Pancreatic cancer remains one of the most challenging malignancies to treat, particularly in the metastatic setting, where immunotherapy has historically shown minimal activity. However, a biologically defined subset of patients with homologous recombination deficiency (HRD), especially those harboring BRCA1, BRCA2, or PALB2 mutations, may be more sensitive to DNA-damaging agents and potentially to immunotherapy-based strategies.
In this context, the POLAR trial explored whether combining PARP inhibition with PD-1 blockade could provide durable disease control in patients who had already benefited from platinum-based chemotherapy.
The study was published in Nature Medicine on March 25, 2026.
Title: Pembrolizumab and olaparib in homologous-recombination-deficient metastatic pancreatic cancer: the phase 2 POLAR trial
Authors: Wungki Park, Catherine A. O’Connor, Joanne F. Chou, Marc Hilmi, Zeynep Tarcan, Carly Schwartz, Mary Larsen, Ramzi Homsi, Karthigayini Sivaprakasam, Shigeaki Umeda, Maria A. Perry, Anna M. Varghese, Kenneth H. Yu, Fiyinfolu Balogun, Alice Zervoudakis, Seth S. Katz, Tae-Hyung Kim, Ken Zhao, Allison L. Richards, Nicolas Lecomte, Daniel Martin Muldoon, Elias Karnoub, Walid Chatila, Jessica Yang, Imane El-Dika, Devika Rao, Smita Joshi, Michael B. Foote, Ryan Sugarman, James J. Harding, Andrew S. Epstein, David Kelsen, Sree Chalassani, Fergus Keane, Joshua D. Schoenfeld, Anupriya Singhal, Erin Diguglielmo, Chaitanya Bandlamudi, Junmin Song, Hulya Sahin Ozkan, Junguei Hong, Haochen Zhang, Agustin III Cardenas, Maria Lao, Jerry Melchor, Ronak Shah, Wenfei Kang, Francesca Mazzoni, Kevin Soares, Mark TA Donoghue, Ernesto Santos, Vineet Rolston, Marsha Reyngold, Alice Chia-chi Wei, Murray Tipping, Olca Basturk, Michael Berger, Richard Kihn Do, Mark Schattner, William R. Jarnagin, Nadeem Riaz, Vinod Balachandran, Dana Pe’er, Marinela Capanu, Christine Iacobuzio-Donahue, and Eileen M. O’Reilly
Methods
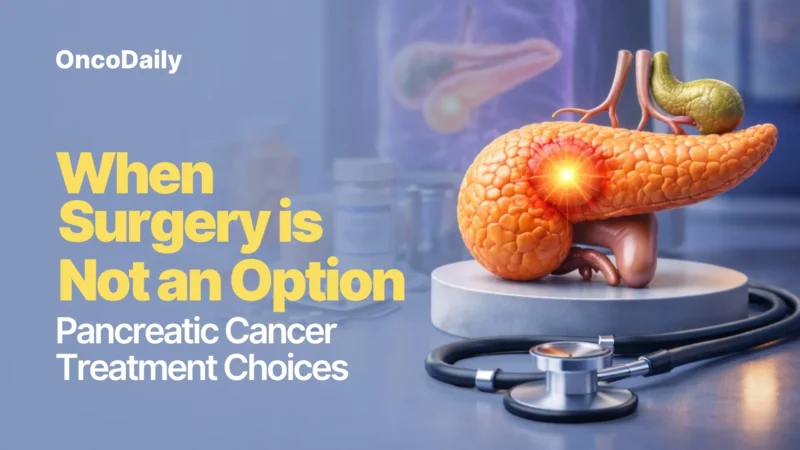
POLAR is a phase 2, single-center, non-randomized basket trial evaluating maintenance pembrolizumab in combination with olaparib in patients with metastatic pancreatic cancer who had achieved at least stable disease after platinum-based chemotherapy.
A total of 63 patients were enrolled and prospectively assigned to three biomarker-defined cohorts. Cohort A included patients with core HRD mutations (BRCA1, BRCA2, or PALB2), cohort B included patients with non-core HRD mutations (such as ATM, CHEK2, and others), and cohort C included patients without HRD alterations but with platinum-sensitive disease.
