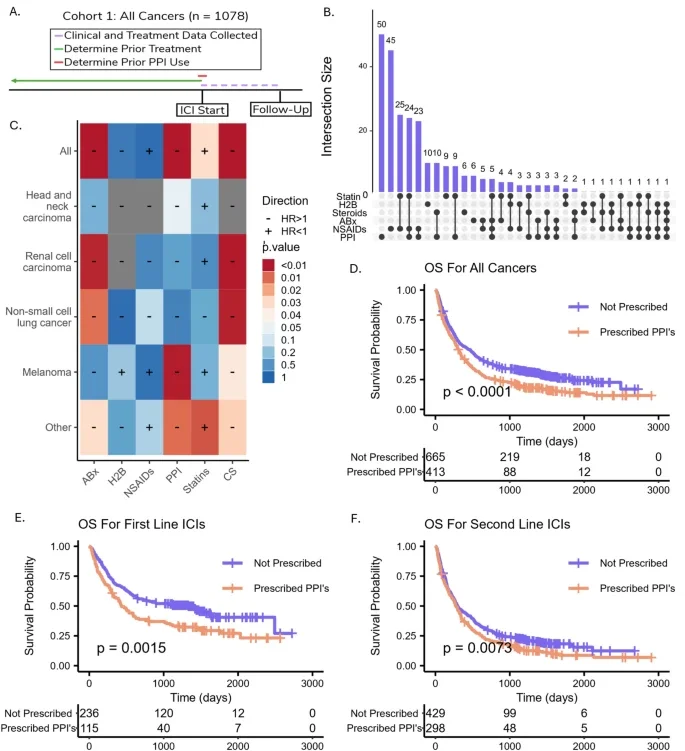
The first and largest cohort included 1,078 patients with advanced cancers treated with ICIs between 2011 and 2019. This retrospective dataset was used to evaluate the relationship between PPI exposure and overall survival. Patients were classified as PPI users if a PPI was listed as an active medication at the time of ICI initiation, based on electronic medical record extraction combined with detailed manual chart review. Importantly, the study captured both prescription and over-the-counter PPI use through provider medication reconciliation, improving exposure assessment. Survival analyses were then stratified by line of therapy, prior chemotherapy exposure, and timing of chemotherapy relative to ICI initiation.
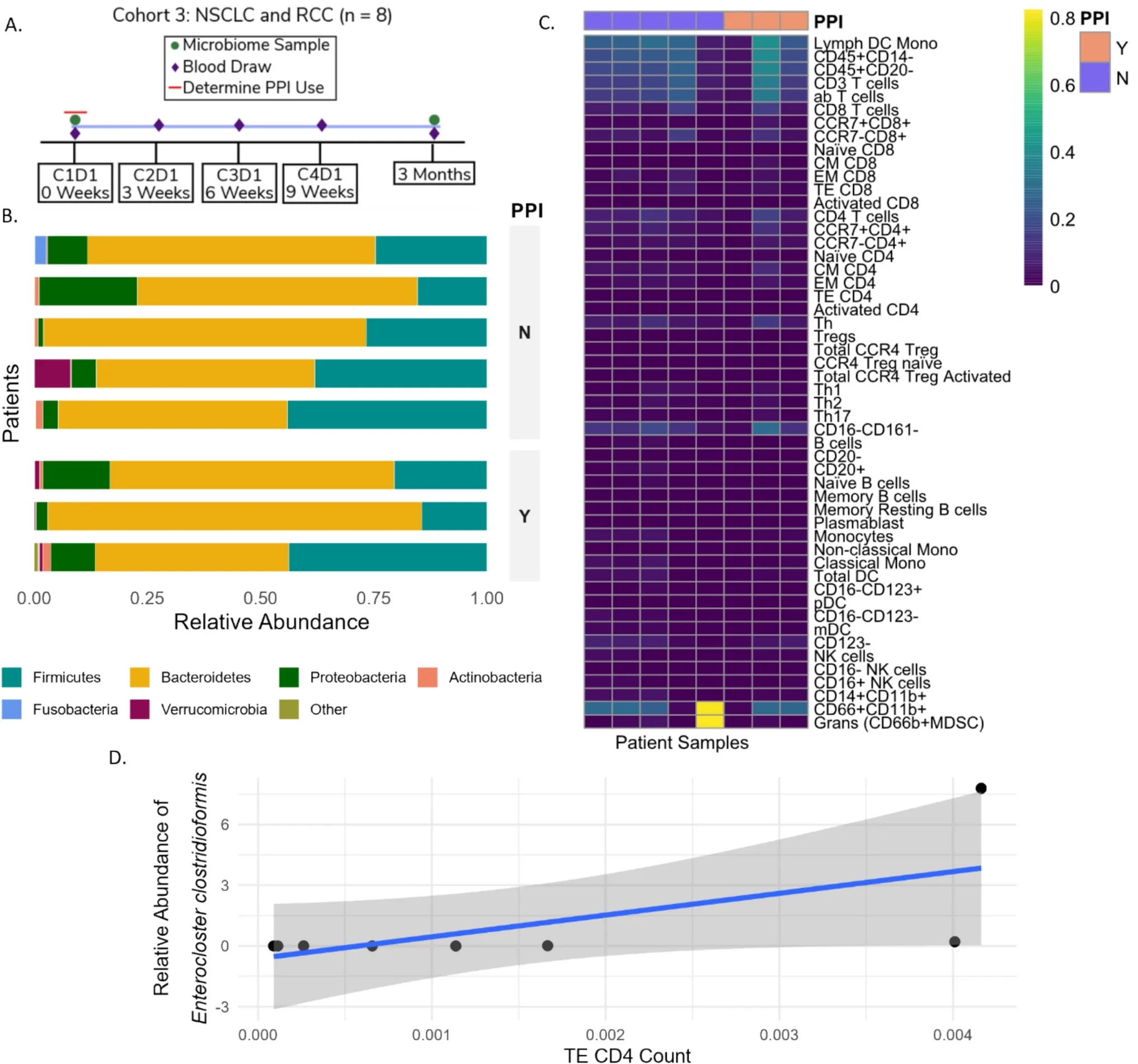
To move beyond purely clinical associations, the authors then evaluated two prospective translational cohorts. One cohort included 42 patients with metastatic melanoma receiving ICIs, in whom stool samples were analyzed using metagenomic sequencing to assess microbiome diversity and taxonomic composition. A third exploratory cohort included 8 patients with NSCLC or renal cell carcinoma, in whom both stool and serial blood samples were available. In this cohort, metagenomic profiling of the microbiome was combined with CyTOF-based immune phenotyping to explore whether PPI-associated microbial signatures correlated with systemic immune cell populations.
This design allowed the study to ask three increasingly mechanistic questions: whether PPIs are associated with worse survival, whether PPIs are associated with altered microbiome composition, and whether those microbiome changes might relate to immune-cell differences relevant to immunotherapy efficacy.
Clinical Results: PPI Use Was Associated With Inferior Survival
In the large retrospective cohort, 38% of patients were taking a PPI at the start of immunotherapy, underscoring how common this exposure is in routine oncology practice. Across tumor types, PPI use was associated with significantly shorter overall survival, with a hazard ratio of 1.39. This means that, overall, PPI exposure was associated with a 39% relative increase in the risk of death among patients treated with ICIs.
This adverse association was not confined to a single treatment context. It remained evident in:
- first-line immunotherapy, where the hazard ratio was 1.57
- second-line and later-line immunotherapy, where the hazard ratio was 1.25
Thus, the signal was present regardless of whether ICIs were being used early or later in the treatment course.
The effect also appeared to vary by tumor type, with the strongest association observed in melanoma. This is biologically notable because melanoma has been one of the malignancies in which microbiome-immunotherapy links have been most consistently demonstrated in prior research.
