Dual immune checkpoint inhibition with ipilimumab plus nivolumab (I–N) represents an established therapeutic strategy for advanced non–small cell lung cancer (NSCLC), capable of producing durable responses even in tumors with low PD-L1 expression. However, clinical benefit remains restricted to a subset of patients, and the optimal integration of chemotherapy into dual immunotherapy regimens continues to represent a major unresolved clinical challenge.
Current treatment selection largely relies on tumor-derived biomarkers, particularly PD-L1 expression, which incompletely captures response heterogeneity. Increasing evidence suggests that systemic host factors — particularly the gut microbiome — play a critical role in shaping antitumor immunity through modulation of T-cell activation, antigen presentation, and inflammatory signaling pathways.
This prospective study investigated whether baseline gut microbiota composition could function as a biologically meaningful determinant of response to dual checkpoint blockade and inform the need for chemotherapy intensification.
Study Design and Translational Framework
In this multicenter prospective analysis, patients with advanced or recurrent NSCLC received:
- Ipilimumab + nivolumab (I–N)
or - I–N combined with chemotherapy
Baseline fecal samples underwent 16S rRNA gene sequencing to quantify microbial diversity and taxonomic composition. Parallel translational analyses incorporated multiplex immunofluorescence evaluation of tumor-infiltrating immune populations, enabling integrated assessment of:
- Microbiome ecology
- Tumor immune microenvironment
- Clinical outcomes
Efficacy endpoints included objective response rate (ORR), progression-free survival (PFS), and overall survival (OS), analyzed alongside microbiome-derived parameters using multivariable and inverse probability weighting approaches.
Clinical Outcomes
Across the overall cohort:
- ORR: 44%
- Disease control rate: 63%
- Median PFS: 5.8 months
- Median OS: 24.8 months
Although addition of chemotherapy increased response rates numerically (59% vs 24%), survival differences between treatment strategies were not uniformly observed in unselected patients, underscoring biological heterogeneity beyond treatment intensity alone.
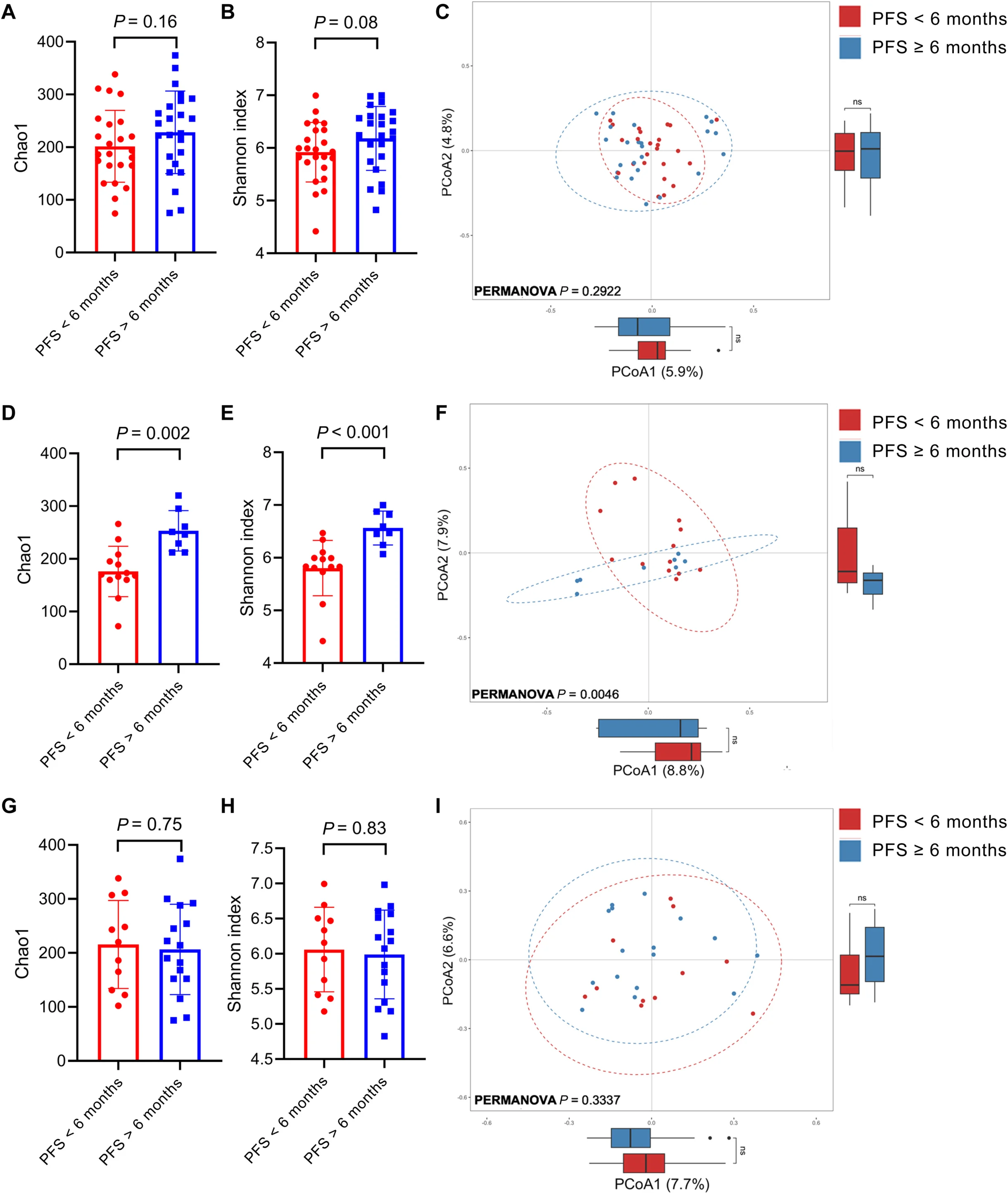
Microbiome Diversity as a Determinant of Immunotherapy Sensitivity
A central finding of the study was that baseline gut microbiota diversity selectively stratified benefit from dual checkpoint blockade.
Among patients treated with I–N alone, high α-diversity was associated with:
- Significantly prolonged PFS
- Increased intratumoral CD8⁺ T-cell infiltration
- Enrichment of PD-1⁺CD8⁺ effector populations
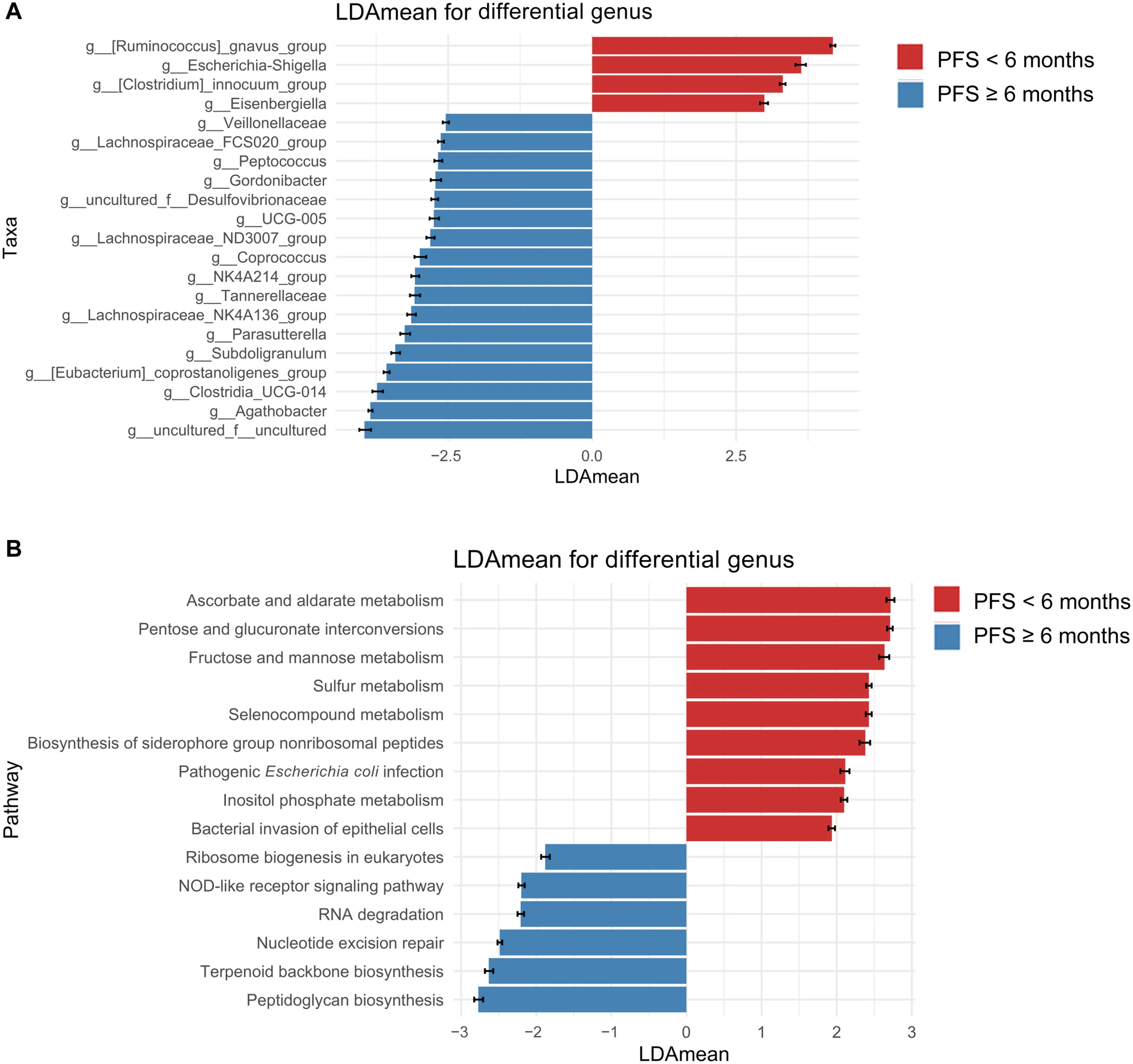
Responder profiles demonstrated expansion of short-chain fatty acid (SCFA)–producing commensal taxa, including members of Lachnospiraceae, Eubacterium, and Agathobacter, previously implicated in immune-metabolic activation and interferon-γ–mediated cytotoxic responses.
Notably, this microbiome–outcome association disappeared when chemotherapy was added, suggesting that cytotoxic therapy may partially overcome microbiome-dependent immune resistance mechanisms.
