Neoadjuvant treatment has transformed the management of early HER2-positive breast cancer and triple-negative breast cancer, not only by increasing the chance of breast conservation and less extensive axillary surgery, but also by giving clinicians a real-time readout of treatment sensitivity through pathologic response. For years, pathologic complete response has been recognized as a favorable marker in these biologically aggressive subtypes. But an important clinical question has remained: once a patient achieves pathologic complete response, do baseline factors such as tumor size, nodal status, and subtype still matter? And conversely, in patients with residual disease, how much prognostic weight do these baseline features continue to carry?
A new retrospective analysis takes a closer look at this issue in a modern cohort of patients with early HER2-positive breast cancer and triple-negative breast cancer treated with neoadjuvant therapy. The study shows that achieving pCR remains strongly associated with excellent outcomes, while baseline clinical factors appear to retain prognostic value primarily in patients who do not achieve pCR (Corti et al., 2026).
Why This Study Matters
In current practice, neoadjuvant therapy plays a central role in both HER2-positive breast cancer and triple-negative breast cancer. Beyond tumor downstaging, the neoadjuvant approach helps guide post-neoadjuvant therapy. In HER2-positive disease, patients with residual disease may receive adjuvant trastuzumab emtansine (T-DM1) based on the KATHERINE trial, while in triple-negative breast cancer residual disease can support consideration of capecitabine, and in select germline BRCA1/2 carriers, olaparib may also be used (von Minckwitz et al., 2019; Masuda et al., 2017; Tutt et al., 2021). In addition, neoadjuvant pembrolizumab-based regimens have further changed the treatment landscape in stage II–III triple-negative breast cancer after KEYNOTE-522 (Schmid et al., 2020; Schmid et al., 2022; Schmid et al., 2024).
Because treatment intensification after surgery is increasingly driven by the presence or absence of residual disease, understanding how pCR interacts with initial stage has become more than an academic question. Recent studies have suggested that even among patients who achievepathologic complete response, initial stage may still influence prognosis, although the magnitude of that effect remains uncertain (van Mackelenbergh et al., 2023; Carroll et al., 2024).
Study Design and Population
This was a single-center retrospective study including patients with early HER2-positive breast cancer or triple-negative breast cancer who underwent surgery after neoadjuvant treatment. The investigators included patients with available data on pathologic response, recurrence-free survival, and overall survival. Pathologic complete response was defined as ypT0/is, ypN0, while all other outcomes were categorized as residual disease (Corti et al., 2026).
A total of 863 patients were included. Median age at surgery was 50.2 years, and median follow-up was 3.5 years. Most patients had stage II disease at diagnosis, and the majority of tumors were high grade, particularly among triple-negative breast cancers. Overall, 43.3% of patients achievedpathologic complete response, while 56.7% had residual disease after neoadjuvant treatment (Corti et al., 2026).
Among subtypes, pCR rates were not equal. The highest pCR rate was seen in HR-negative/HER2-positive tumors at 71.8%, while the lowest was observed in triple-negative breast cancer at 32.3%. This is consistent with the biologic sensitivity of HER2-positive tumors to modern HER2-directed treatment and the persistent challenge of triple-negative disease despite contemporary neoadjuvant strategies (Corti et al., 2026).