The Effect of PPIs Was Modulated by Prior Chemotherapy
One of the most interesting aspects of this study is that the negative impact of PPIs was not uniform across all patients. Prior chemotherapy appeared to substantially modulate this association.
Among patients who had not received prior chemotherapy, PPI use was clearly associated with shorter overall survival, with a hazard ratio of 1.57. Among those who had received prior chemotherapy, the association was weaker but still present overall, with a hazard ratio of 1.20.
However, when the authors examined the timing of chemotherapy, an important distinction emerged:
- If the most recent chemotherapy had been given more than 1 year before starting ICI, PPI use remained significantly associated with worse survival, with a hazard ratio of 1.79
- If chemotherapy had been given within 1 year before ICI initiation, the association between PPI use and survival was no longer significant, with a hazard ratio of 1.09
This finding suggests that recent chemotherapy may alter the microbiome so profoundly that the additional perturbation associated with PPIs no longer exerts a distinct measurable effect. In other words, chemotherapy may partially “flatten” the microbiome-related survival differences that would otherwise be seen with PPI exposure.
This is an important concept because it implies that the biologic effect of PPIs is context-dependent, not simply binary. Their impact may depend on the pre-existing state of the host microbiome and on whether other treatments have already disrupted it.
PPIs Appeared More Relevant Than H2 Blockers
The study also examined H2 blockers, another class of acid-suppressive medications. This comparison is clinically relevant because it raises the question of whether all acid suppression is equally problematic, or whether the effect is specific to PPIs.
The results suggested that PPIs had a stronger and more consistent negative association with survival than H2 blockers. H2-blocker use was not significantly associated with overall survival across the major analyses, including first-line and later-line ICI settings and subgroup analyses based on chemotherapy exposure.
Although the study does not prove that H2 blockers are safer alternatives, it does suggest that the negative signal is not merely due to acid suppression in general, but may relate specifically to the broader microbiome-disruptive effects of PPIs.
Multivariable Analysis and Confounding
Because retrospective analyses are vulnerable to confounding, the investigators tested whether the PPI-survival association remained after adjustment for clinically relevant covariates.