All patients received olaparib 300 mg twice daily and pembrolizumab administered intravenously every 3 weeks for 6 months, followed by every 6 weeks until disease progression or unacceptable toxicity.
The co-primary endpoints in cohort A were an objective response rate of at least 43% and a 6-month progression-free survival rate of at least 77%.
Results
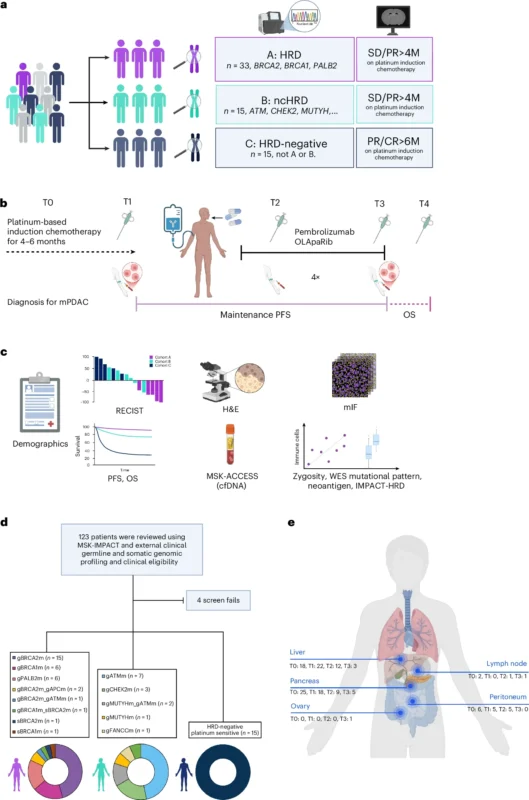
At the time of analysis, 63 patients had been treated, and 46 were evaluable by RECIST criteria. Among RECIST-evaluable patients in cohort A (n=20), the objective response rate was 35% (95% CI: 15–59%), and the 6-month progression-free survival rate was 64% (95% CI: 49–82%), compared with 47% in cohort B and 13% in cohort C. These results did not meet the predefined primary endpoints. At a median follow-up of 37 months (95% CI: 27–47), outcomes were durable in cohort A.
Despite this, clinically meaningful activity was observed. Median progression-free survival in cohort A was 8.3 months (95% CI: 5.3–not reached), and median overall survival reached 28 months (95% CI: 12–not reached). Long-term outcomes were notable, with 2-year and 3-year overall survival rates of 56% and 44%, respectively.
Importantly, a subgroup of patients demonstrated particularly durable benefit, with 7.9% of patients achieving progression-free survival beyond 36 months. When including patients with non-measurable disease at baseline—many of whom had already experienced deep responses to platinum chemotherapy— the exploratory response rate increased to 52% (95% CI: 34–69%), and prolonged disease control was observed in several patients.
Across cohorts, outcomes followed a clear biomarker gradient. In cohort B, the objective response rate was 8%, with a median progression-free survival of 4.8 months and median overall survival of 18 months. In cohort C, which included HRD-negative patients, outcomes were more limited, with an objective response rate of 14%, median progression-free survival of 3.3 months, and median overall survival of 10 months. Baseline CA19-9 was independently associated with progression-free survival, with a hazard ratio of 1.02 (95% CI: 1.02–1.03).
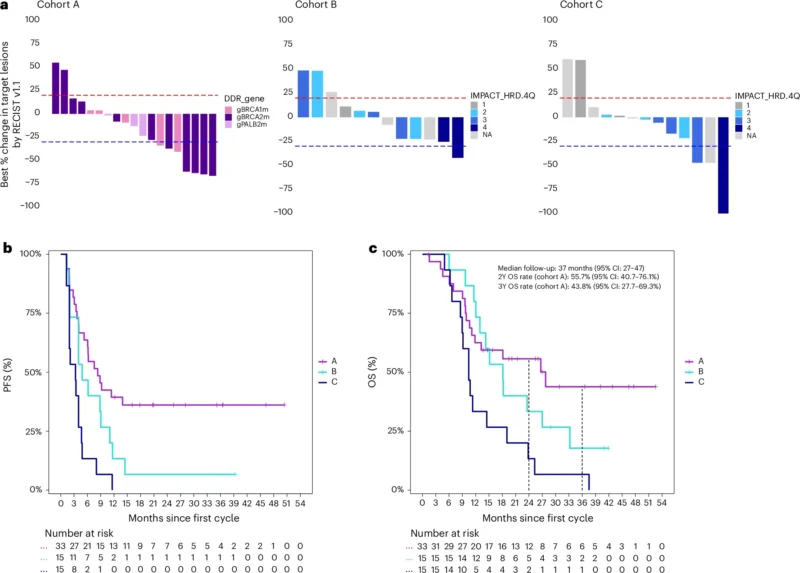
Disease control was achieved in the majority of patients, particularly in cohort A, where the disease control rate reached 80%, compared with 75% in cohort B and 50% in cohort C. Among responders in cohort A, the median duration of response was 6.8 months, while the median duration of disease control reached 32 months when including patients with non-measurable disease.