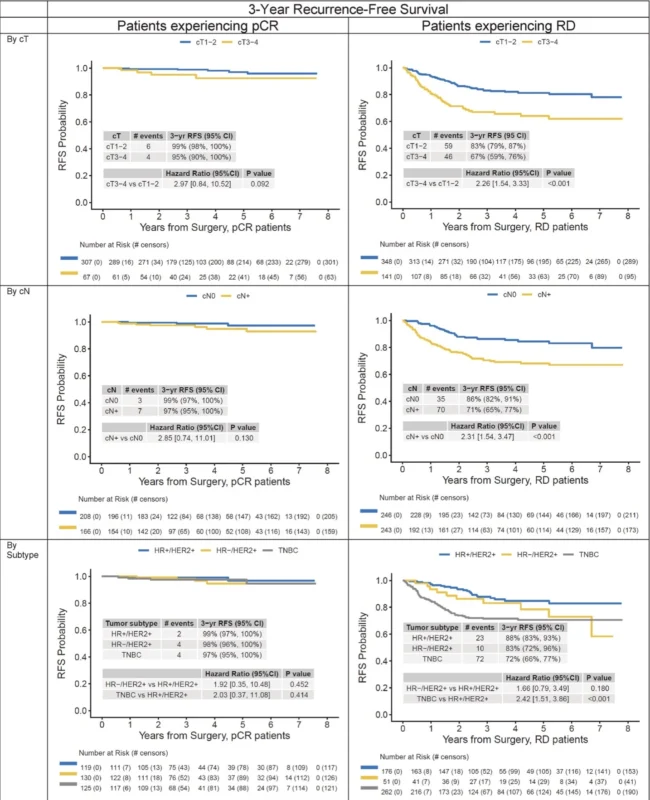
Pathologic Complete Response Was Associated with Outstanding Outcomes
The clearest message from this study is that patients who achievedpathologic complete response had excellent outcomes, regardless of subtype.
Across the full cohort, 3-year recurrence-free survival was 98% in patients with pCR, compared with 79% in those with residual disease. The separation was striking within each major subtype. In HER2-positive breast cancer, 3-year recurrence-free survival was 99% with pCR versus 87% with residual disease. In triple-negative breast cancer, it was 97% with pCR versus 72% with residual disease (Corti et al., 2026).
The same pattern held for overall survival. Across the entire cohort, 3-year overall survival was 100% in the pCR group and 88% in the residual disease group. In triple-negative breast cancer, 3-year overall survival was 99% among patients with pCR and 81% among those with residual disease. In HER2-positive disease, survival remained exceptionally high in both groups, but still favored patients with pCR, with 100% versus 96% 3-year overall survival (Corti et al., 2026).
These numbers reinforce what clinicians already appreciate in practice: in aggressive early breast cancer, pCR remains one of the strongest favorable prognostic signals available.
Residual Disease Identified the Patients Whose Baseline Risk Still Mattered
The most clinically important nuance of the analysis emerged when the investigators separated patients according to pathologic response.
In the overall cohort, larger clinical tumor size, positive nodal status, triple-negative subtype, and residual disease were all associated with worse recurrence-free survival and overall survival. However, when patients were stratified by pathologic response, those associations largely disappeared in the pCR group and remained concentrated in the residual disease group (Corti et al., 2026).
Among patients with pCR, recurrence-free survival was numerically lower in those with cT3–4 disease than in those with cT1–2 disease, and also numerically lower in node-positive versus node-negative patients, but these differences were not statistically significant. No significant associations were seen by subtype either. Similarly, for overall survival, patients with pCR had uniformly excellent outcomes regardless of clinical tumor size, nodal status, or subtype (Corti et al., 2026).
That is an important observation. It suggests that once a patient reaches pCR, the negative impact of presenting with larger tumors or nodal involvement may be substantially attenuated, at least within the follow-up available in this study.
By contrast, in patients with residual disease, the baseline risk factors remained highly relevant. Larger tumor size, positive nodes, and triple-negative subtype were all associated with significantly worse recurrence-free survival and overall survival. On multivariable analysis, among patients with residual disease, cT3–4 disease, node-positive disease, and triple-negative subtype all remained independently associated with poorer outcomes (Corti et al., 2026).
This means that residual disease is not simply a binary marker of incomplete response. It identifies a subgroup in which baseline disease burden and tumor biology continue to shape recurrence risk in a meaningful way.
What This Means for HER2-Positive Disease
For patients with HER2-positive breast cancer, the data are reassuring in those who achieve pCR. Their 3-year recurrence-free survival approached 99%, and overall survival was essentially 100% over the observed follow-up. These results align with prior pooled evidence showing that pCR after neoadjuvant anti-HER2 therapy is associated with excellent outcomes, while residual disease identifies a population at higher risk that can benefit from escalation with T-DM1 (van Mackelenbergh et al., 2023; von Minckwitz et al., 2019).