Chemotherapy as a Compensatory Immune Modulator
Inverse probability–weighted analyses demonstrated that:
- Patients with low microbiome diversity derived significant PFS benefit from chemo-immunotherapy.
- Patients with high diversity showed no clear survival advantage from chemotherapy addition.
These findings support a biologically coherent model in which chemotherapy functions as an immune-restorative intervention, potentially through induction of immunogenic cell death, enhanced antigen release, and dendritic cell activation in microbiome-impaired immune environments.
Thus, microbiome status may represent a previously unrecognized determinant of chemotherapy necessity within immunotherapy-based regimens.
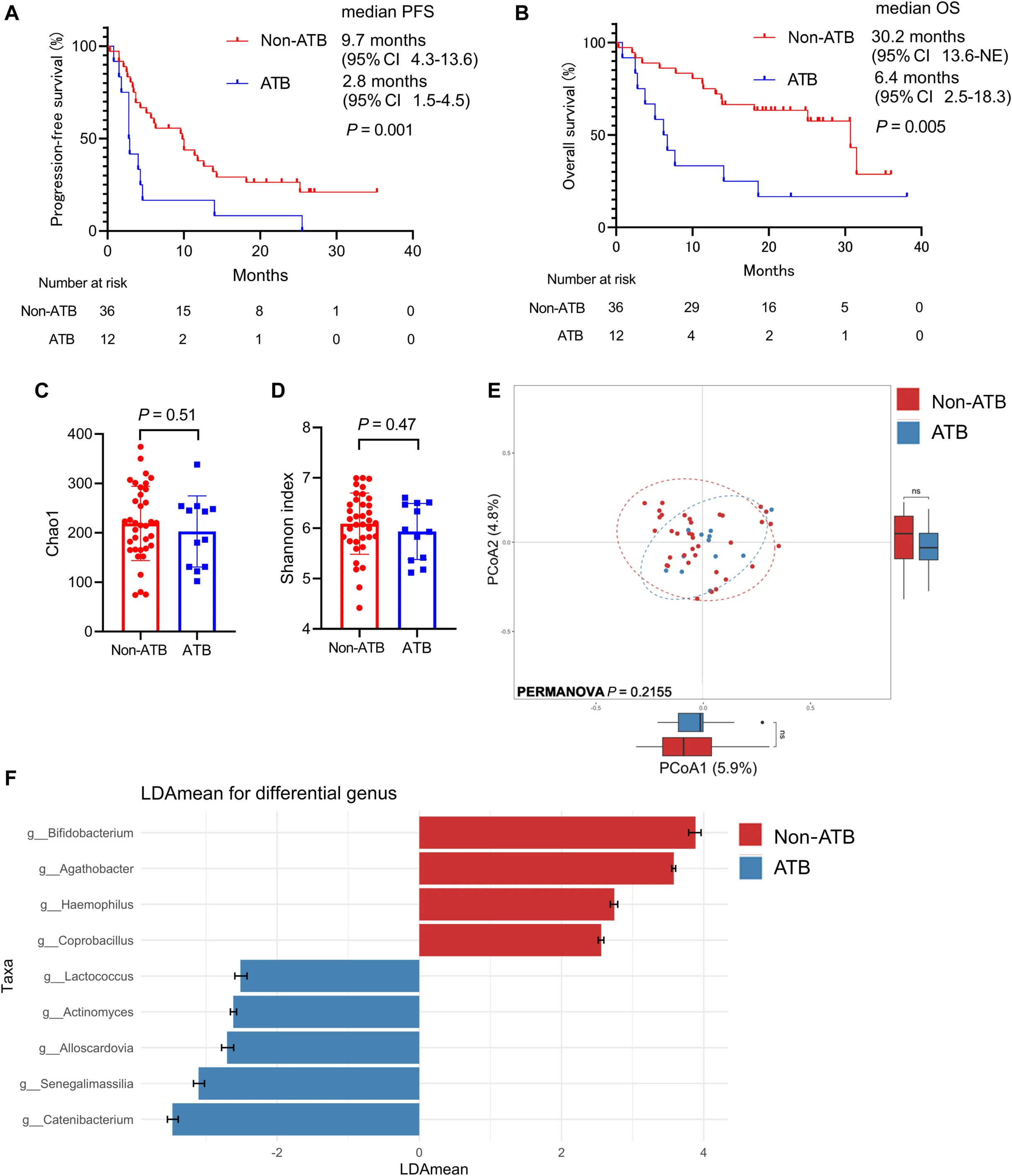
Antibiotic Exposure and Immunotherapy Resistance
Consistent with emerging literature, antibiotic exposure within 30 days prior to treatment initiation independently correlated with inferior PFS and OS.
Importantly, this effect occurred despite preserved global diversity indices, suggesting that functional microbial disruption rather than diversity loss alone may impair antitumor immunity.
These observations reinforce the concept that peri-treatment microbiome perturbation represents a clinically relevant modifier of checkpoint inhibitor efficacy.
Biological Integration: The Gut–Tumor Immune Axis
Mechanistically, SCFA-producing microbiota appear to enhance antitumor immunity through:
- Metabolic support of CD8⁺ T-cell function