Subgroup analyses within cohort A revealed heterogeneity according to mutation type. Patients with BRCA2 and PALB2 mutations demonstrated longer progression-free and overall survival compared with those harboring BRCA1 mutations. Median progression-free survival was 9.9 months for BRCA2, 12.0 months for PALB2, and 6.1 months for BRCA1. Median overall survival was 28.0, 27.0, and 18.0 months, respectively.
Translational insights
The study incorporated an extensive translational program that provided important biological insights. Circulating tumor DNA analyses showed that patients with durable benefit often had very low or undetectable variant allele frequencies, whereas increases in ctDNA were associated with shorter progression-free survival.
Tumors in cohort A exhibited a more immunogenic profile, including higher frameshift indel burden, increased tumor mutational burden, and higher genomic instability scores. These features were associated with increased tumor-infiltrating lymphocytes, particularly CD8+ T cells, which correlated with improved clinical outcomes.
Together, these findings support the hypothesis that HRD tumors may be inherently more immunogenic and therefore more susceptible to combined PARP inhibition and immune checkpoint blockade.
Safety
The combination was generally well tolerated, with no new safety signals. No grade 4 or 5 treatment-related adverse events were reported.
The most common grade 3 treatment-related adverse event was anemia, occurring in 15% of patients, along with rare abdominal infection (1.6%). Immune-related adverse events included pneumonitis, colitis, hyperthyroidism, and hyperglycemia, with grade 3 immune toxicities observed in a small number of patients.
Expert Highlights
The POLAR trial contributes to a growing shift in how pancreatic cancer is conceptualized, moving away from the idea of a uniformly immunotherapy-resistant disease toward a more nuanced, biology-driven approach.
Wungki Park, a clinician-scientist at Memorial Sloan Kettering Cancer Center and the Gerstner Sloan Kettering Graduate School of Biomedical Sciences, focusing on next-generation therapies in GI cancers, including KRAS-directed strategies and immune reprogramming, highlights:
“The POLAR study was designed around the idea that pancreatic cancer should not be approached as a uniformly immunotherapy-resistant disease. Instead, we focused on whether a biologically defined subset, specifically homologous recombination-deficient tumors, may have increased susceptibility when treated in the right clinical context, particularly in the maintenance setting after platinum response. In that sense, I see POLAR as contributing to a shift toward more biomarker-driven and context-specific strategies in metastatic pancreatic cancer.”

Beyond the clinical outcomes, the study also provides an important conceptual framework for future treatment strategies, emphasizing the role of tumor biology in guiding therapeutic decisions rather than broadly applying immunotherapy across all patients.
“From a practice and field perspective, the key insight is not just the clinical signal itself, but the framework it supports. The data suggest that genomic features such as HRD may be linked to underlying tumor immunogenicity, which opens the door to more rational combinations and sequencing strategies, not broadly applying immunotherapy across all patients. It reinforces the importance of aligning treatment with tumor biology and continuing to refine patient selection.”
Conclusion
Although the POLAR trial did not meet its prespecified primary endpoints, it demonstrated meaningful and durable clinical activity in a subset of patients with HRD metastatic pancreatic cancer.
The results suggest that combining PARP inhibition with PD-1 blockade may extend disease control beyond chemotherapy in biologically selected patients, particularly those with BRCA2 and PALB2 mutations.
These findings reinforce the importance of biomarker-driven strategies and support further investigation of precision immunotherapy approaches in pancreatic cancer.
The full article is available in Nature Medicine.