The current study adds to that framework by suggesting that in HER2-positive disease, once pCR is achieved, initial clinical stage may matter less than previously feared. But in those with residual disease, baseline clinical burden still helps define prognosis.
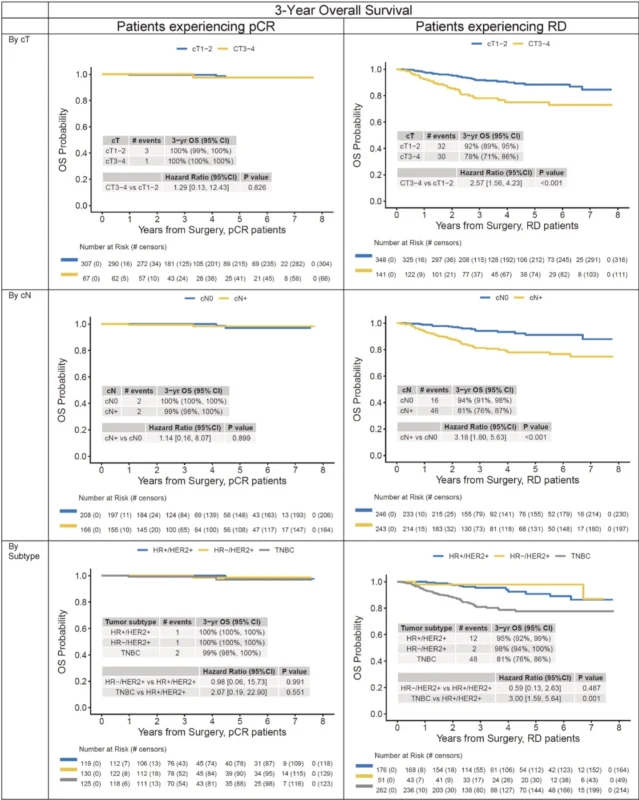
What This Means for Triple-Negative Breast Cancer
In triple-negative breast cancer, the distinction between pCR and residual disease was even more dramatic. Patients with pCR had a 3-year recurrence-free survival of 97% and overall survival of 99%, compared with 72% and 81%, respectively, in patients with residual disease (Corti et al., 2026).
This underscores how central pCR remains in triple-negative breast cancer, even in the modern era. It also reinforces the importance of post-neoadjuvant escalation strategies in patients with residual disease, where prognosis remains substantially worse. These findings fit well with the current treatment model in TNBC, where pembrolizumab-based neoadjuvant therapy, adjuvant pembrolizumab continuation, capecitabine, and olaparib all play a role in selected high-risk patients (Schmid et al., 2020; Schmid et al., 2024; Masuda et al., 2017; Tutt et al., 2021).
Important Limitations
The authors appropriately note several limitations. This was a retrospective single-center study, which introduces inherent bias. Follow-up was relatively short at 3.5 years, which is especially relevant in HER2-positive and hormone receptor-positive/HER2-positive disease where later events can still occur. The study also could not fully account for all post-neoadjuvant treatments received over time, nor did it systematically capture all early discontinuations of neoadjuvant therapy due to toxicity or tolerability (Corti et al., 2026).
In addition, because the event count in the pCR group was low, the study may have been underpowered to detect smaller but real differences according to stage among patients who achieved pCR. So while the findings suggest that baseline factors are no longer strongly prognostic after pCR, larger datasets and longer follow-up will be needed to confirm how complete that risk attenuation truly is.
Final Takeaway
This study delivers a practical message for clinicians treating early HER2-positive breast cancer and triple-negative breast cancer: pathologic complete response remains a powerful marker of excellent prognosis, and in patients who achieve pCR, baseline clinical stage may lose much of its prognostic weight. In contrast, for patients with residual disease, tumor size, nodal status, and subtype still matter and continue to define risk after surgery (Corti et al., 2026).
That makes residual disease more than a postoperative finding. It is a biologically meaningful signal that identifies patients who remain vulnerable, and it reinforces why post-neoadjuvant escalation strategies are so important in modern breast oncology.
Full article here