PPI use remained significantly associated with worse survival when adjusting for:
- Cancer stage
- Charlson Comorbidity Index (CCI)
This strengthens the argument that the association is not simply a reflection of sicker patients being more likely to receive PPIs.
However, when the analysis adjusted for antibiotic exposure, the statistical significance of the PPI association was lost. This is highly informative biologically. Antibiotics are among the strongest known disruptors of the microbiome and have repeatedly been linked to poorer immunotherapy outcomes. The fact that antibiotic exposure attenuated the PPI signal suggests that these medications may converge on overlapping biologic pathways, most likely involving microbiome disruption.
This does not invalidate the PPI finding. Rather, it places it within a larger framework of microbiome-sensitive concomitant medications that may collectively shape response to immunotherapy.
Microbiome Findings in the Melanoma Cohort
The prospective melanoma cohort was used to assess whether PPI exposure was accompanied by detectable differences in gut microbial composition.
At baseline, both PPI users and non-users had gut communities dominated by Firmicutes and Bacteroidota, which is broadly typical. Interestingly, there were no significant differences in alpha diversity or beta diversity between groups. This means that PPIs were not associated with a global loss of diversity or large-scale restructuring of community architecture in this cohort.
However, when the investigators looked more closely at individual taxa, they identified specific compositional shiftsassociated with PPI use. PPI users showed relative enrichment of:
- Lactobacillaceae
- Veillonella
In contrast, non-users showed relative enrichment of:
- Clostridiaceae
- Bacteroides
- and some Bifidobacterium species, which were absent in the PPI group
These findings are important because microbiome effects on immunotherapy often depend not on overall diversity alone, but on the relative abundance of functionally important organisms. Several taxa depleted in PPI users have been implicated in more favorable ICI responses in prior studies, whereas some enriched taxa, such as Veillonella, have been linked to inflammatory or dysbiotic states.
Thus, even in the absence of major diversity changes, PPI exposure appears to shift the microbiome toward a composition that may be less supportive of optimal anti-tumor immunity.
Exploratory Immune Correlates
The NSCLC/RCC cohort allowed the investigators to go one step further and explore whether microbiome changes associated with PPIs might correspond to measurable changes in circulating immune populations.
This cohort was very small, so the results must be interpreted cautiously. No major group-level differences in immune-cell abundance were detected between PPI users and non-users. That said, in exploratory correlation analyses, the abundance of Enterocloster clostridioformis was significantly associated with the abundance of terminal effector CD4 T cells.
This is a provocative observation because prior preclinical work has linked Enterocloster clostridioformis to immune-suppressive effects after microbiome perturbation, including the promotion of regulatory immune states and reduced sensitivity to PD-1 blockade. The present study does not prove that the same mechanism is operating here, but it provides a translational signal consistent with that broader biologic model.
The implication is that PPI-associated microbial changes may not be biologically neutral. They may influence the balance of circulating immune subsets in ways that could affect responsiveness to checkpoint inhibition.
Limitations
The study has several important limitations.
The core survival analysis was retrospective and observational, so causality cannot be established. Residual confounding remains possible despite multivariable adjustment.
The prospective microbiome and immune cohorts were relatively small, especially the NSCLC/RCC cohort, limiting statistical power and making the translational findings exploratory.
All cohorts were derived from a single institution, which may limit generalizability.
Lifestyle factors known to influence the microbiome, such as diet, were not systematically controlled.
And although over-the-counter PPI exposure was captured through chart review, underreporting remains possible.
These limitations mean the findings should be interpreted as strongly hypothesis-generating rather than definitive proof of causation.
Key Takeaway Messages
- PPI use was associated with significantly shorter overall survival in patients with advanced cancers treated with ICIs.
- This effect was observed in both first-line and later-line immunotherapy settings.
- The association was strongest in melanoma and was attenuated in patients who had received chemotherapy within 1 year before ICI initiation.
- H2 blockers did not show the same negative association, suggesting drug-class specificity rather than a generic acid-suppression effect.
- PPI use was associated with distinct taxonomic shifts in the gut microbiome, including enrichment of Lactobacillaceae and Veillonella and depletion of taxa such as Bifidobacterium.
- Exploratory data suggested that PPI-associated microbes may correlate with circulating immune-cell subsets, supporting a microbiome–immune mechanism.
- These findings reinforce the need to consider concomitant medications as biologically relevant variables in immunotherapy-treated patients.
Conclusion
This study offers an important and increasingly relevant message for immuno-oncology: common supportive medications may meaningfully influence the efficacy of immune checkpoint inhibition.
PPIs were associated with inferior survival in ICI-treated patients, and this association appears to be linked to shifts in gut microbiome composition and, potentially, to downstream immune-cell changes. The effect was not uniform across all settings, but instead was modulated by prior chemotherapy exposure, emphasizing that microbiome-related determinants of immunotherapy response are dynamic and treatment-context dependent.
You Can Read All Article Here.