- Promotion of IFN-γ–mediated cytotoxic activity
- Activation of NOD-like receptor signaling
- Reduction of regulatory T-cell–mediated immunosuppression
Conversely, dysbiosis-associated taxa linked to intestinal inflammation were enriched among non-responders and associated with immune exhaustion phenotypes.
Collectively, these findings position the gut microbiome as an upstream regulator of tumor immune competence.
Clinical Implications
This study challenges the conventional escalation paradigm in advanced NSCLC management.
Rather than universally intensifying therapy, results suggest that treatment selection may evolve toward host-informed immunotherapy sequencing, where microbiome profiling helps determine:
- When dual checkpoint blockade alone is sufficient
- When chemotherapy is required to overcome immune resistance
Such an approach aligns with the broader transition from single biomarker models toward systems-level precision oncology.
Key Takeaway Messages
- Gut microbiome diversity independently modulates response to dual PD-1/CTLA-4 blockade in NSCLC.
- High microbial diversity identifies patients capable of achieving benefit from immunotherapy without chemotherapy escalation.
- Chemotherapy may compensate for microbiome-associated immune dysfunction in low-diversity states.
- Antibiotic exposure negatively impacts outcomes, supporting microbiome preservation as a therapeutic consideration.
- Microbiome profiling may emerge as a clinically actionable biomarker guiding immunotherapy–chemotherapy integration.
You Can Read Full Article Here